* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 4
Self-assembled monolayer wikipedia , lookup
High-temperature superconductivity wikipedia , lookup
State of matter wikipedia , lookup
Electromigration wikipedia , lookup
Shape-memory alloy wikipedia , lookup
Geometrical frustration wikipedia , lookup
Paleostress inversion wikipedia , lookup
X-ray crystallography wikipedia , lookup
History of metamaterials wikipedia , lookup
Low-energy electron diffraction wikipedia , lookup
Condensed matter physics wikipedia , lookup
Nanochemistry wikipedia , lookup
Radiation damage wikipedia , lookup
Electronic band structure wikipedia , lookup
Tight binding wikipedia , lookup
Semiconductor wikipedia , lookup
Work hardening wikipedia , lookup
Crystallographic defects in diamond wikipedia , lookup
Colloidal crystal wikipedia , lookup
Dislocation wikipedia , lookup
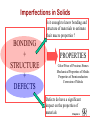
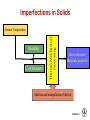
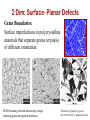
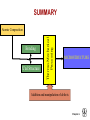
Solidification & Crystalline Imperfections Chapter 4- Today’s Agenda Processing Structure Properties Ex: casting Atomic/Molecular St. Bond structure Crystal Structure Defect Structures Microstructure Energy Band Structure Mechanical Electrical Optical Thermal Magnetic Performance Cost Reliability Efficiency Service Life ... Melt SOLIDIFICATION Chapter 4- Solidification of Metals • The solidification of metals and alloys is an important industrial process since most metals are melted and then cast into semifinished of finished shape. • When molten alloys are cast, solidification starts at the wallls of the mold Chapter 4- 1. Solidification of Polycrystalline Material • Almost all engineering crystalline materials are composed of many crystals. • Two steps of solidification: 1. Nucleation: Formation of stable nuclei in the melt 2. Growth: Crystals grow until they meet each other Liquid Nuclei Crystals that will form grains Grains Grain boundaries Chapter 4- 2. Solidification of Single Crystals There are also materials that consist of only one crystal. Examples: • High-temperature creep-resistant gas turbine blades (At high temperatures, grain boundaries become weaker than the grain bodies) • Silicon single crystals for semiconductor devices. (Grain boundaries would disrupt the flow of electrons in devices) Chapter 4- 3. Metallic Solid Solutions • Most engineering metals are combined with other metals or nonmetals to provide increased strength, higher corrosion resistance or other desired properties «METAL ALLOY» • Simplest type of alloy = Solid Solution: A solid that consists of two or more elements atamically dispersed in a single-phase structure. Two types of solid solutions: Substitutional alloy (e.g., Cu in Ni) Interstitial alloy (e.g., C in Fe) OR Chapter 4- Conditions for Substitutional Solid Solutions • W. Hume – Rothery rule 1. r (atomic radius) < 15% 2. Proximity in periodic table • i.e., similar electronegativities 3. Same crystal structure for pure metals 4. Valency • All else being equal, a metal will have a greater tendency to dissolve a metal of higher valency than one of lower valency Chapter 4- Second Phase • While solute atoms are being added, new compounds / structures may form beyond solubility limit, or solute forms local PRECIPITATES. • Nature of the impurities, their concentration, reactivity, temperature and pressure, etc decides the formation of solid solution or a second phase. Solid solution of B in A plus particles of a new phase (usually for a larger amount of B) Second phase particle --different composition --often different structure. Chapter 4- CRYSTALLINE IMPERFECTIONS Why are we interested IMPERFECTIONS IN SOLIDS ? “Crystals are like people, it is the defects in them which tend to make them interesting!” - Colin Humphreys. Crystals in nature are never perfect, they have defects ! Chapter 4- 1 Imperfections in Solids Is it enough to know bonding and structure of materials to estimate their macro properties ? BONDING + STRUCTURE + DEFECTS PROPERTIES Color/Price of Precious Stones Mechanical Properties of Metals Properties of Semiconductors Corrosion of Metals Defects do have a significant impact on the properties of materials Chapter 4- Imperfections in Solids Bonding X’tal Structure Thermo-Mechanical Processing Atomic Composition Microstructure: Materials properties Addition and manipulation of defects Chapter 4- Perfection… In terms of: 1. Chemical composition – pure 2. Atomic arrangement – defect free • • • Both are critical in determining the performance of material. Real engineering materials are not perfect. Properties can be altered through defect engineering. 12 Chapter 4- Classification of Defects • • • • The defects are classified on the basis of dimensionality: 0-dimensional: point defects 1-dimensional: line defects 2-dimensional: interfacial defects 3-dimensional: bulk defects 13 Chapter 4- 0 dim: Point Defects - localized disruption in regularity of the lattice on and between lattice sites 3 Types: 1. Substitutional Impurity - occupies normal lattice site - dopant ☺, e.g., P in Si - contaminant Li+ in NaCl Interstitial 2. Interstitial Impurity - occupies position between lattice sites - alloying element ☺, e.g., C in Fe - contaminant, H in Fe 3. Vacancy - unoccupied lattice site - formed at time of crystallization Vacancy Substitutional Self-interstitial 14 Chapter 4- • Vacancies: POINT DEFECTS -vacant atomic/lattice sites in a structure. distortion of planes Vacancy • Self-Interstitials: -"extra" atoms positioned between atomic sites. distortion of planes selfinterstitial Chapter 4- 3 Population of vacancies in a crystal • In a crystal containing N atomic sites, the number nd of vacant sites: nd = the number of defects (in equilibrium at T) N = the total number of atomic sites per mole ΔHd = the energy necessary to form the defect T = the absolute temperature (K) k = the Boltzmann constant A = proportionality constant 16 Chapter 4- EQUIL. CONCENTRATION: POINT DEFECTS • Equilibrium concentration varies with temperature! No. of defects Activation energy Q ND exp D kT N No. of potential Temperature defect sites. Boltzmann's constant (1.38 x 10-23 J/atom K) (8.62 x 10 -5 eV/atom K) Each lattice site is a potential vacancy site Chapter 4- 4 ESTIMATING VACANCY CONC. 3 • Find the equil. # of vacancies in 1m of Cu at 1000C. • Given: 0.9eV/atom Q ND exp D = 2.7 · 10-4 kT N For 1m 3, N = 1273K 8.62 x 10-5 eV/atom-K NA x 1m3 = 8.0 x 1028 sites x ACu • Answer: Chapter 4- Point Defects: Vacancies & Interstitials • Most common defects in crystalline solids are point defects. • At high temperatures, atoms frequently and randomly change their positions leaving behind empty lattice sites. • In general, diffusion (mass transport by atomic motion) - can only occur because of vacancies. Chapter 4- OBSERVING EQUIL. VACANCY CONC. • Low energy electron microscope view of a (110) surface of NiAl. • Increasing T causes surface island of atoms to grow. • Why? The equil. vacancy conc. increases via atom motion from the crystal to the surface, where they join the island. Reprinted with permission from Nature (K.F. McCarty, J.A. Nobel, and N.C. Bartelt, "Vacancies in Solids and the Stability of Surface Morphology", Nature, Vol. 412, pp. 622-625 (2001). Image is 5.75 mm by 5.75 mm.) Copyright (2001) Macmillan Publishers, Ltd. Question: Where do vacancies go ? Chapter 4- 7 Point Defects in Ionic Crystals Maintain global charge neutrality 1. Schottky Imperfection formation of equivalent (not necessarily equal) numbers of cationic and anionic vacancies 2. Frenkel Imperfection formation of an ion vacancy and an ion interstitial 21 Chapter 4- Exercise on Point Defects Schematic representation of a variety of point defects: (1) vacancy; ANSWER (2) self-interstitial; Ei > Ev , so ? (3) interstitial impurity;less distortion caused (4,5) substitutional impurities The arrows represent the local stresses introduced by the point defects. Chapter 4- 1 Dimensional: Line Defects Dislocations • Dislocations are linear defects: the interatomic bonds are distorted only in the vicinity of the dislocation line. This area is called the dislocation core. • Dislocations also create small elastic deformations of the lattice at large distances. Chapter 4- 23 DISLOCATIONS •Material permanently deforms as dislocation moves through the crystal. • Bonds break and reform, but only along the dislocation line at any point in time, not along the whole plane at once. • Dislocation line separates slipped and unslipped material. Chapter 4- LINE DEFECTS Dislocations: • are line defects, • cause slip between crystal plane when they move, • produce permanent (plastic) deformation. Schematic of a Zinc Crystal (HCP): • before deformation • after tensile elongation slip steps Chapter 4- 11 Dislocations and Materials Strength Easily form dislocations and allow mobility; Not limited with coordination numbers Remember Covalent Bond ! How many bonds to break ? Finding an equivalent site ? Very large Burgers vector size; Finding an equivalent site and overcoming repulsive forces ! Chapter 4- 2 Dim: Surface- Planar Defects Grain Boundaries: Surface imperfections in polycrystalline materials that separate grains (crystals) of different orientation. SEM (Scanning electron microscope) image (showing grains and grain boundaries) Photomicrographs 27 of typical Chapter 4microstructures of annealed brass Polycrystalline Materials Grain Boundaries • regions between crystals • transition from lattice of one region to that of the other • slightly disordered • low density in grain boundaries – high mobility – high diffusivity – high chemical reactivity Adapted from Fig. 4.7, Callister 7e. Chapter 4- AREA DEFECTS: GRAIN BOUNDARIES Grain boundaries: • • • • are boundaries between crystals. are produced by the solidification process, for example. have a change in crystal orientation across them. impede dislocation motion. Metal Ingot Schematic ~ 8cm grain boundaries Adapted from Fig. 4.7, Callister 6e. heat flow Adapted from Fig. 4.10, Callister 6e. (Fig. 4.10 is from Metals Handbook, Vol. 9, 9th edition, Metallography and Microstructures, Am. Society for Metals, Metals Park, OH, 1985.) Chapter 4- 15 3 Dim: Volume Defects • Form when a cluster of point defects join F. Gao,J. Qu, Materials Letters Volume 73, 15 Apr.2012, 92–94 • Vacancies join: – voids – Pores • Cracks • Cluster of impurity atoms join precipitate Chapter 4- Experimental Techniques for Identification of Microstructure and Defects Chapter 4- Optical Microscopy • Useful up to 2000X magnification. • Polishing removes surface features (e.g., scratches) • Etching changes reflectance, depending on crystal orientation. crystallographic planes Adapted from Fig. 4.13(b) and (c), Callister 7e. (Fig. 4.13(c) is courtesy of J.E. Burke, General Electric Co. Micrograph of brass (a Cu-Zn alloy) 0.75mm Chapter 4- Microscopy Optical resolution ca. 10-7 m = 0.1 m = 100 nm For higher resolution need higher frequency – X-Rays? Difficult to focus. – Electrons • wavelengths ca. 3 pm (0.003 nm) – (Magnification - 1,000,000X) • Atomic resolution possible • Electron beam focused by magnetic lenses. Chapter 4- Scanning Tunneling Microscopy (STM) • Atoms can be arranged and imaged! Photos produced from the work of C.P. Lutz, Zeppenfeld, and D.M. Eigler. Reprinted with permission from International Business Machines Corporation, copyright 1995. Carbon monoxide molecules arranged on a platinum (111) surface. Iron atoms arranged on a copper (111) surface. These Kanji characters represent the word “atom”. Chapter 4- SUMMARY Bonding X’tal Structure Thermo-Mechanical Processing Atomic Composition MICROSTRUCTURE Addition and manipulation of defects Chapter 4- Disorder Single Crystal Polycrystalline Amorphous Grain boundaries Grains 36 Chapter 4- Summary • Point, Line, and Area defects exist in solids. • The number and type of defects can be varied and controlled (e.g., T controls vacancy conc.) • Defects affect material properties (e.g., grain boundaries control crystal slip). • Defects may be desirable or undesirable (e.g., dislocations may be good or bad, depending on whether plastic deformation is desirable or not.) Chapter 4-



































!["[Photographer`s name]/[Collection Name]/Getty Images" or as](http://s1.studyres.com/store/data/010577066_1-1c512b5ed79bdb618cc6f2911d80860a-150x150.png)






