* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Metal-Metal Bonds
Evolution of metal ions in biological systems wikipedia , lookup
Hydroformylation wikipedia , lookup
Metalloprotein wikipedia , lookup
Metal carbonyl wikipedia , lookup
Bond valence method wikipedia , lookup
Cluster chemistry wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Jahn–Teller effect wikipedia , lookup
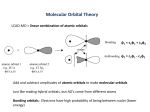
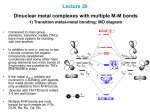
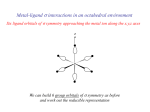
Metal-Metal Bonds Chapter 15 Metal-Metal Bonds • Single, double, triple, and quadruple bonds are possible in transition metal complexes. – Figure 15-7; [Re3Cl12]3- and [Re2Cl8]2– The metal-metal bond distance in the dimer is 224 pm. It was the first complex found to have a quadruple bond. Look at other complexes that have metal-metal bonds. Possible Bonding Interaction between Metal d Orbitals • Discuss the possible bonding interactions between the two metal atoms. – d orbital interactions. • The interaction between dx2-y2 and dxy orbitals is weak. Why? – This interactions produces the bonding. • Occupancy of the molecular orbitals contributes only a small amount to the overall bonding. – Figure 15.9 in book. • What about the spacing between and *? Possible Bonding Interaction in a Complex Containing M-M Bonds • In an actual complex (e.g. [Re2Cl8]2-) the ligands interact with the metal orbitals. • What is the symmetry of this molecule? • With which orbital combination do the ligands interact most strongly? • LGO treatment (ligands pointing toward the metal atoms). • Interaction occur with multiple orbital combinations on the metal but the strongest is with the MO’s (bonding and antibonding) – Let’s look at the IR’s. Possible Bonding Interaction in a Complex Containing M-M Bonds Possible Bonding Interaction in a Complex Containing M-M Bonds • New molecular orbitals form when including the ligand interactions. – Simplistic picture showing interaction only between the ligands and certain d orbital combinations on the metal atoms. • Why isn’t the complex staggered? What will rotation do to the energy of the MO’s? Possible Bonding Interaction in a Complex Containing M-M Bonds • Compounds of the staggered conformation. – What is the symmetry of [Os2Cl8]2-? – Why is it staggered and why does it possess a lower bond order? • Formal shortness ratio. – Table