e-the-quantum-numberssv-2
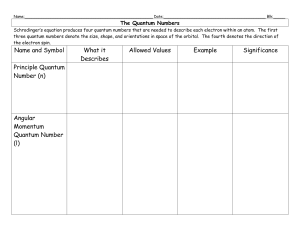
... The Quantum Numbers Schrodinger’s equation produces four quantum numbers that are needed to describe each electron within an atom. The first three quantum numbers denote the size, shape, and orientations in space of the orbital. The fourth denotes the direction of the electron spin. ...
... The Quantum Numbers Schrodinger’s equation produces four quantum numbers that are needed to describe each electron within an atom. The first three quantum numbers denote the size, shape, and orientations in space of the orbital. The fourth denotes the direction of the electron spin. ...
uncertainty: einstein, heisenberg, bohr, and the struggle for the soul
... atoms, influences their absorption and emission of light, and also transports energy between them. In addition, electrons were now to be seen not as orbiting nuclei in the atom, but as "virtual oscillators," each one corresponding to a particular spectroscopic line. However, contrary to classical ph ...
... atoms, influences their absorption and emission of light, and also transports energy between them. In addition, electrons were now to be seen not as orbiting nuclei in the atom, but as "virtual oscillators," each one corresponding to a particular spectroscopic line. However, contrary to classical ph ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 18. Illustrate the Pauli Exclusion Principle for the ground state of He atom. 19. At what distance from the nucleus is the probability of finding the electron a maximum for a 1S electron in hydrogen? 20. While the order is the same for both C3v and C3h point groups, their classes are different reaso ...
... 18. Illustrate the Pauli Exclusion Principle for the ground state of He atom. 19. At what distance from the nucleus is the probability of finding the electron a maximum for a 1S electron in hydrogen? 20. While the order is the same for both C3v and C3h point groups, their classes are different reaso ...
A system consist of two particles,each of which has two possible
... 1. A system consist of two particles,each of which has two possible quantum stats with energies Eo and 2Eo .Write the complete expression for the partition function if: (a) The particle are distinguishable. (b) The particle obey Maxwell-Boltzmann statistics. (c) The particle obey Fermi-Dirac statist ...
... 1. A system consist of two particles,each of which has two possible quantum stats with energies Eo and 2Eo .Write the complete expression for the partition function if: (a) The particle are distinguishable. (b) The particle obey Maxwell-Boltzmann statistics. (c) The particle obey Fermi-Dirac statist ...
dual nature of light
... Diffraction :➢Light bends around the corner but Newton refused this phenomenon. ...
... Diffraction :➢Light bends around the corner but Newton refused this phenomenon. ...
Periodic boundary physics etc
... In physics, specifically quantum mechanics, the Schrödinger equation is an equation that describes how the quantum state of a physical system changes in time. It is as central to quantum mechanics as Newton's laws are to classical mechanics. In the standard interpretation of quantum mechanics, the q ...
... In physics, specifically quantum mechanics, the Schrödinger equation is an equation that describes how the quantum state of a physical system changes in time. It is as central to quantum mechanics as Newton's laws are to classical mechanics. In the standard interpretation of quantum mechanics, the q ...
ExamView Pro
... e. reason why photons are emitted. 6. What is "excluded" by the Pauli exclusion principle? a. certain values of angular momentum. b. precise values of both position and momentum. c. electrons in the same quantum state. d. none of the above. ...
... e. reason why photons are emitted. 6. What is "excluded" by the Pauli exclusion principle? a. certain values of angular momentum. b. precise values of both position and momentum. c. electrons in the same quantum state. d. none of the above. ...
PARTICLE IN AN INFINITE POTENTIAL WELL
... This process may be performed for any other observable. For example, the average momentum or the expectation value of the momentum of a particle in the n-th state of the box. The only difference in the procedure to determine the expectation value of position is to replace the position operator with ...
... This process may be performed for any other observable. For example, the average momentum or the expectation value of the momentum of a particle in the n-th state of the box. The only difference in the procedure to determine the expectation value of position is to replace the position operator with ...
Quantum Mechanics
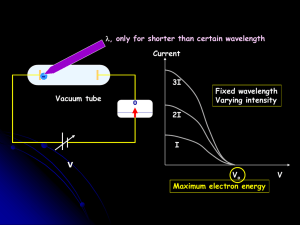
... • Electrons leave the metal with a given energy • Explanation: Photons have a collision with electrons and transfer energy/momentum to the ...
... • Electrons leave the metal with a given energy • Explanation: Photons have a collision with electrons and transfer energy/momentum to the ...
Lecture 1 - UW Canvas
... If we use light with to measure the position of an object, x, its uncertainty, x, cannot be less than ~ because of diffraction. If we use photons with p = h/ to measure the momentum of an object, p, p of the object cannot be less than ~h/ since the photon changes the momentum of the object ...
... If we use light with to measure the position of an object, x, its uncertainty, x, cannot be less than ~ because of diffraction. If we use photons with p = h/ to measure the momentum of an object, p, p of the object cannot be less than ~h/ since the photon changes the momentum of the object ...
quantum1
... •Describes the time evolution of your wavefunction. •Takes the place of Newton’s laws and conserves energy of the system. •Since “particles” aren’t particles but wavicles, it won’t give us a precise position of an individual particle, but due to the statistical nature of things, it will precisely de ...
... •Describes the time evolution of your wavefunction. •Takes the place of Newton’s laws and conserves energy of the system. •Since “particles” aren’t particles but wavicles, it won’t give us a precise position of an individual particle, but due to the statistical nature of things, it will precisely de ...
Dr. Vikram Panchal Institute Of Chemistry CH-2 Worksheet: -2
... 4. A 25 watt bulb emits monochromatic yellow light of wavelength of 0.57m.Calculate the rate of emission of quanta per second. 5. Calculate the energy associated with the first orbit of Li2+. (Z=3 ) 6. Neon gas is generally used in the sign boards. If it emits strongly at 616nm. Calculate (a) The f ...
... 4. A 25 watt bulb emits monochromatic yellow light of wavelength of 0.57m.Calculate the rate of emission of quanta per second. 5. Calculate the energy associated with the first orbit of Li2+. (Z=3 ) 6. Neon gas is generally used in the sign boards. If it emits strongly at 616nm. Calculate (a) The f ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 12. What is a hermitian operator and its significance? Show that eigen functions corresponding to two different eigen values of a hermitian operator are orthogonal. 13. Show that in spherical polar coordinates the operator for the Z-component of angular momentum becomes Lz = -iħδ/δφ. Show that the f ...
... 12. What is a hermitian operator and its significance? Show that eigen functions corresponding to two different eigen values of a hermitian operator are orthogonal. 13. Show that in spherical polar coordinates the operator for the Z-component of angular momentum becomes Lz = -iħδ/δφ. Show that the f ...
Quantum Chemistry - Winona State University
... Postulates of Quantum Theory • The state of a system is defined by a function (usually denoted and called the wavefunction or state function) that contains all the information that can be known about the system. • Every physical observable is represented by a linear operator called the “Hermitian ...
... Postulates of Quantum Theory • The state of a system is defined by a function (usually denoted and called the wavefunction or state function) that contains all the information that can be known about the system. • Every physical observable is represented by a linear operator called the “Hermitian ...
Quantum Physics and Human Affairs
... quantized field is like water in a bucket that, for unknown reasons, must contain either exactly 1 gallon, or 2 gallons, or 3 gallons, etc. of water and never any intermediate amount such as 1.7 gallons. Such a "quantized bucket of water" could gain or lose water only in sudden 1-gallon increments o ...
... quantized field is like water in a bucket that, for unknown reasons, must contain either exactly 1 gallon, or 2 gallons, or 3 gallons, etc. of water and never any intermediate amount such as 1.7 gallons. Such a "quantized bucket of water" could gain or lose water only in sudden 1-gallon increments o ...
Bohr–Einstein debates

The Bohr–Einstein debates were a series of public disputes about quantum mechanics between Albert Einstein and Niels Bohr. Their debates are remembered because of their importance to the philosophy of science. An account of the debates was written by Bohr in an article titled ""Discussions with Einsteinon Epistemological Problems in Atomic Physics"". Despite their differences of opinion regarding quantum mechanics, Bohr and Einstein had a mutual admiration that was to last the rest of their lives.The debates represent one of the highest points of scientific research in the first half of the twentieth century because it called attention to an element of quantum theory, quantum non-locality, which is absolutely central to our modern understanding of the physical world. The consensus view of professional physicists has been that Bohr proved victorious, and definitively established the fundamental probabilistic character of quantum measurement.