Gases - Teacher Notes
... to 9.8 atm at constant T. What is the new volume? P1V1=P2V2 (0.98 atm)(20.5 L) = (9.8 atm)(V2) 20.09 atm*L/9.8 atm= V2 2.1 L = V2 30.6 mL of carbon dioxide at 740 torr is expanded at constant temperature to 750 mL. What is the final pressure in kPa? (30.6mL)(740Torr)=(750 mL)(P2) 22,644 mL*Torr/750 ...
... to 9.8 atm at constant T. What is the new volume? P1V1=P2V2 (0.98 atm)(20.5 L) = (9.8 atm)(V2) 20.09 atm*L/9.8 atm= V2 2.1 L = V2 30.6 mL of carbon dioxide at 740 torr is expanded at constant temperature to 750 mL. What is the final pressure in kPa? (30.6mL)(740Torr)=(750 mL)(P2) 22,644 mL*Torr/750 ...
How Do Gases Behave?
... to 9.8 atm at constant T. What is the new volume? P1V1=P2V2 (0.98 atm)(20.5 L) = (9.8 atm)(V2) 20.09 atm*L/9.8 atm= V2 2.1 L = V2 30.6 mL of carbon dioxide at 740 torr is expanded at constant temperature to 750 mL. What is the final pressure in kPa? (30.6mL)(740Torr)=(750 mL)(P2) 22,644 mL*Torr/750 ...
... to 9.8 atm at constant T. What is the new volume? P1V1=P2V2 (0.98 atm)(20.5 L) = (9.8 atm)(V2) 20.09 atm*L/9.8 atm= V2 2.1 L = V2 30.6 mL of carbon dioxide at 740 torr is expanded at constant temperature to 750 mL. What is the final pressure in kPa? (30.6mL)(740Torr)=(750 mL)(P2) 22,644 mL*Torr/750 ...
Gas Chromatography
... compound. The comparison of retention times is what gives GC its analytical usefulness. A gas chromatograph uses a flow-through narrow tube known as the column, through which different chemical constituents of a sample pass in a gas stream (carrier gas, mobile phase) at different rates depending on ...
... compound. The comparison of retention times is what gives GC its analytical usefulness. A gas chromatograph uses a flow-through narrow tube known as the column, through which different chemical constituents of a sample pass in a gas stream (carrier gas, mobile phase) at different rates depending on ...
Gas review
... 1. A gas is composed of molecules that are separated from each other by distances far greater than their own dimensions. The molecules can be considered to be points; that is, they possess mass but have negligible volume. 2. Gas molecules are in constant motion in random directions. Collisions among ...
... 1. A gas is composed of molecules that are separated from each other by distances far greater than their own dimensions. The molecules can be considered to be points; that is, they possess mass but have negligible volume. 2. Gas molecules are in constant motion in random directions. Collisions among ...
Show - Evonik
... referred to herein but not any in-depth health and safety information. The information in this GPS Safety Summary is supplied on the condition that the persons receiving the same will make their own determination as to its suitability for their purposes prior to use. This GPS Safety Summary does not ...
... referred to herein but not any in-depth health and safety information. The information in this GPS Safety Summary is supplied on the condition that the persons receiving the same will make their own determination as to its suitability for their purposes prior to use. This GPS Safety Summary does not ...
Hydrogen Sulfide (H2S) Technical Information
... products. These high performance materials have high surface areas and unique morphologies which make them uniquely different from conventional counterparts. This high chemical reactivity led to development of several formulations that are very effective at treating a broad range of chemical hazards ...
... products. These high performance materials have high surface areas and unique morphologies which make them uniquely different from conventional counterparts. This high chemical reactivity led to development of several formulations that are very effective at treating a broad range of chemical hazards ...
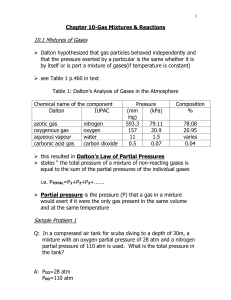
Chapter 10 Notes
... react with 125L of carbon monoxide produced during a 100km trip? A: 2CO(g) + O2(g)-----2CO2(g) 125L V=? VO2=125L CO x 1mol/2mol =62.5L ,therefore the volume of O2 required is 62.5L Answer questions 1, 3-5 on pages 468 & 469. ...
... react with 125L of carbon monoxide produced during a 100km trip? A: 2CO(g) + O2(g)-----2CO2(g) 125L V=? VO2=125L CO x 1mol/2mol =62.5L ,therefore the volume of O2 required is 62.5L Answer questions 1, 3-5 on pages 468 & 469. ...
Document
... Of the elements on the periodic table, some exist as a gas at room temperature, these include the Noble Gases, Group 8A (18), H2, N2, O2, F2, Cl2, and many oxides of nonmetals such as CO, CO2, NO, NO2, SO2, and SO3. ...
... Of the elements on the periodic table, some exist as a gas at room temperature, these include the Noble Gases, Group 8A (18), H2, N2, O2, F2, Cl2, and many oxides of nonmetals such as CO, CO2, NO, NO2, SO2, and SO3. ...
Chapter 6 notes 2015
... If you have two gases at the same temperature and pressure, than : the average kinetic energy of the two gases is the same. (same temp.) ... if there is any difference in the pressure exerted by the two gases, it would be due to the number of particles. however, since we have already stated th ...
... If you have two gases at the same temperature and pressure, than : the average kinetic energy of the two gases is the same. (same temp.) ... if there is any difference in the pressure exerted by the two gases, it would be due to the number of particles. however, since we have already stated th ...
284
... If 25.0 g of ethyl alcohol is burned in air (excess oxygen), calculate the mass of carbon dioxide produced. 33. Small quantities of oxygen gas can be generated in the laboratory by the decomposition of hydrogen peroxide. The unbalanced equation for the reaction is H2O2(aq) H2O(l) + O2(g) Calculate ...
... If 25.0 g of ethyl alcohol is burned in air (excess oxygen), calculate the mass of carbon dioxide produced. 33. Small quantities of oxygen gas can be generated in the laboratory by the decomposition of hydrogen peroxide. The unbalanced equation for the reaction is H2O2(aq) H2O(l) + O2(g) Calculate ...
CP Chemistry Practice Mid
... 25. Water has a specific heat of 4.184 J/g°C while glass (Pyrex) has a specific heat of 0.780 J/g°C. If 10.0 J of heat is added to 1.00 g of each of these, which will experience the larger increase of temperature? a. glass b. water c. They both will experience the same change in temperature since on ...
... 25. Water has a specific heat of 4.184 J/g°C while glass (Pyrex) has a specific heat of 0.780 J/g°C. If 10.0 J of heat is added to 1.00 g of each of these, which will experience the larger increase of temperature? a. glass b. water c. They both will experience the same change in temperature since on ...
Introductory Chemistry: A Foundation FOURTH EDITION by Steven
... 10. Repeat – A trick of the trade, when you are forced to attack an element that is in 3 or more compounds – find where it is uncombined. You can find a factor to make it any amount you want, even if that factor is a fraction! – We want to make the O on the left equal 5, therefore we will multiply i ...
... 10. Repeat – A trick of the trade, when you are forced to attack an element that is in 3 or more compounds – find where it is uncombined. You can find a factor to make it any amount you want, even if that factor is a fraction! – We want to make the O on the left equal 5, therefore we will multiply i ...
ap unit 5 worksheet answers
... 3. A fixed quantity of gas at 23⁰C exhibits a pressure of 748 torr and occupies a volume of 10.3 L. Calculate the volume the gas will occupy if the temperature is increased to 145 ⁰C 15 L 4. A fixed quantity of gas at 21⁰C exhibits a pressure of 748 torr and occupies a volume of 10.3 L. Calculate th ...
... 3. A fixed quantity of gas at 23⁰C exhibits a pressure of 748 torr and occupies a volume of 10.3 L. Calculate the volume the gas will occupy if the temperature is increased to 145 ⁰C 15 L 4. A fixed quantity of gas at 21⁰C exhibits a pressure of 748 torr and occupies a volume of 10.3 L. Calculate th ...
presentation on power generation from biogas in 2
... The gas from the bottom of the Scrubber enters the packed Column while liquid is collected in the Tank for recalculation. In the II stage of scrubbing, gas is scrubbed with caustic solution in a packed Column. The Column is provided with ceramic rings to have enhanced surface area for mass transfer. ...
... The gas from the bottom of the Scrubber enters the packed Column while liquid is collected in the Tank for recalculation. In the II stage of scrubbing, gas is scrubbed with caustic solution in a packed Column. The Column is provided with ceramic rings to have enhanced surface area for mass transfer. ...
Chapter 5 Gases
... • Occupy containers uniformly and completely • The densities of gases are much smaller than those of liquids and solid and highly variable depending on temperature and pressure • Expand infinitely • Diffuse and mix rapidly ...
... • Occupy containers uniformly and completely • The densities of gases are much smaller than those of liquids and solid and highly variable depending on temperature and pressure • Expand infinitely • Diffuse and mix rapidly ...
Lab Stuff:
... 1. Carbon atoms make 4 bonds, and can arrange these bonds by using single, double, or triple bonds to other atoms. 2. Electron-dot and structural formulas describe the bonds between atoms in alkanes, alkenes and alkynes. 3. Petroleum is a nonrenewable resource, and many everyday materials are produc ...
... 1. Carbon atoms make 4 bonds, and can arrange these bonds by using single, double, or triple bonds to other atoms. 2. Electron-dot and structural formulas describe the bonds between atoms in alkanes, alkenes and alkynes. 3. Petroleum is a nonrenewable resource, and many everyday materials are produc ...
Lab Stuff
... 1. Carbon atoms make 4 bonds, and can arrange these bonds by using single, double, or triple bonds to other atoms. 2. Electron-dot and structural formulas describe the bonds between atoms in alkanes, alkenes and alkynes. 3. Petroleum is a nonrenewable resource, and many everyday materials are produc ...
... 1. Carbon atoms make 4 bonds, and can arrange these bonds by using single, double, or triple bonds to other atoms. 2. Electron-dot and structural formulas describe the bonds between atoms in alkanes, alkenes and alkynes. 3. Petroleum is a nonrenewable resource, and many everyday materials are produc ...
Lab Stuff - WW-P K
... 1. Carbon atoms make 4 bonds, and can arrange these bonds by using single, double, or triple bonds to other atoms. 2. Electron-dot and structural formulas describe the bonds between atoms in alkanes, alkenes and alkynes. 3. Petroleum is a nonrenewable resource, and many everyday materials are produc ...
... 1. Carbon atoms make 4 bonds, and can arrange these bonds by using single, double, or triple bonds to other atoms. 2. Electron-dot and structural formulas describe the bonds between atoms in alkanes, alkenes and alkynes. 3. Petroleum is a nonrenewable resource, and many everyday materials are produc ...
de Caux - Combustion of Methane Demonstration
... Water has a high specific heat capacity and, therefore, demonstrates a relatively small change in temperature for a given amount of heat absorbed. This capacity to absorb heat without undergoing a large temperature change is why water is used to prevent burns during this demonstration. Methane is a ...
... Water has a high specific heat capacity and, therefore, demonstrates a relatively small change in temperature for a given amount of heat absorbed. This capacity to absorb heat without undergoing a large temperature change is why water is used to prevent burns during this demonstration. Methane is a ...
Exam 3 - Canvas by Instructure
... Hint: There is more than one correct answer that is consistent with the data provided. D. Based on your from part C above, which gas would have the larger a & b terms in the van der Waals equation? Explain your reasoning by describing the different structures of the unknown gases you chose. ...
... Hint: There is more than one correct answer that is consistent with the data provided. D. Based on your from part C above, which gas would have the larger a & b terms in the van der Waals equation? Explain your reasoning by describing the different structures of the unknown gases you chose. ...
Chapter 3 Part 2 Review
... photosynthesis in the forest. One morning, it wakes up with dew on its leaves. The sunshine immediately begins to evaporate the dew. Suddenly, a storm rolls in and a giant wind knocks the tree to the ground. A limb is struck by lighting and partially burns. Over the next year, the remaining parts of ...
... photosynthesis in the forest. One morning, it wakes up with dew on its leaves. The sunshine immediately begins to evaporate the dew. Suddenly, a storm rolls in and a giant wind knocks the tree to the ground. A limb is struck by lighting and partially burns. Over the next year, the remaining parts of ...
METHODOLOGY FOR RESIDUAL OXYGEN CALORIMETRY
... Requirement Index (CARI) of the fuel, which can then be mathematically correlated to the Wobbe Index. Differences between the CARI and Wobbe Index values can be cancelled out by the use of calibration gases thus giving this technique the ability to report Wobbe Index values along with CARI. With the ...
... Requirement Index (CARI) of the fuel, which can then be mathematically correlated to the Wobbe Index. Differences between the CARI and Wobbe Index values can be cancelled out by the use of calibration gases thus giving this technique the ability to report Wobbe Index values along with CARI. With the ...