Answers - Shelton State
... 9. Write the products and use the solubility chart in your textbook to determine whether the precipitation reaction below would occur. ...
... 9. Write the products and use the solubility chart in your textbook to determine whether the precipitation reaction below would occur. ...
AP Chem Mr. Dehne Name: ___________ Date: Per#: ___ AP
... The partial pressure of CH4(g) is 0.175atm and that of O2(g) is 0.250atm in a mixture of two gases. a. What is the mole fraction of each gas in the mixture? b. If the mixture occupies a volume of 10.5L at 65oC, calculate the total number of moles of gas in the mixture. c. Calculate the number of gra ...
... The partial pressure of CH4(g) is 0.175atm and that of O2(g) is 0.250atm in a mixture of two gases. a. What is the mole fraction of each gas in the mixture? b. If the mixture occupies a volume of 10.5L at 65oC, calculate the total number of moles of gas in the mixture. c. Calculate the number of gra ...
QUATERLY 3 REVIEW CHAPTER 12- Stoichiometry Define the law
... 5. Define Percent Yield and calculate it from given data 6. a. What mass of iron must react with excess oxygen in order to form 4.81 moles of iron (III) oxide? b. What mass of iron is needed to react with 40.0 g of oxygen? 7. Predict the mass of barium phosphate that will be formed if 20.2g of bariu ...
... 5. Define Percent Yield and calculate it from given data 6. a. What mass of iron must react with excess oxygen in order to form 4.81 moles of iron (III) oxide? b. What mass of iron is needed to react with 40.0 g of oxygen? 7. Predict the mass of barium phosphate that will be formed if 20.2g of bariu ...
Propane - Power Fuels LLC
... water is not available to protect the container shell from weakening, the area will be required to be evacuated. If gas has not ignited, liquid or vapor may be dispersed by water spray or ...
... water is not available to protect the container shell from weakening, the area will be required to be evacuated. If gas has not ignited, liquid or vapor may be dispersed by water spray or ...
Name__________________________ Honors Chemistry Final
... 3. Xe gas effuses from a certain container at a slow rate. An unknown gas effuses from the same container at a rate 3x faster than the Xe gas. What is the molar mass of the unknown gas? ...
... 3. Xe gas effuses from a certain container at a slow rate. An unknown gas effuses from the same container at a rate 3x faster than the Xe gas. What is the molar mass of the unknown gas? ...
42.89 KB
... mm of Hg at 30 °C) Molar Mass of sucrose (C12H22O11) is 342.3 g/mol. A) B) C) D) E) ...
... mm of Hg at 30 °C) Molar Mass of sucrose (C12H22O11) is 342.3 g/mol. A) B) C) D) E) ...
Summary of Gas Laws
... If a liquid is heated to sufficiently high temperature, its vapor pressure can become equal to the applied (atmospheric) pressure Gas phase in the form of bubbles is formed within the volume of the liquid and the bubbles rise to the surface and burst releasing the vapor into the air This process is ...
... If a liquid is heated to sufficiently high temperature, its vapor pressure can become equal to the applied (atmospheric) pressure Gas phase in the form of bubbles is formed within the volume of the liquid and the bubbles rise to the surface and burst releasing the vapor into the air This process is ...
Chemistry 111 Study Sheet - Answers
... T Gas molecules move in straight lines and change direction when they collide with other molecules or the walls of the container. c) F Collisions are not elastic, energy is lost to friction. d) T At low pressures, distance between gas molecules is large in comparison to their size. e) T At low press ...
... T Gas molecules move in straight lines and change direction when they collide with other molecules or the walls of the container. c) F Collisions are not elastic, energy is lost to friction. d) T At low pressures, distance between gas molecules is large in comparison to their size. e) T At low press ...
Final Review Sheet Answers (the 6 page packet)
... however, the CO2 molecule does have a dipole moment between the C and O atoms. The dipole cancels in the molecule overall making it nonpolar, but in reality, the oxygens are still slightly more negative than the carbon and can interact with other CO2 molecules. For all of these reasons, the balloon ...
... however, the CO2 molecule does have a dipole moment between the C and O atoms. The dipole cancels in the molecule overall making it nonpolar, but in reality, the oxygens are still slightly more negative than the carbon and can interact with other CO2 molecules. For all of these reasons, the balloon ...
Ch. 11: Gases
... VIII. Gases and Stoichiometry • Gases can be either products or reactants in a reaction, so they can be involved in stoichiometry problems. • Mole relationships allow gas calculations via the ideal gas equation. • Note that at STP (0 °C and 1 atm) 1 mole of gas occupies 22.4 L. ...
... VIII. Gases and Stoichiometry • Gases can be either products or reactants in a reaction, so they can be involved in stoichiometry problems. • Mole relationships allow gas calculations via the ideal gas equation. • Note that at STP (0 °C and 1 atm) 1 mole of gas occupies 22.4 L. ...
File
... 50.0 milliliters. What volume will the gas have at a pressure of 20.0 kPa and a temperature of 200. K? 1) 50.0 mL 2) 200. mL 3) 12.5 mL 4) 100. mL An ideal gas is made up of gas particles that 1) can be liquefied 2) attract each other 3) are in random motion 4) have volume A 1-liter flask contains t ...
... 50.0 milliliters. What volume will the gas have at a pressure of 20.0 kPa and a temperature of 200. K? 1) 50.0 mL 2) 200. mL 3) 12.5 mL 4) 100. mL An ideal gas is made up of gas particles that 1) can be liquefied 2) attract each other 3) are in random motion 4) have volume A 1-liter flask contains t ...
Molecular Mass of a gas
... Use R = 62.4 L·torr·mol-1·K-1 in your calculations. Use the proper number of significant digits throughout the calculations. Show the mathematical setup in the ...
... Use R = 62.4 L·torr·mol-1·K-1 in your calculations. Use the proper number of significant digits throughout the calculations. Show the mathematical setup in the ...
Chemistry
... Properties of matter and changes of matter Matter: anything that has mass and takes up ...
... Properties of matter and changes of matter Matter: anything that has mass and takes up ...
Gas Laws Practice Test.Ans.Key
... We assume the volume of the gas to be equal to the volume of the container, but at high pressure, the available free space for molecular movement is decreased, increasing the number of collisions. This results in an increase in the observed pressure. ...
... We assume the volume of the gas to be equal to the volume of the container, but at high pressure, the available free space for molecular movement is decreased, increasing the number of collisions. This results in an increase in the observed pressure. ...
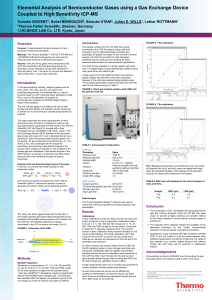
Elemental Analysis of Semiconductor Gases Using a Gas Exchange
... J-SCIENCE LAB Gas Exchange Device were used to measure trace amounts of semiconductor gases. Results: AsH3 and GeH4 gases were measured by the iCAP Qs connected to the Gas Exchange Device as described. Linear calibration curves were obtained for AsH3 and GeH4 from 1 vol.ppt to 0.5 vol.ppb and detect ...
... J-SCIENCE LAB Gas Exchange Device were used to measure trace amounts of semiconductor gases. Results: AsH3 and GeH4 gases were measured by the iCAP Qs connected to the Gas Exchange Device as described. Linear calibration curves were obtained for AsH3 and GeH4 from 1 vol.ppt to 0.5 vol.ppb and detect ...
a Gas
... - has a specific volume but it assumes the shape of its container. - it has a ability to flow -the distances between atoms are greater than in a solid ...
... - has a specific volume but it assumes the shape of its container. - it has a ability to flow -the distances between atoms are greater than in a solid ...
CHAPTER 5 REVIEW PACKET – GAS LAWS
... 10. Magnesium can be used as a “getter” in evacuated enclosures, to react with the last traces of oxygen. (The magnesium is usually heated by passing an electric current through a wire or ribbon of the metal.) If an enclosure of 0.382 L has a partial pressure of O2 of 3.50 x 10-6 torr at 27.00C, wh ...
... 10. Magnesium can be used as a “getter” in evacuated enclosures, to react with the last traces of oxygen. (The magnesium is usually heated by passing an electric current through a wire or ribbon of the metal.) If an enclosure of 0.382 L has a partial pressure of O2 of 3.50 x 10-6 torr at 27.00C, wh ...
R= 8.31 J/mol K = 0.0821 L atm/mol K = 62.4 L torr/mol K PV = nRT
... ______15. How much heat is needed to raise the temperature of 1.00 mole of water from 10.0̊C to 50.0̊ C? A) 167 J B) 3.01 kJ C) 9.29 J D) 3760 J E) 40.0 kJ ______16. The pressure and kelvin temperature of a gas with a volume of 10.00 L are both doubled. The new volume of the gas is A) 10.00 L B) 2. ...
... ______15. How much heat is needed to raise the temperature of 1.00 mole of water from 10.0̊C to 50.0̊ C? A) 167 J B) 3.01 kJ C) 9.29 J D) 3760 J E) 40.0 kJ ______16. The pressure and kelvin temperature of a gas with a volume of 10.00 L are both doubled. The new volume of the gas is A) 10.00 L B) 2. ...
File
... ______15. How much heat is needed to raise the temperature of 1.00 mole of water from 10.0̊C to 50.0̊ C? A) 167 J B) 3.01 kJ C) 9.29 J D) 3760 J E) 40.0 kJ ______16. The pressure and kelvin temperature of a gas with a volume of 10.00 L are both doubled. The new volume of the gas is A) 10.00 L B) 2.5 ...
... ______15. How much heat is needed to raise the temperature of 1.00 mole of water from 10.0̊C to 50.0̊ C? A) 167 J B) 3.01 kJ C) 9.29 J D) 3760 J E) 40.0 kJ ______16. The pressure and kelvin temperature of a gas with a volume of 10.00 L are both doubled. The new volume of the gas is A) 10.00 L B) 2.5 ...
Santee Education Complex Chemistry Mini Assessment 4
... 7) A sample of unknown gas at STP has a density of 0.630 g per liter. What is the molecular mass of this gas? a. 2.81 g b. 14.1 g c. 22.4 g d. 63 g 8) Under which conditions are gases most soluble in water? a. High pressure and high pressure b. High pressure and low temperature c. Low pressure and h ...
... 7) A sample of unknown gas at STP has a density of 0.630 g per liter. What is the molecular mass of this gas? a. 2.81 g b. 14.1 g c. 22.4 g d. 63 g 8) Under which conditions are gases most soluble in water? a. High pressure and high pressure b. High pressure and low temperature c. Low pressure and h ...
C:\Users\Jim\Documents\usb key backups\Nov. 17\sch3u\unit 4
... Higher temperatures mean higher average speed. However, temperature is proportional to kinetic energy, not speed. Ek = .5 m v2 At the same temperature, molecules with larger molar masses are moving more slowly. You can measure this by releasing two gases at the same moment and timing how long it tak ...
... Higher temperatures mean higher average speed. However, temperature is proportional to kinetic energy, not speed. Ek = .5 m v2 At the same temperature, molecules with larger molar masses are moving more slowly. You can measure this by releasing two gases at the same moment and timing how long it tak ...
Test Chap 5 gas laws
... 20 °C. If the total pressure is 400 kPa, what is the partial pressure of the hydrogen? (3 pts) 16. What is the pressure exerted by some nitrogen gas collected in a tube filled with water on a day when the room temperature is 18.0 °C and the room pressure is 750.0 mmHg? [The partial pressure of water ...
... 20 °C. If the total pressure is 400 kPa, what is the partial pressure of the hydrogen? (3 pts) 16. What is the pressure exerted by some nitrogen gas collected in a tube filled with water on a day when the room temperature is 18.0 °C and the room pressure is 750.0 mmHg? [The partial pressure of water ...
Chemistry 2nd Semester Final Review
... CO2(g) + CaO(s). How many grams of calcium carbonate will I need to form 3.45 liters of carbon dioxide? 8. How many liters of water can be made from 55 grams of oxygen gas and an excess of hydrogen at a pressure of 12.4 atm and a temperature of 85C? 9. How many liters of water can be made from 34 g ...
... CO2(g) + CaO(s). How many grams of calcium carbonate will I need to form 3.45 liters of carbon dioxide? 8. How many liters of water can be made from 55 grams of oxygen gas and an excess of hydrogen at a pressure of 12.4 atm and a temperature of 85C? 9. How many liters of water can be made from 34 g ...