STOICHIOMETRY:
... The word stoichiometry derives from two Greek words: stoicheion (meaning "element") and metron (meaning "measure"). Stoichiometry deals with calculations about the masses, volumes or concentrations of reactants and products involved in a chemical reaction. The reason we balance chemical reactions is ...
... The word stoichiometry derives from two Greek words: stoicheion (meaning "element") and metron (meaning "measure"). Stoichiometry deals with calculations about the masses, volumes or concentrations of reactants and products involved in a chemical reaction. The reason we balance chemical reactions is ...
MECHANISTIC STUDIES ON THE MONOAMINE OXIDASE B
... carbon-hydrogen bond cleavage takes place. Such a step is normally identified as the "rate-determining step" of the enzymatic reaction. An enzyme catalyzed reaction usually consists of a large number of separate reaction steps of similar energy, the rate of only one of which is affected by isotopic ...
... carbon-hydrogen bond cleavage takes place. Such a step is normally identified as the "rate-determining step" of the enzymatic reaction. An enzyme catalyzed reaction usually consists of a large number of separate reaction steps of similar energy, the rate of only one of which is affected by isotopic ...
Astrochemistry and Star Formation
... probes of physical conditions. Molecules have unique values as probes because (i) their spectra are so rich and diverse that they can be used to understand the details of their surroundings and even heterogeneities in these details, and (ii) chemical processes that both form and destroy molecules yi ...
... probes of physical conditions. Molecules have unique values as probes because (i) their spectra are so rich and diverse that they can be used to understand the details of their surroundings and even heterogeneities in these details, and (ii) chemical processes that both form and destroy molecules yi ...
chemical reactions
... Elements listed higher will displace any elements listed below them. For example Na will displace any elements listed below it from one of its compounds. 2 Na (s) + MgCl2 (aq) 2 NaCl (aq) + Mg (s) Na (s) + AgCl (aq) NaCl (aq) + Ag (s) ...
... Elements listed higher will displace any elements listed below them. For example Na will displace any elements listed below it from one of its compounds. 2 Na (s) + MgCl2 (aq) 2 NaCl (aq) + Mg (s) Na (s) + AgCl (aq) NaCl (aq) + Ag (s) ...
METABOLISM CATABOLISM AND ANABOLISM ATP MOLECULE
... what remains is keto acid and may be converted to pyruvic acid, acetyl-CoA, or one of the acids of the citric acid cycle during shortage of amino acids, citric acid cycle intermediates can be aminated and converted to amino acids in gluconeogenesis, keto acids are used to synthesis glucose ...
... what remains is keto acid and may be converted to pyruvic acid, acetyl-CoA, or one of the acids of the citric acid cycle during shortage of amino acids, citric acid cycle intermediates can be aminated and converted to amino acids in gluconeogenesis, keto acids are used to synthesis glucose ...
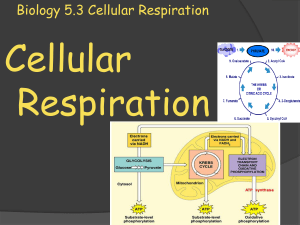
Biology 5.3 Cellular Respiration
... In the first stage of cellular respiration, glucose is broken down to pyruvate during glycolysis. Glycolysis is an anaerobic process (no oxygen required), and it results in a gain of two ATP molecules. In the second stage of cellular respiration, the pyruvate passes through either aerobic respiratio ...
... In the first stage of cellular respiration, glucose is broken down to pyruvate during glycolysis. Glycolysis is an anaerobic process (no oxygen required), and it results in a gain of two ATP molecules. In the second stage of cellular respiration, the pyruvate passes through either aerobic respiratio ...
Lactic acid fermentation
... alcohol; lactic acid Animals and some bacteria employ _________ fermentation, while plants use _________ fermentation. lactic acid; alcohol The point of fermentation, whether alcohol, or lactic acid, is… ATP can be generated, although in moderate amounts. Energy within NADH is used to generate this ...
... alcohol; lactic acid Animals and some bacteria employ _________ fermentation, while plants use _________ fermentation. lactic acid; alcohol The point of fermentation, whether alcohol, or lactic acid, is… ATP can be generated, although in moderate amounts. Energy within NADH is used to generate this ...
Role of Water as a Solvent
... hydroxide and potassium hydrogenphthalate (KHP) to standardize the base solution, by placing 50.00 mg of solid potassium hydrogenphthalate in a flask with a few drops of an indicator. A buret is filled with the base, and the initial buret reading is 0.55 ml; at the end of the titration the buret rea ...
... hydroxide and potassium hydrogenphthalate (KHP) to standardize the base solution, by placing 50.00 mg of solid potassium hydrogenphthalate in a flask with a few drops of an indicator. A buret is filled with the base, and the initial buret reading is 0.55 ml; at the end of the titration the buret rea ...
Oxidative Medicine and Cellular Longevity Special Issue
... Glutathione (GSH), a tripeptide of glycine, cysteine, and glutamate, is a major cellular antioxidant that neutralizes free radicals and reactive oxygen species, detoxifies reactive electrophile products of lipid peroxidation, and maintains the thiol status of proteins. When GSH reacts with oxidants, ...
... Glutathione (GSH), a tripeptide of glycine, cysteine, and glutamate, is a major cellular antioxidant that neutralizes free radicals and reactive oxygen species, detoxifies reactive electrophile products of lipid peroxidation, and maintains the thiol status of proteins. When GSH reacts with oxidants, ...
Covalent Bonding - whitburnscience
... All discrete covalent molecules have low melting and boiling points and tend to be liquids and gases at room temperature. Between all molecules in the liquid or solid state weak forces called van der waals’ forces exist these forces become larger as the size of the molecule increases, it is these fo ...
... All discrete covalent molecules have low melting and boiling points and tend to be liquids and gases at room temperature. Between all molecules in the liquid or solid state weak forces called van der waals’ forces exist these forces become larger as the size of the molecule increases, it is these fo ...
Campbell`s Biology, 9e (Reece et al.)
... Chapter 9 Cellular Respiration and Fermentation 1) What is the term for metabolic pathways that release stored energy by breaking down complex molecules? A) anabolic pathways B) catabolic pathways C) fermentation pathways D) thermodynamic pathways E) bioenergetic pathways Answer: B 3) When electrons ...
... Chapter 9 Cellular Respiration and Fermentation 1) What is the term for metabolic pathways that release stored energy by breaking down complex molecules? A) anabolic pathways B) catabolic pathways C) fermentation pathways D) thermodynamic pathways E) bioenergetic pathways Answer: B 3) When electrons ...
Chem 11 Notes Booklet (pdf version)
... ◘ Note that the number given under the symbol is not the mass number of that particular atom. It is an averaged mass number called the atomic mass and will be used later. 4. Ions a) Creating Ions We know that atoms are electrically neutral because they have equal numbers of protons (p+) and electr ...
... ◘ Note that the number given under the symbol is not the mass number of that particular atom. It is an averaged mass number called the atomic mass and will be used later. 4. Ions a) Creating Ions We know that atoms are electrically neutral because they have equal numbers of protons (p+) and electr ...
Main-group elements as transition metals
... labels. This type of activation by main-group species is general, as shown by the fact that several other unsaturated heavier main-group molecules, including the carbene-like :GeAr2 and :SnAr2 as well as the monovalent :GaAr species, have been recently shown to react directly with H2 (Fig. 3a and b) ...
... labels. This type of activation by main-group species is general, as shown by the fact that several other unsaturated heavier main-group molecules, including the carbene-like :GeAr2 and :SnAr2 as well as the monovalent :GaAr species, have been recently shown to react directly with H2 (Fig. 3a and b) ...
PowerPoint
... hydroxide and potassium hydrogenphthalate (KHP) to standardize the base solution, by placing 50.00 mg of solid potassium hydrogenphthalate in a flask with a few drops of an indicator. A buret is filled with the base, and the initial buret reading is 0.55 ml; at the end of the titration the buret rea ...
... hydroxide and potassium hydrogenphthalate (KHP) to standardize the base solution, by placing 50.00 mg of solid potassium hydrogenphthalate in a flask with a few drops of an indicator. A buret is filled with the base, and the initial buret reading is 0.55 ml; at the end of the titration the buret rea ...
Recovery Following Exercise
... The fast replenishment stage • This is the first component of the oxygen debt that is replenished and requires up to 4 litres of oxygen. •Within this component, the very first amount of oxygen consumed is used to resaturate myoglobin with oxygen. •The rest of the oxygen is required to produce suffi ...
... The fast replenishment stage • This is the first component of the oxygen debt that is replenished and requires up to 4 litres of oxygen. •Within this component, the very first amount of oxygen consumed is used to resaturate myoglobin with oxygen. •The rest of the oxygen is required to produce suffi ...
Chemistry
... describe, interpret and/or predict the effect of different types of bonding (ionic bonding; covalent bonding; hydrogen bonding; other intermolecular interactions; metallic bonding) on the physical properties of substances ...
... describe, interpret and/or predict the effect of different types of bonding (ionic bonding; covalent bonding; hydrogen bonding; other intermolecular interactions; metallic bonding) on the physical properties of substances ...
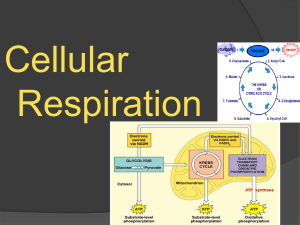
Biology 5.3 Cellular Respiration - Chemistry
... In the first stage of cellular respiration, glucose is broken down to pyruvate during glycolysis. Glycolysis is an anaerobic process (no oxygen required), and it results in a gain of two ATP molecules. ...
... In the first stage of cellular respiration, glucose is broken down to pyruvate during glycolysis. Glycolysis is an anaerobic process (no oxygen required), and it results in a gain of two ATP molecules. ...
Fat-Soluble
... • Found in sour fruits such as oranges, pineapples, and cranberries, as well as some animal products. (Most mammals make their own vitamin C, except for us.) • Lack of vitamin C causes scurvy, a disease that leads to gum damage, bleeding, joint pain, fatigue, and eventually death. • It is difficult ...
... • Found in sour fruits such as oranges, pineapples, and cranberries, as well as some animal products. (Most mammals make their own vitamin C, except for us.) • Lack of vitamin C causes scurvy, a disease that leads to gum damage, bleeding, joint pain, fatigue, and eventually death. • It is difficult ...
CHAPtER 9 Properties and reactions of organic compounds
... essentially infinite because there are so many combinations of organic compounds. However, certain general patterns involving addition, decomposition, combination, substitution or rearrangement of atoms or groups of atoms can be used to describe many common and useful reactions. It is not unusual to ...
... essentially infinite because there are so many combinations of organic compounds. However, certain general patterns involving addition, decomposition, combination, substitution or rearrangement of atoms or groups of atoms can be used to describe many common and useful reactions. It is not unusual to ...
Cellular Respiration
... If oxygen is not present, Pyruvic Acid goes along another pathway. It does not produce ATP, but other organic molecules that regenerates NAD+ to keep the cycle going. In Lactic Acid Fermentation, it converts to lactic acid. In Alcoholic Fermentation, it converts to ethyl alcohol and carbon dioxide. ...
... If oxygen is not present, Pyruvic Acid goes along another pathway. It does not produce ATP, but other organic molecules that regenerates NAD+ to keep the cycle going. In Lactic Acid Fermentation, it converts to lactic acid. In Alcoholic Fermentation, it converts to ethyl alcohol and carbon dioxide. ...
Chemical Basis of Life - SBCC Biological Sciences Department
... chloride ions react to form a type of chemical bond called an ionic bond (electrovalent bond). Sodium ions (Na+) and chloride ions (Cl–) uniting in this manner form the compound sodium chloride (NaCl), or table salt (fig. 2.4b). Some ions have an electrical charge greater than 1—for example, Ca+2 (o ...
... chloride ions react to form a type of chemical bond called an ionic bond (electrovalent bond). Sodium ions (Na+) and chloride ions (Cl–) uniting in this manner form the compound sodium chloride (NaCl), or table salt (fig. 2.4b). Some ions have an electrical charge greater than 1—for example, Ca+2 (o ...
respiration - Sakshieducation.com
... Anaerobes that can tolerate aerobic conditions are called as facultative anaerobes. e.g. Yeasts. There are two stages in anaerobic respiration known as Glycolysis and Fermentation. Glycolysis results in the formation of 2 molecules of Pyruvic acid, 2 NADH2 and net gain of 2 ATP. Ethyl alcohol is for ...
... Anaerobes that can tolerate aerobic conditions are called as facultative anaerobes. e.g. Yeasts. There are two stages in anaerobic respiration known as Glycolysis and Fermentation. Glycolysis results in the formation of 2 molecules of Pyruvic acid, 2 NADH2 and net gain of 2 ATP. Ethyl alcohol is for ...
8872 Chemistry H1 syllabus for 2016
... describe, interpret and/or predict the effect of different types of bonding (ionic bonding; covalent bonding; hydrogen bonding; other intermolecular interactions; metallic bonding) on the physical properties of substances ...
... describe, interpret and/or predict the effect of different types of bonding (ionic bonding; covalent bonding; hydrogen bonding; other intermolecular interactions; metallic bonding) on the physical properties of substances ...
Chapter 8 - Plant Biology
... electrons in oxidation-reduction reactions. If a cofactor (sometimes called a coenzyme) is loosely bound to its enzyme and is capable of accepting and donating electrons (or hydrogen atoms; electrons plus protons), it may serve as a carrier of electrons (or hydrogen atoms) from one reaction to anoth ...
... electrons in oxidation-reduction reactions. If a cofactor (sometimes called a coenzyme) is loosely bound to its enzyme and is capable of accepting and donating electrons (or hydrogen atoms; electrons plus protons), it may serve as a carrier of electrons (or hydrogen atoms) from one reaction to anoth ...
9.1-10.5 Organic Chemistry
... Problem: What is the relative order of the boiling points of hexane, hex-1-ene, cyclohexane, cyclohexene, and benzene? Prediction: Determine the number of electrons in each molecule and use these numbers to determine the order of boiling points Analysis: On the basis of the evidence given, determine ...
... Problem: What is the relative order of the boiling points of hexane, hex-1-ene, cyclohexane, cyclohexene, and benzene? Prediction: Determine the number of electrons in each molecule and use these numbers to determine the order of boiling points Analysis: On the basis of the evidence given, determine ...
Radical (chemistry)

In chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valency electrons.With some exceptions, these unpaired electrons make free radicals highly chemically reactive towards other substances, or even towards themselves: their molecules will often spontaneously dimerize or polymerize if they come in contact with each other. Most radicals are reasonably stable only at very low concentrations in inert media or in a vacuum.A notable example of a free radical is the hydroxyl radical (HO•), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (:CH2) which have two unpaired electrons. In contrast, the hydroxyl anion (HO−) is not a radical, since the unpaired electron is resolved by the addition of an electron; singlet oxygen and singlet carbene are not radicals as the two electrons are paired.Free radicals may be created in a number of ways, including synthesis with very dilute or rarefied reagents, reactions at very low temperatures, or breakup of larger molecules. The latter can be affected by any process that puts enough energy into the parent molecule, such as ionizing radiation, heat, electrical discharges, electrolysis, and chemical reactions. Indeed, radicals are intermediate stages in many chemical reactions.Free radicals play an important role in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. In living organisms, the free radicals superoxide and nitric oxide and their reaction products regulate many processes, such as control of vascular tone and thus blood pressure. They also play a key role in the intermediary metabolism of various biological compounds. Such radicals can even be messengers in a process dubbed redox signaling. A radical may be trapped within a solvent cage or be otherwise bound.Until late in the 20th century the word ""radical"" was used in chemistry to indicate any connected group of atoms, such as a methyl group or a carboxyl, whether it was part of a larger molecule or a molecule on its own. The qualifier ""free"" was then needed to specify the unbound case. Following recent nomenclature revisions, a part of a larger molecule is now called a functional group or substituent, and ""radical"" now implies ""free"". However, the old nomenclature may still occur in the literature.