* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Topic 2.3 The Atom Electron Configuration
Survey
Document related concepts
Transcript
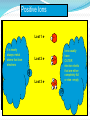
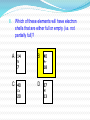
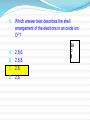
Assessment Objectives 2.3.4 Deduce the electron arrangement for atoms and ions up to Z = 20. Key Terms Electron Electron Configuration Electron Shell Ion Cation Anion References Textbook on moodle Starter What are the symbols for the following elements: Sulphur Phosphorous Chlorine Aluminium Berilium Potassium Starter Activity Write the answers to the following in the back of your book. Name 3 sub-atomic particles and write down as much information about each one as you can remember. What can you say about the numbers of protons and electrons in an atom? Assessment Objective 2.3.4 Deduce the electron arrangement for atoms and ions up to Z = 20. History 1913 - Danish physicist Niels Bohr realised that the secret of atomic structure lay in its discreteness, that energy could only be absorbed or emitted at certain values. The Bohr Model Bohr realised that the electrons could only be at specific energy levels (or states) around the atom. Models of Atoms This property of atoms can be represented by a number of models which helps us explain how chemical reactions work. (HL students will learn about another one) How Are Electrons Arranged? Electrons are not evenly spread. The exist in layers known as shells. The arrangement of electrons in these shells is often called the electron configuration. 1st Shell 2nd Shell 3rd Shell 4th Shell How Many Electrons per Shell? Each shell has a maximum number of electrons that it can hold. The maximum 1st Shell: 2 electrons 2nd Shell: 8 electrons 3rd Shell: 8 electrons Which Shells Do Electrons go into? Opposites attract. Protons are + and electrons are – charged. Electrons will occupy the shells nearest the nucleus unless these shells are already full. 1st Shell: Fills this first 2nd Shell: Fill this next 3rd Shell: And so on Working Out Electron Arrangements 1. How many electrons do the element atoms have? (This will equal the atomic number). 2. Keeping track of the total used, feed them into the shells working outwards until you have used them all up. Drawing neat diagrams helps you keep track! 1st Shell: Fills this first 2nd Shell: Fill this next Electron Shells Electrons occupy shells around the nucleus of an atom. Each shell can hold different numbers of electrons before it is full. 1st shell can hold 2 electrons 2nd shell can hold 8 electrons 3rd shell can hold 8 electrons 4th shell can hold 18 electrons The shells MUST be filled up from the centre outwards. The Electrons in Carbon The Electrons in Neon The Electrons in Silicon Electrons in Phosphorus The Electrons in Argon The Electrons in Sodium The Electrons in Fluorine The Electrons in Aluminium The Electrons in Nitrogen The Electrons in Sulfur The Electrons in Oxygen The Electrons in Chlorine The Electrons in Magnesium Ions and Electron Structures 1. Ions are atoms that have either extra electrons added or electrons removed. e.g. Atoms Protons Electrons Lose 1 electron 1+ ion Gain 1 electron Protons Electrons So in ions the number of electrons no longer equals the number of protons 1- ion Protons Electrons Positive Ions These are ions formed by the atom losing one or more electrons. Lost 1 e- + They are called cations. This is because during Itelectrolysis is nearly Ions usually(Or they move towards the cathode. always metal have because they are “pussytive”!). Lost 2 e atoms that lose 2+ OUTER electrons The charge on the ions is equal to electrons that the atom has lost. Lost 3 e 3+ electron shells that number are either the completely full or else empty In equations the charge is usually shown above and to the right of the symbol. (E.g. Mg2+ ). of The Electrons in a Sodium Ion In the sodium atom Atomic number = number of protons Number of electrons = 11 Na 23 = 11 Na 11 Na+ Bye! Electron lost Electron arrangement: 2.8.1 (Incomplete Shell) Electron arrangement: 2.8 (Full Shells) The Electrons in a Magnesium Ion In the magnesium atom Atomic number = number of protons Number of electrons = 12 24 = 12 Mg 12 Bye! Mg2+ Mg Bye! 2 electrons lost Electron arrangement = 2.8.2 (Incomplete shell) Electron arrangement 2.8 (Full Shells) Activity The Lithium Ion •How many electrons? 3 •How many electrons in the first shell? 2 •How many electrons in the second shell? 1 What electron arrangement? 7 Li 3 2.1 1st Shell = 2: full 2nd Shell = 1: not full How many electrons to lose? 1 Li + New electron arrangement? Include a diagram 2.(0) Bye! Activity The Boron Ion •How many electrons? 5 •How many electrons in the first shell? 2 •How many electrons in the second shell? 3 What electron arrangement? 11 B 5 2.3 1st Shell = 2: full 2nd Shell = 3: not full How many electrons to lose? Bye! Bye! 3 B3+ New electron arrangement? 2.(0) Bye! Negative Ions. These are ions formed by the atom gaining one or more electrons. Gain 1 e They are called anions. This is because during Ions usually anode. have It is nearly always nonelectrolysis they move towards the Gain 2 e metal atoms 2that gain electrons The charge on the ions is equal to electrons that the atom Gain 3 e- - OUTER electron shells the that number are either full has gained.completely or else empty 3- In equations the charge is usually shown above and to the right of the symbol. (E.g.. O2- ). of The Electrons in a Sulphide Ion. In the sulphur atom Atomic number = number of protons Number of electrons = 16 32 = 16 S 16 S2- S 2 electrons gained Electron arrangement: 2.8.6 (incomplete shell) Electron arrangement 2.8.8 (Full shells) The Electrons in a Fluoride Ion. In the fluorine atom Atomic number = number of protons Number of electrons = 9 19 =9 F 9 F2- F 1 electron gained Electron arrangement: 2.8.7 (incomplete shell) Electron arrangement 2.8.8 (Full shells) Activity The Oxide Ion 8 •How many electrons? •How many electrons in the first shell? 2 •How many electrons in the second shell? 6 What electron arrangement? 16 O 8 2.6 1st Shell = 2: full 2nd Shell = 6: not full How many electrons to gain? 2 O2New electron arrangement? 2.8 Activity Drag the words at the top to their correct places in the sentences Activity The following sentences are all mixed up. Drag and drop the Words in red until they are in the correct places Activity Word Check 1. Which of the following is not a sub-atomic particle? A. Proton. B. Isotope. C. Neutron. D. Electron. 2. The element Cobalt has a relative atomic mass of 59 and an atomic number of 27. Which of these is a true statement about each neutral cobalt atom? A. It contains 59 neutrons. B. It contains 27 electrons. C. It contains 32 protons. D. It contains equal numbers of neutrons and electrons. 3. The Periodic Table displays iron as shown below. This indicates that Fe atoms: A. contain 56 neutrons. 56 Fe 26 B. contain 30 electrons. C. contain 26 protons. D. contains more protons than neutrons. 4. Bromine consists of a mixture of two isotopes: Bromine-79 and Bromine-81 Which of the following is true: 80 Br A. Both isotopes contain 35 protons. 35 B. Bromine 79 contains 46 neutrons. C. Bromine 81 contains 44 neutrons. D. Bromine-81 is more reactive than bromine- 79. 5. Natural boron consists of approx. 20% boron-10 and 80% boron-11. What will the relative atomic mass of natural boron will be? ? A. 10.0 B. 11.0 C. 10.5 D. 10.8 B 5 6. Which answer best describes the shell arrangement of the electrons in a sodium atom? 23 Na 11 A. 2,8,1. B. 2,2,7. C. 2,8,8,3. D. 2,8,8,1. 7. Which answer best describes the shell arrangement of the electrons in an oxygen atom? 16 A. 2,8,6. B. 2,8,8. C. 2,8. D. 2,6. O 8 Which of these elements will have electron shells that are either full or empty (i.e. not partially full)? 8. A 14 N B 7 C 40 Ca 20 40 Ar 18 D 27 Al 13 9. Which answer best describes the shell arrangement of the electrons in an oxide ion O2-? 16 A. 2,8,6. B. 2,8,8. C. 2,8. D. 2,6. O 8 Plenary in shells around the Where do we find electrons in an atom? nucleus 2,8,8,18 How many electrons fit into each shell? What symbol do we use to show electrons? X How do we arrange the electrons in the shells? in pairs What do we notice about the XX all full electrons shells of the noble gases? Draw the electron configuration of chlorine XX X X X X X X X X X X X X X Homework Memorise the symbols for the following elements. Silver (Ag) Tin (Sn) Iodine (I) Lead (Pb) Mercury (Hg) Gold (Au)