* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Amino Acids Found in Proteins
Survey
Document related concepts
Transcript
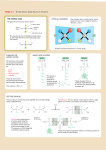
Amino Acids Found in Proteins - Part 1 ©1998 by Alberts, Bray, Johnson, Lewis, Raff, Roberts, Walter . http://www.essentialcellbiology.com Published by Garland Publishing, a member of the Taylor & Francis Group. FAMILIES OF AMINO ACIDS BASIC SIDE CHAINS The common amino acids are grouped according to whether their side chains are acidic basic uncharged polar nonpolar lysine arginine histidine (Lys, or K) (Arg, or R) (His, or H) H O N C C H CH2 H O N C C H CH2 CH2 CH2 + NH3 Thus: alanine = Ala = A THE AMINO ACID C R CH2 C HC NH NH2 2N CH NH+ These nitrogens have a relatively weak affinity for an H+ and are only partly positive at neutral pH. C +H The a -carbon atom is asymmetric, which allows for two mirror image (or stereo-) isomers, L and D. The general formula of an amino acid is amino group H2N C H HN CH2 OPTICAL ISOMERS H O C CH2 This group is very basic because its positive charge is stabilized by resonance. CH2 These 20 amino acids are given both three-letter and one-letter abbreviations. H N a -carbon atom carboxyl COOH group COO– NH3+ COO– NH3+ side-chain group L R is commonly one of 20 different side chains. At pH 7 both the amino and carboxyl groups are ionized. H + H3N C COO Ca D Ca R H R H Proteins consist exclusively of L-amino acids. R PEPTIDE BONDS Amino acids are commonly joined together by an amide linkage, called a peptide bond. H H N H C R O C N OH H H2O R H C H Peptide bond: The four atoms in each gray box form a rigid planar unit. There is no rotation around the C–N bond. O H C N OH H H O C C R R N C H H O C OH SH Proteins are long polymers of amino acids linked by peptide bonds, and they are always written with the N-terminus toward the left. The sequence of this tripeptide is histidine-cysteine-valine. amino, or N-, terminus +H N 3 H O C C CH2 HC N H C C O H H N C CH NH+ carboxyl, or C-, terminus COO– CH CH3 C HN CH2 CH3 These two single bonds allow rotation, so that long chains of amino acids are very flexible.