FINAL EXAM REVIEW PROBLEMS
... For each of the following reactions, predict the direction the equilibrium will shift when the volume of the container is increased. a. H2 (g) + F2 (g) 2 HF (g) b. CO (g) + 2 H2 (g) CH3OH (g) c. 2 SO3 (g) 2 SO2 (g) + O2 (g) 57. For the reaction: 2 SO2 (g) + O2 (g) 2 SO3 (g) a. If the reactio ...
... For each of the following reactions, predict the direction the equilibrium will shift when the volume of the container is increased. a. H2 (g) + F2 (g) 2 HF (g) b. CO (g) + 2 H2 (g) CH3OH (g) c. 2 SO3 (g) 2 SO2 (g) + O2 (g) 57. For the reaction: 2 SO2 (g) + O2 (g) 2 SO3 (g) a. If the reactio ...
AP Chemistry Summer Assignment
... 54. A sample of dolomitic limestone containing only CaCO3 and MgCO3 was analyzed. (a) When a 0.2800 gram sample of this limestone was decomposed by heating, 0.00308 moles of CO2 were evolved. How many grams of CO2 were produced? ...
... 54. A sample of dolomitic limestone containing only CaCO3 and MgCO3 was analyzed. (a) When a 0.2800 gram sample of this limestone was decomposed by heating, 0.00308 moles of CO2 were evolved. How many grams of CO2 were produced? ...
worksheer format 11-12
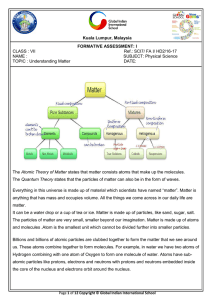
... being solid, liquid, gas, plasma and a new one called Bose-Einstein condensates.. As of 1995, scientists have identified five states of matter.Matter can change states as well as participate in chemical changes, depending on their chemical properties and composition. ...
... being solid, liquid, gas, plasma and a new one called Bose-Einstein condensates.. As of 1995, scientists have identified five states of matter.Matter can change states as well as participate in chemical changes, depending on their chemical properties and composition. ...
C:\Users\Sadhan Chakrabarty\Desktop\0909.xps
... same gas is added to the vessel without altering the temperature. What will be the change in pressure? What is the SI unit of coefficient of linear expansion of a solid ? or Write down the relation among the linear, surface and volume expansion coefficients of a solid. What type of spherical mirror ...
... same gas is added to the vessel without altering the temperature. What will be the change in pressure? What is the SI unit of coefficient of linear expansion of a solid ? or Write down the relation among the linear, surface and volume expansion coefficients of a solid. What type of spherical mirror ...
Gases - Teacher Notes
... 0 Kelvin: absolute zero (atom stops moving completely) Is there a maximum temperature in the universe? ...
... 0 Kelvin: absolute zero (atom stops moving completely) Is there a maximum temperature in the universe? ...
Family
... Bonding – The combining of atoms. Molecule – 1. The smallest particle of a substance that retains the chemical and physical properties of the substance and is composed of two or more atoms. 2. A group of like or different atoms held together by chemical ...
... Bonding – The combining of atoms. Molecule – 1. The smallest particle of a substance that retains the chemical and physical properties of the substance and is composed of two or more atoms. 2. A group of like or different atoms held together by chemical ...
Answers to Final Exam Review
... c. E1> E2, as CaCl2 is an ionic bond b. E1< E2, as CaCl2 is a covalent bond d. E1< E2, as CaCl2 is an ionic bond 32. Given the pairs of atoms below, predict whether the bond formed between the atoms is either ionic or covalent, and if ionic, write the formula for the predicted compound. a. Na and Fi ...
... c. E1> E2, as CaCl2 is an ionic bond b. E1< E2, as CaCl2 is a covalent bond d. E1< E2, as CaCl2 is an ionic bond 32. Given the pairs of atoms below, predict whether the bond formed between the atoms is either ionic or covalent, and if ionic, write the formula for the predicted compound. a. Na and Fi ...
111 Exam II Outline
... A. Gases are composed of such extremely tiny atoms or molecules that are widely separated by empty space. B. Gas particles move in a random,rapid, and continuous motion, thus has kinetic energy. C. Gas particles moves so rapidly and are so far apart the there is essentially no force of attraction be ...
... A. Gases are composed of such extremely tiny atoms or molecules that are widely separated by empty space. B. Gas particles move in a random,rapid, and continuous motion, thus has kinetic energy. C. Gas particles moves so rapidly and are so far apart the there is essentially no force of attraction be ...
How Do Gases Behave?
... 0 Kelvin: absolute zero (atom stops moving completely) Is there a maximum temperature in the universe? ...
... 0 Kelvin: absolute zero (atom stops moving completely) Is there a maximum temperature in the universe? ...
(1) Dissolves, accompanied by evolution of flammable gas (2
... The phase diagram for a pure substance is shown above. Use this diagram and your knowledge about changes of phase to answer the following questions. (a) What does point V represent? What characteristics are specific to the system only at point V ? (b) What does each point on the curve between V and ...
... The phase diagram for a pure substance is shown above. Use this diagram and your knowledge about changes of phase to answer the following questions. (a) What does point V represent? What characteristics are specific to the system only at point V ? (b) What does each point on the curve between V and ...
Lesson Plans - University High School
... describe temperature as a measure of the average kinetic energy of the particles in a substance describe matter at absolute zero (0 K) apply kinetic-molecular theory and intermolecular forces of attraction to explain the different properties of solids, liquids, and gases ○ definite/indefinite shape ...
... describe temperature as a measure of the average kinetic energy of the particles in a substance describe matter at absolute zero (0 K) apply kinetic-molecular theory and intermolecular forces of attraction to explain the different properties of solids, liquids, and gases ○ definite/indefinite shape ...
Every reaction is reversible: A chemical reaction is in equilibrium
... The mobile phase is a gas known as the 'carrier' gas, usually Nitrogen or Helium. The carrier gas drags the mixture into a heated coil packed with the stationery phase - a liquid (e.g. a silicone oil) held on a solid support (e.g. Celite). Careful control of the oven temperature ensures that volatil ...
... The mobile phase is a gas known as the 'carrier' gas, usually Nitrogen or Helium. The carrier gas drags the mixture into a heated coil packed with the stationery phase - a liquid (e.g. a silicone oil) held on a solid support (e.g. Celite). Careful control of the oven temperature ensures that volatil ...
SCH3U - Norbraten
... Write the word and skeletal equation for the following reactions. Be sure to include the proper states of matter by using the appropriate subscripts. You do not need to balance the equation. If the reaction does not occur write NO REACTION (NR) 10. Magnesium metal burns in air to produce solid magne ...
... Write the word and skeletal equation for the following reactions. Be sure to include the proper states of matter by using the appropriate subscripts. You do not need to balance the equation. If the reaction does not occur write NO REACTION (NR) 10. Magnesium metal burns in air to produce solid magne ...
Word and Skeleton Equations Practice (ws Fall 2010)
... Write the WORD and SKELETON equations for each chemical reaction. Indicate the state of each reactant and product in the skeleton equation. Remember that the following seven elements are diatomic: hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and ...
... Write the WORD and SKELETON equations for each chemical reaction. Indicate the state of each reactant and product in the skeleton equation. Remember that the following seven elements are diatomic: hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and ...
Sample Chem 111 Final
... c) The temperature of the water in both beakers is the same d) The boiling points of the water in the two beakers must be different. e) The temperature in the vigorously boiling water is not uniform. 57. How many sigma (σ) bonds are in the following molecule? a) 3 b) 7 c) 8 d) 9 e) 12 ...
... c) The temperature of the water in both beakers is the same d) The boiling points of the water in the two beakers must be different. e) The temperature in the vigorously boiling water is not uniform. 57. How many sigma (σ) bonds are in the following molecule? a) 3 b) 7 c) 8 d) 9 e) 12 ...
Introductory Chemistry: A Foundation FOURTH EDITION by Steven
... • Matter cannot be created or destroyed • In a chemical reaction, all the atoms present at the beginning are still present at the end • Therefore the total mass cannot change • Therefore the total mass of the reactants will be the same as the total mass of the ...
... • Matter cannot be created or destroyed • In a chemical reaction, all the atoms present at the beginning are still present at the end • Therefore the total mass cannot change • Therefore the total mass of the reactants will be the same as the total mass of the ...
Pollution Control - No Brain Too Small
... Collecting plates are designed to receive and retain the precipitated particles until they are intentionally removed into the hopper. Collecting plates are also part of the electrical power circuit of the precipitator. These collecting plate functions are incorporated into the precipitator design. ...
... Collecting plates are designed to receive and retain the precipitated particles until they are intentionally removed into the hopper. Collecting plates are also part of the electrical power circuit of the precipitator. These collecting plate functions are incorporated into the precipitator design. ...
Name ………………………………………………… Unit 7: States of
... Crude oil is a mixture of many hydrocarbons that have different numbers of carbon atoms. The use of a fractionating tower allows the separation of this mixture based on the boiling points of the hydrocarbons. To begin the separation process, the crude oil is heated to about 400°C in a furnace, caus ...
... Crude oil is a mixture of many hydrocarbons that have different numbers of carbon atoms. The use of a fractionating tower allows the separation of this mixture based on the boiling points of the hydrocarbons. To begin the separation process, the crude oil is heated to about 400°C in a furnace, caus ...
SC72 1st semester Study Guide
... DemocritusDaltonAtomic theoryThompson’s modelGold foil experimentRutherford’s modelBohr’s modelHypothesisTheoryLawPrinciplePractice 1. Draw and describe each step of the development of the atomic model. ...
... DemocritusDaltonAtomic theoryThompson’s modelGold foil experimentRutherford’s modelBohr’s modelHypothesisTheoryLawPrinciplePractice 1. Draw and describe each step of the development of the atomic model. ...
Students know
... stir and heating is just like stirring on the molecular level. C. Gas molecules move faster when heated and this causes them to move out of the solution so they don’t dissolve. D. Gas molecules are lower in energy than water, therefore when the gas molecules are heated they have an increase in their ...
... stir and heating is just like stirring on the molecular level. C. Gas molecules move faster when heated and this causes them to move out of the solution so they don’t dissolve. D. Gas molecules are lower in energy than water, therefore when the gas molecules are heated they have an increase in their ...
7.5.9 Compare physical properties of matter to the chemical property
... oThe boiling point for pure water at sea level is 100 degrees Celsius or 212 degrees Fahrenheit ...
... oThe boiling point for pure water at sea level is 100 degrees Celsius or 212 degrees Fahrenheit ...
AP Chemistry Summer Assignment THIS
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
the nakuru district sec. schools trial examinations - 2015
... 5. The use of materials of lead in roofing and in water pipes is being discouraged. State (i) One reason why these materials have been used in the past. (1 mark) Lead piping had unique ability to resist pin hole leaks lead does not rust/corrode forms a protective layer on its surface 6. State an ...
... 5. The use of materials of lead in roofing and in water pipes is being discouraged. State (i) One reason why these materials have been used in the past. (1 mark) Lead piping had unique ability to resist pin hole leaks lead does not rust/corrode forms a protective layer on its surface 6. State an ...
Resumen Science I Trimestre II Parcial Definitions: Element: pure
... Ratio: is the amount of elements in a compound. Element join in a specific ratio (amount of the element), according to their mass to form a compound. Ex. 16g Mixture: combination of 2 or more substances that are not chemically combined. (Physical change: so each substance has the same chemical makeu ...
... Ratio: is the amount of elements in a compound. Element join in a specific ratio (amount of the element), according to their mass to form a compound. Ex. 16g Mixture: combination of 2 or more substances that are not chemically combined. (Physical change: so each substance has the same chemical makeu ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.