0191 271 0222 Nitrogen (Oxygen Free)
... Protect gas cylinders from external heat sources that may adversely affect their mechanical integrity ...
... Protect gas cylinders from external heat sources that may adversely affect their mechanical integrity ...
AP Chemistry Unit 3 Test Review Topics Covered: Gases Liquids
... (C) The average kinetic energy stays the same. (D) It cannot be determined how the kinetic energy is affected without knowing P1 and P2. ...
... (C) The average kinetic energy stays the same. (D) It cannot be determined how the kinetic energy is affected without knowing P1 and P2. ...
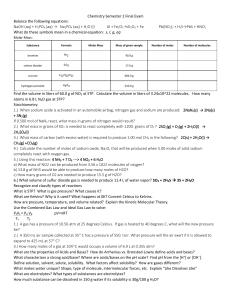
Chemistry Semester 2 Final Exam Chemistry Semester 2 Final Exam
... 3.) What mass of carbon (with excess water) is required to produce 1.00 mol CH4 in the following? 2C(s) + 2H2O(l) → CH4(g) +CO2(g) 24.02 g 4.) Calculate the number of moles of sodium oxide, Na2O, that will be produced when 5.00 moles of solid sodium completely react with oxygen gas. 2.5 mol 5.) Usin ...
... 3.) What mass of carbon (with excess water) is required to produce 1.00 mol CH4 in the following? 2C(s) + 2H2O(l) → CH4(g) +CO2(g) 24.02 g 4.) Calculate the number of moles of sodium oxide, Na2O, that will be produced when 5.00 moles of solid sodium completely react with oxygen gas. 2.5 mol 5.) Usin ...
AP gas notes 2010
... particles of a real gas occupy space and exert attractive forces. • The deviations are most recognizable when gases are at extremely low temperatures or at very high pressures ( > 10 atm) • This is due to the small KE of the particles and the particles will be closer ...
... particles of a real gas occupy space and exert attractive forces. • The deviations are most recognizable when gases are at extremely low temperatures or at very high pressures ( > 10 atm) • This is due to the small KE of the particles and the particles will be closer ...
Chemistry 332
... Explain why the following equation supports the law of conservation of matter. ...
... Explain why the following equation supports the law of conservation of matter. ...
Practice Test #1
... The average velocity of the gas particles is directly proportional to the pressure. Gas particles are very small compared with the average distance between particlesGas particles collide with the walls of their container and in doing so give rise to pressure. Gasesare made up of tiny particles in co ...
... The average velocity of the gas particles is directly proportional to the pressure. Gas particles are very small compared with the average distance between particlesGas particles collide with the walls of their container and in doing so give rise to pressure. Gasesare made up of tiny particles in co ...
Exam Review
... 10. Determine whether the following molecules are polar or non-polar. Show your work (EN calculations and diagrams with dipoles). a. CH4 b. H2O c. NH3 d. CO2 e. HCl ...
... 10. Determine whether the following molecules are polar or non-polar. Show your work (EN calculations and diagrams with dipoles). a. CH4 b. H2O c. NH3 d. CO2 e. HCl ...
110 EXAM IV MATERIAL Tro Spr 2015
... 4. Smelling salts contain ammonium carbonate, which can decompose to form ammonia, a mild heart stimulant. The ammonium carbonate decomposes according to the following reaction: (NH4)2CO3(s) 2NH3(g) + CO2(g) + H2O(l) ...
... 4. Smelling salts contain ammonium carbonate, which can decompose to form ammonia, a mild heart stimulant. The ammonium carbonate decomposes according to the following reaction: (NH4)2CO3(s) 2NH3(g) + CO2(g) + H2O(l) ...
Chemistry 2nd Semester Final Review
... Most things are mixtures There are only two types of pure substances: elements and compounds A mixture is anything that is not a single element or a single compound, but a mixture of more than one element or compound Mixtures can be separated through physical means. For example, if you want to remov ...
... Most things are mixtures There are only two types of pure substances: elements and compounds A mixture is anything that is not a single element or a single compound, but a mixture of more than one element or compound Mixtures can be separated through physical means. For example, if you want to remov ...
practice quiz5
... absolute temperature scale is the same. A) True B) False Question 13 The Kelvin temperature scale is useful when comparing: A) various gas samples at different densities B) volume of a gas sample with temperature at constant pressure C) pressure of gas samples at different volumes and constant tempe ...
... absolute temperature scale is the same. A) True B) False Question 13 The Kelvin temperature scale is useful when comparing: A) various gas samples at different densities B) volume of a gas sample with temperature at constant pressure C) pressure of gas samples at different volumes and constant tempe ...
the nakuru district sec. schools trial examinations - 2015
... 28. A piece of burning magnesium was inserted in a gas jar of carbon(IV) oxide (a) State and explain the observations made (2 marks) The reaction is exothermic ½ producing heat which decomposes carbon(IV)oxide to black½ carbon specks and oxygen gas. Oxygen gas supports½ continued burning of mag ...
... 28. A piece of burning magnesium was inserted in a gas jar of carbon(IV) oxide (a) State and explain the observations made (2 marks) The reaction is exothermic ½ producing heat which decomposes carbon(IV)oxide to black½ carbon specks and oxygen gas. Oxygen gas supports½ continued burning of mag ...
Chemistry- The Gas Phase
... Ideal Gas: a hypothetical gas whose pressurevolume-temperature behavior can be completely accounted for the ideal gas equation. (molecules or atoms of gas don’t interact with each other at all) This happens when: 1. Volume of container is large compared to the V of the gas (P is low) 2. Molecules o ...
... Ideal Gas: a hypothetical gas whose pressurevolume-temperature behavior can be completely accounted for the ideal gas equation. (molecules or atoms of gas don’t interact with each other at all) This happens when: 1. Volume of container is large compared to the V of the gas (P is low) 2. Molecules o ...
Pollution Control - No Brain Too Small
... shaken to dislodge the dust, causing it to fall with the force of gravity to hoppers below. The dust is then removed by a conveyor system for disposal or recycling. Depending upon dust characteristics and the gas volume to be treated, there are many different sizes, types and designs of electrostati ...
... shaken to dislodge the dust, causing it to fall with the force of gravity to hoppers below. The dust is then removed by a conveyor system for disposal or recycling. Depending upon dust characteristics and the gas volume to be treated, there are many different sizes, types and designs of electrostati ...
+ O2 (g)
... Organic vs. Inorganic In the18th century, compounds from living things were called organic; compounds from the nonliving environment were called inorganic. ...
... Organic vs. Inorganic In the18th century, compounds from living things were called organic; compounds from the nonliving environment were called inorganic. ...
Name……………………………………............................. Index number
... Write your name, index number class and admission number in the spaces provided Sign and write the date of examination in the spaces provided. Answer all the questions in the spaces provided. Mathematical tables and silent electronic calculators may be used. All working must be clearly shown where n ...
... Write your name, index number class and admission number in the spaces provided Sign and write the date of examination in the spaces provided. Answer all the questions in the spaces provided. Mathematical tables and silent electronic calculators may be used. All working must be clearly shown where n ...
CHEM 1405 Practice Exam 3 (2015)
... 9) Assuming similar conditions, how many liters of water vapor, H2O, react to produce 1L of hydrogen gas? C(s) + H2O(g) CO(g) + H2(g) A) 1 L ...
... 9) Assuming similar conditions, how many liters of water vapor, H2O, react to produce 1L of hydrogen gas? C(s) + H2O(g) CO(g) + H2(g) A) 1 L ...
Chapter 4: Properties of Gases
... When comparing energies of a double and a single bond we find that when we break a double bond (a) A higher energy is given out. (c)A higher energy is taken in. ...
... When comparing energies of a double and a single bond we find that when we break a double bond (a) A higher energy is given out. (c)A higher energy is taken in. ...
AP Ch 5 Gases . ppt
... 0.421 moles of C2H6, and 0.116 moles of C3H8. If the total pressure of the gases is 1.37 atm, what is the partial pressure of propane (C3H8)? ...
... 0.421 moles of C2H6, and 0.116 moles of C3H8. If the total pressure of the gases is 1.37 atm, what is the partial pressure of propane (C3H8)? ...
Study Guide (Semester 2)
... To be able to write a skeleton chemical equation from words. Directions: Write a complete balanced equation for each chemical reaction. Box your answer. 1. Tin foil will oxidize when exposed to oxygen forming tin (II) oxide. ...
... To be able to write a skeleton chemical equation from words. Directions: Write a complete balanced equation for each chemical reaction. Box your answer. 1. Tin foil will oxidize when exposed to oxygen forming tin (II) oxide. ...
Semester 2 Final Exam
... bonds with Br EXCEPT: (A) C (B) H (C) K (D) O 48. Bonds between which of the following elements is expected to be most polar? (A) C–H (B) N–O (C) P–F (D) S–Br 49. Which type of bond is described as “a lattice of positive ions in a sea of electrons”? (A) covalent (B) hydrogen (C) ionic (D) metallic 5 ...
... bonds with Br EXCEPT: (A) C (B) H (C) K (D) O 48. Bonds between which of the following elements is expected to be most polar? (A) C–H (B) N–O (C) P–F (D) S–Br 49. Which type of bond is described as “a lattice of positive ions in a sea of electrons”? (A) covalent (B) hydrogen (C) ionic (D) metallic 5 ...
Characteristics of Gases Pressure Gas Laws The Ideal
... Some nitrogen gas is in a 2.00-L tank at a pressure of 3.00 atm. The tank is connected to a 5.00-L tank that is completely empty (evacuated), and a valve connects the two tanks., If the valve is opened, determine the total pressure in this two-tank system after the nitrogen stops flowing. No tempera ...
... Some nitrogen gas is in a 2.00-L tank at a pressure of 3.00 atm. The tank is connected to a 5.00-L tank that is completely empty (evacuated), and a valve connects the two tanks., If the valve is opened, determine the total pressure in this two-tank system after the nitrogen stops flowing. No tempera ...
AP Chemistry Unit 3 Test Review Topics Covered: Gases Liquids
... D) water molecules have a higher average kinetic energy due to the lower atmospheric pressure E) water contains a greater concentration of dissolved Gas 13) Under which of the following conditions of temperature and pressure will H2 gas be expected to behave most like an ideal gas? (A) 50 K and 0.10 ...
... D) water molecules have a higher average kinetic energy due to the lower atmospheric pressure E) water contains a greater concentration of dissolved Gas 13) Under which of the following conditions of temperature and pressure will H2 gas be expected to behave most like an ideal gas? (A) 50 K and 0.10 ...
General Chemistry Sample Exam 2 and Outline
... it. The reaction produces sodium sulfate, carbon dioxide and water. i) Write the molecular, ionic and net ionic equation and balance the equation (include the phases). ii) What is the limiting reagent and how much remains if 35 ml of 6.0 M sulfuric acid is spilled and 50 grams of sodium bicarbonate ...
... it. The reaction produces sodium sulfate, carbon dioxide and water. i) Write the molecular, ionic and net ionic equation and balance the equation (include the phases). ii) What is the limiting reagent and how much remains if 35 ml of 6.0 M sulfuric acid is spilled and 50 grams of sodium bicarbonate ...