5.7 Nuclear Radiation
... • Easily pass through paper, wood, and human body (extremely dangerous). • They can be stopped, although not completely, by several meters of concrete or several centimeters of lead. ...
... • Easily pass through paper, wood, and human body (extremely dangerous). • They can be stopped, although not completely, by several meters of concrete or several centimeters of lead. ...
Radioactivity Notes Day 1 and 2 Apr 23 and Apr 24
... o Beta radiation penetrates better than alpha particles o It takes a thin sheet of aluminum foil to stop a beta particle. ...
... o Beta radiation penetrates better than alpha particles o It takes a thin sheet of aluminum foil to stop a beta particle. ...
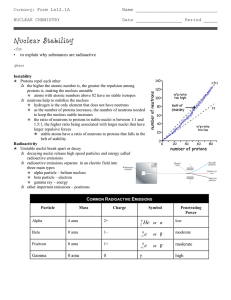
Nuclear Stability
... protons is, making the nucleus unstable p atoms with atomic numbers above 82 have no stable isotopes q neutrons help to stabilize the nucleus p hydrogen is the only element that does not have neutrons p as the number of protons increases, the number of neutrons needed to keep the nucleus stable incr ...
... protons is, making the nucleus unstable p atoms with atomic numbers above 82 have no stable isotopes q neutrons help to stabilize the nucleus p hydrogen is the only element that does not have neutrons p as the number of protons increases, the number of neutrons needed to keep the nucleus stable incr ...
AP Chem
... 2. What is meant by mass defect? 3. Describe how alpha, beta, and gamma rays each behave when passed through an electric field. Draw a picture. 4. Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? ...
... 2. What is meant by mass defect? 3. Describe how alpha, beta, and gamma rays each behave when passed through an electric field. Draw a picture. 4. Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? ...
Download: Worksheet - New York Science Teacher
... b.) Any of the following can be determined given two of the three variables: initial amount of isotope present, the fraction of isotope remaining after a given amount of time, or the half-life of the isotope ...
... b.) Any of the following can be determined given two of the three variables: initial amount of isotope present, the fraction of isotope remaining after a given amount of time, or the half-life of the isotope ...
Ernest Rutherford Essay Research Paper Rutherford was
... can be stopped by a single sheet of paper. Alpha particles electrically charge molecules in the air through which they travel. Beta particles consist of streams of electrons traveling at very high speeds, often approaching the speed of light. They have a mass of 0.000 55 atomic mass unit and a charg ...
... can be stopped by a single sheet of paper. Alpha particles electrically charge molecules in the air through which they travel. Beta particles consist of streams of electrons traveling at very high speeds, often approaching the speed of light. They have a mass of 0.000 55 atomic mass unit and a charg ...
Document
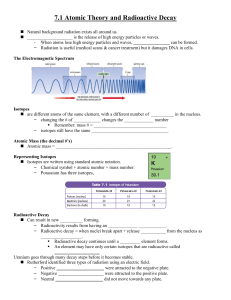
... Radioactive decay continues until a _________ element forms. An element may have only certain isotopes that are radioactive called ____________________ Uranium goes through many decay steps before it becomes stable. Rutherford identified three types of radiation using an electric field. – Posi ...
... Radioactive decay continues until a _________ element forms. An element may have only certain isotopes that are radioactive called ____________________ Uranium goes through many decay steps before it becomes stable. Rutherford identified three types of radiation using an electric field. – Posi ...
Santee Education Complex Chemistry Mini Assessment 11
... Santee Education Complex Chemistry Mini Assessment 11-Nuclear Chemistry Please answer the following questions by choosing the most correct response (1 pt each). 1) Which is most likely to be radioactive? a. Ion of sodium b. Isotope of uranium c. Proton of hydrogen d. Proton of oxygen 2) Which form o ...
... Santee Education Complex Chemistry Mini Assessment 11-Nuclear Chemistry Please answer the following questions by choosing the most correct response (1 pt each). 1) Which is most likely to be radioactive? a. Ion of sodium b. Isotope of uranium c. Proton of hydrogen d. Proton of oxygen 2) Which form o ...
Student 5
... and the conservation atomic number. The number of protons and neutrons remain the same for conservation of Mass number. The charge remains the same, for conservation atomic number. [2] What are the products of nuclear reactions and what are their properties? There are three possible products of nucl ...
... and the conservation atomic number. The number of protons and neutrons remain the same for conservation of Mass number. The charge remains the same, for conservation atomic number. [2] What are the products of nuclear reactions and what are their properties? There are three possible products of nucl ...
PreAP Chemistry Radioactivity WS Name Period ____ Match the
... ---------- 6. The center most part of an atom where the protons and neutrons are contained ...
... ---------- 6. The center most part of an atom where the protons and neutrons are contained ...
Terms to Know
... apart into two roughly equal and smaller parts (lighter nuclei) in a process known as nuclear fission. Gamma radiation : Gamma radiation is one of the three forms of radiation emitted by radioactive materials, the others being alpha and beta radiation. Gamma radiation consists of gamma rays, high en ...
... apart into two roughly equal and smaller parts (lighter nuclei) in a process known as nuclear fission. Gamma radiation : Gamma radiation is one of the three forms of radiation emitted by radioactive materials, the others being alpha and beta radiation. Gamma radiation consists of gamma rays, high en ...
NUCLEAR CHANGES
... What happens when an element undergoes radioactive decay? • During radioactive decay an unstable nuclei of an isotope emits particles and releases energy, to become a stable isotope. ...
... What happens when an element undergoes radioactive decay? • During radioactive decay an unstable nuclei of an isotope emits particles and releases energy, to become a stable isotope. ...
Chinese Civil War, Duck and Cover, and Bomb Shelters
... “Duck and Cover” “Dropping immediately and covering exposed skin provide[s] protection against blast and thermal effects....Immediately drop facedown. A log, a large rock, or any depression in the earth's surface provides some protection. Close eyes. Protect exposed skin from heat by putting hands ...
... “Duck and Cover” “Dropping immediately and covering exposed skin provide[s] protection against blast and thermal effects....Immediately drop facedown. A log, a large rock, or any depression in the earth's surface provides some protection. Close eyes. Protect exposed skin from heat by putting hands ...
Fall Out Shelter Reading Activity and Discussion Questions
... the backyard. The idea was to get as much mass as possible between survivors, the detonation, and its after-effects. Shelter types were: expedience, personal or family, community, multipurpose, and hidden. Usually, an expedience fallout shelter was a trench with a strong roof buried under three feet ...
... the backyard. The idea was to get as much mass as possible between survivors, the detonation, and its after-effects. Shelter types were: expedience, personal or family, community, multipurpose, and hidden. Usually, an expedience fallout shelter was a trench with a strong roof buried under three feet ...
Fallout shelter

A fallout shelter is an enclosed space specially designed to protect occupants from radioactive debris or fallout resulting from a nuclear explosion. Many such shelters were constructed as civil defense measures during the Cold War.During a nuclear explosion, matter vaporized in the resulting fireball is exposed to neutrons from the explosion, absorbs them, and becomes radioactive. When this material condenses in the rain, it forms dust and light sandy materials that resembles ground pumice. The fallout emits alpha and beta particles, as well as gamma rays.Much of this highly radioactive material falls to earth, subjecting anything within the line of sight to radiation, becoming a significant hazard. A fallout shelter is designed to allow its occupants to minimize exposure to harmful fallout until radioactivity has decayed to a safer level.