the properties and structure of matter
... • Physical properties used to describe matter can be classified as: 1) Extensive – depends on the amount of matter in the sample - e.g. Mass, volume, length 2) Intensive – depends on the type of matter, not the amount present - Hardness, density, boiling point ...
... • Physical properties used to describe matter can be classified as: 1) Extensive – depends on the amount of matter in the sample - e.g. Mass, volume, length 2) Intensive – depends on the type of matter, not the amount present - Hardness, density, boiling point ...
AP Chemistry MC Review Questions
... 35. _____Represents an atom that is chemically unreactive 36. _____Represents an atom in an excited state 37. _____Represents an atom that has four valence electrons 38. _____Represents an atom of a transition metal ...
... 35. _____Represents an atom that is chemically unreactive 36. _____Represents an atom in an excited state 37. _____Represents an atom that has four valence electrons 38. _____Represents an atom of a transition metal ...
presentation on power generation from biogas in 2
... in the conversion of complex organic materials ultimately to methane 1 CH42 and CO2. Acetate and H2 + CO2 from primary fermentations can be directly converted to methane, although H2 + CO2 can also be consumed by homoacetogens. But note how the syntrophs play a key role in anoxic decomposition by co ...
... in the conversion of complex organic materials ultimately to methane 1 CH42 and CO2. Acetate and H2 + CO2 from primary fermentations can be directly converted to methane, although H2 + CO2 can also be consumed by homoacetogens. But note how the syntrophs play a key role in anoxic decomposition by co ...
File
... To set the properties right click and select SMART Response Question Object->Properties.. ...
... To set the properties right click and select SMART Response Question Object->Properties.. ...
Methane Production from Municipal Solid Waste
... Volatile solids (VS) content (determined by weight loss on ignition at 550°C) has been used to estimate the biodegradability of MSW components, but this measure overestimates the biodegradability of paper. Paper products have a very high volatile solids content. Newsprint, office paper, and cardboar ...
... Volatile solids (VS) content (determined by weight loss on ignition at 550°C) has been used to estimate the biodegradability of MSW components, but this measure overestimates the biodegradability of paper. Paper products have a very high volatile solids content. Newsprint, office paper, and cardboar ...
Bellin College Homework Supplement
... a. copper in copper wire b. a chocolate-chip cookie c. nitrox, a combination of oxygen and nitrogen used to fill scuba tanks 24. Classify each of the following as a physical or chemical change: a. A gold ingot is hammered to form gold leaf. b. Gasoline burns in air. c. Garlic is chopped into small p ...
... a. copper in copper wire b. a chocolate-chip cookie c. nitrox, a combination of oxygen and nitrogen used to fill scuba tanks 24. Classify each of the following as a physical or chemical change: a. A gold ingot is hammered to form gold leaf. b. Gasoline burns in air. c. Garlic is chopped into small p ...
CST REVIEW Percent Error 1. 2. What is the formula for density?
... 34. List the following in order of increasing electronegativity: K, Br, Ca, As 35. Atoms and molecules in liquids move in a random pattern relative to one another because the intermolecular forces are too weak to hold the atoms or molecules in a solid form. Under the same conditions of pressure and ...
... 34. List the following in order of increasing electronegativity: K, Br, Ca, As 35. Atoms and molecules in liquids move in a random pattern relative to one another because the intermolecular forces are too weak to hold the atoms or molecules in a solid form. Under the same conditions of pressure and ...
Name: Period:______ PHYSICAL SCIENCE 1st Semester Final
... Every sample of a given substance has the same properties because a substance has a fixed, uniform composition. An element has a fixed composition because it contains only one type of atom. A compound always contains two or more elements joined in a fixed proportion. The properties of a mixt ...
... Every sample of a given substance has the same properties because a substance has a fixed, uniform composition. An element has a fixed composition because it contains only one type of atom. A compound always contains two or more elements joined in a fixed proportion. The properties of a mixt ...
Effects of antioxidants for the degradation of flame
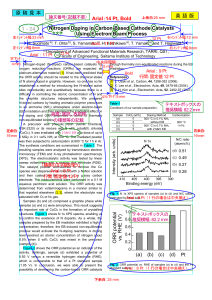
... Nitrogen-doped (N-doped) carbon-based catalysts for oxygen reduction reactions (ORRs) are expected as platinum-alternative material [1]. It has been predicted that the ORR activity should be related to the chemical states of N atoms doped in graphite. However, no one has so far established a method ...
... Nitrogen-doped (N-doped) carbon-based catalysts for oxygen reduction reactions (ORRs) are expected as platinum-alternative material [1]. It has been predicted that the ORR activity should be related to the chemical states of N atoms doped in graphite. However, no one has so far established a method ...
Spring Exam 2 - Chemistry
... This is VERY IMPORTANT! Under IDENTIFICATION NUMBER, put in your 8 DIGIT STUDENT ID NUMBER (do not use the 9 at the beginning of your number) beginning in column A and continuing through column H, column I will be blank, (do NOT use column J at this time); be sure to fill in the correct circles (a c ...
... This is VERY IMPORTANT! Under IDENTIFICATION NUMBER, put in your 8 DIGIT STUDENT ID NUMBER (do not use the 9 at the beginning of your number) beginning in column A and continuing through column H, column I will be blank, (do NOT use column J at this time); be sure to fill in the correct circles (a c ...
Review - cloudfront.net
... When two substances react to form products, the reactant which is used up is called the ____. a. determining reagent c. excess reagent b. limiting reagent d. catalytic reagent Why does the pressure inside a container of gas increase if more gas is added to the container? a. There is an increase in t ...
... When two substances react to form products, the reactant which is used up is called the ____. a. determining reagent c. excess reagent b. limiting reagent d. catalytic reagent Why does the pressure inside a container of gas increase if more gas is added to the container? a. There is an increase in t ...
Gas Volumes and the Ideal Gas Law
... atoms, which turns out to be correct. The simplest possible molecule of water indicated two hydrogen atoms and one oxygen atom per molecule, which is also correct. Experiments eventually showed that all elements that are gases near room temperature, except the noble gases, normally exist as diatomic ...
... atoms, which turns out to be correct. The simplest possible molecule of water indicated two hydrogen atoms and one oxygen atom per molecule, which is also correct. Experiments eventually showed that all elements that are gases near room temperature, except the noble gases, normally exist as diatomic ...
Chemistry 5350 Advanced Physical Chemistry Fall Semester 2013
... well. The well depth is a fraction of the total energy, and the molecule is unaffected by the attractive part of the potential. 2. Explain why attractive interactions between molecules in a gas make the pressure less than predicted by the ideal gas equation. The ideal gas equation assumes that the m ...
... well. The well depth is a fraction of the total energy, and the molecule is unaffected by the attractive part of the potential. 2. Explain why attractive interactions between molecules in a gas make the pressure less than predicted by the ideal gas equation. The ideal gas equation assumes that the m ...
Chemistry I Exam
... C. The distance travelled per hour is constant. D. The student rested for one hour half way through the trip. E. The graph is a curve rather than a straight line because the speed of the bicycle decreased as the time of day increased. ...
... C. The distance travelled per hour is constant. D. The student rested for one hour half way through the trip. E. The graph is a curve rather than a straight line because the speed of the bicycle decreased as the time of day increased. ...
SAMPLE QUESTION PAPER CHEMISTRY (043) CLASS XII (2013-14)
... ½ T1/2 = 3465 s ½ 22. (a) chemical reaction the metal is converted to a compound, which forms a vapour, which is decomposed to get pure metal ...
... ½ T1/2 = 3465 s ½ 22. (a) chemical reaction the metal is converted to a compound, which forms a vapour, which is decomposed to get pure metal ...
Show - Evonik
... Safety Summary is supplied on the condition that the persons receiving the same will make their own determination as to its suitability for their purposes prior to use. This GPS Safety Summary does not supersede or replace required regulatory and/or legal communication documents. Performance of the ...
... Safety Summary is supplied on the condition that the persons receiving the same will make their own determination as to its suitability for their purposes prior to use. This GPS Safety Summary does not supersede or replace required regulatory and/or legal communication documents. Performance of the ...
432 Final Exam Study Guide
... D. an increase in the average kinetic energy of the particles ___3. Evaporation is type of A. boiling B. vaporization C. sublimation D. condensation ___4. Compared with gases, liquids: A. have stronger intermolecular attractions. B. have more space between their particles. C. are much less dense. D. ...
... D. an increase in the average kinetic energy of the particles ___3. Evaporation is type of A. boiling B. vaporization C. sublimation D. condensation ___4. Compared with gases, liquids: A. have stronger intermolecular attractions. B. have more space between their particles. C. are much less dense. D. ...
MP 2 workbook 2016
... Matter is anything that has mass and takes up space. It includes all the physical “stuff” around us—rocks, houses, water, air, people, ants, trees, etc. All matter can exist as a solid, a liquid, or a gas. Matter includes all the things we can weigh (to find the mass) and measure to find the volume ...
... Matter is anything that has mass and takes up space. It includes all the physical “stuff” around us—rocks, houses, water, air, people, ants, trees, etc. All matter can exist as a solid, a liquid, or a gas. Matter includes all the things we can weigh (to find the mass) and measure to find the volume ...
Chapter 9 Atomic Absorption and Atomic Fluorescence Spectrometry
... follow Beer’s law with absorbance being directly proportional to concentration. In fact, however, departures from linearity are often encountered, and it is foolhardy to perform an atomic absorption analysis without experimentally determining whether or not a linear relationship does exist. A calibr ...
... follow Beer’s law with absorbance being directly proportional to concentration. In fact, however, departures from linearity are often encountered, and it is foolhardy to perform an atomic absorption analysis without experimentally determining whether or not a linear relationship does exist. A calibr ...
Fall Final Rev 2014
... iv. Molecules are moving faster, so they strike container walls more often and with greater force. v. Molecules are in constant random motion, so they eventually distribute throughout entire container, no matter how large it is. b. 738 mm Hg c. Both have same kinetic energy (same temperature); Xe at ...
... iv. Molecules are moving faster, so they strike container walls more often and with greater force. v. Molecules are in constant random motion, so they eventually distribute throughout entire container, no matter how large it is. b. 738 mm Hg c. Both have same kinetic energy (same temperature); Xe at ...
PRACTICE EXAM for FALL 2013 FINAL EXAM (Unit 6 + review) 1
... iv. Molecules are moving faster, so they strike container walls more often and with greater force. v. Molecules are in constant random motion, so they eventually distribute throughout entire container, no matter how large it is. b. 738 mm Hg c. Both have same kinetic energy (same temperature); Xe at ...
... iv. Molecules are moving faster, so they strike container walls more often and with greater force. v. Molecules are in constant random motion, so they eventually distribute throughout entire container, no matter how large it is. b. 738 mm Hg c. Both have same kinetic energy (same temperature); Xe at ...
7 - Mona Shores Blogs
... Assume hematite is the only source of iron in this ore. (A) 1.11 mol (B) 1059 mol (C) 3.18 mol (D) 6.36 mol 14. A 2.0 mL sample of HCl(g) is mixed with a 1.5 mL of NH3(g). What is the volume if the resulting mixture after the reaction is complete? (Assume all measurements are carried out at the same ...
... Assume hematite is the only source of iron in this ore. (A) 1.11 mol (B) 1059 mol (C) 3.18 mol (D) 6.36 mol 14. A 2.0 mL sample of HCl(g) is mixed with a 1.5 mL of NH3(g). What is the volume if the resulting mixture after the reaction is complete? (Assume all measurements are carried out at the same ...
Chemical Reactions
... Count the number and type of each type of atom on both sides If there are different numbers of atoms on each side, you must add coefficients to compounds to change the number of atoms Figure out what number to multiply each compound by in order to make the numbers of atoms add up Remember: you can ...
... Count the number and type of each type of atom on both sides If there are different numbers of atoms on each side, you must add coefficients to compounds to change the number of atoms Figure out what number to multiply each compound by in order to make the numbers of atoms add up Remember: you can ...
Ductility-the ability to be stretched into wires
... return to its original form after being stretched (__.P) ...
... return to its original form after being stretched (__.P) ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.