Kinetics
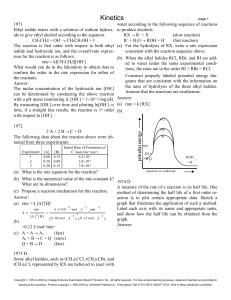
... CH3CH2I + OH- CH3CH2OH + IThe reaction is first order with respect to both ethyl iodide and hydroxide ion, and the overall-rate expression for the reaction is as follows: rate = k[CH3CH2I][OH-] What would you do in the laboratory to obtain data to confirm the order in the rate expression for eithe ...
... CH3CH2I + OH- CH3CH2OH + IThe reaction is first order with respect to both ethyl iodide and hydroxide ion, and the overall-rate expression for the reaction is as follows: rate = k[CH3CH2I][OH-] What would you do in the laboratory to obtain data to confirm the order in the rate expression for eithe ...
GLASS AND CERAMIC MATERIALS Study Support Jozef Vlček
... towards molten glass. Both actions, creation of glacial nucleus as well as the growth of crystals are dependent on temperature. Maximum rate of both actions are not present by the same temperature, more in fig. 4. In most cases is the maximal rate of creation of nucleus recorded by lower temperature ...
... towards molten glass. Both actions, creation of glacial nucleus as well as the growth of crystals are dependent on temperature. Maximum rate of both actions are not present by the same temperature, more in fig. 4. In most cases is the maximal rate of creation of nucleus recorded by lower temperature ...
(K c ) [A] - Knockhardy
... CHEMICAL EQUILIBRIUM (2) INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at AS and A2 level Chemistry. It is based on the requirements of the AQA and OCR specifications but is suitable for other examination boards. Individual students may use ...
... CHEMICAL EQUILIBRIUM (2) INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at AS and A2 level Chemistry. It is based on the requirements of the AQA and OCR specifications but is suitable for other examination boards. Individual students may use ...
Chapter 14
... A) oxygen is a reactant in combustion and concentration of oxygen is higher in pure oxygen than is in air. B) oxygen is a catalyst for combustion. C) oxygen is a product of combustion. D) nitrogen is a product of combustion and the system reaches equilibrium at a lower temperature. E) nitrogen is a ...
... A) oxygen is a reactant in combustion and concentration of oxygen is higher in pure oxygen than is in air. B) oxygen is a catalyst for combustion. C) oxygen is a product of combustion. D) nitrogen is a product of combustion and the system reaches equilibrium at a lower temperature. E) nitrogen is a ...
Student Study Guide Chemistry 534
... Gases play a very important role in our daily existence. Since we are surrounded by an ocean of gas called the atmosphere, many of the properties of gases are already familiar to us. We know that we can squeeze a balloon into a smaller shape and that perfume released into the corner of a room can, i ...
... Gases play a very important role in our daily existence. Since we are surrounded by an ocean of gas called the atmosphere, many of the properties of gases are already familiar to us. We know that we can squeeze a balloon into a smaller shape and that perfume released into the corner of a room can, i ...
Thermodynamics: the Second Law
... to it, and water can be decomposed by the passage of an electric current. However, in each case we need to act in some way on the system to bring about the non-spontaneous change. There must be some feature of the world that accounts for the distinction between the two types of change. Throughout th ...
... to it, and water can be decomposed by the passage of an electric current. However, in each case we need to act in some way on the system to bring about the non-spontaneous change. There must be some feature of the world that accounts for the distinction between the two types of change. Throughout th ...
CHAPTER-7
... Ans. 1.If QcKc net reaction goes from right to left (towards backward direction)
3. If Qc=Kc no net reaction occurs (reaction is at equilibrium)
14) State le Chatelier’s principle?
Ans. If a system under equilibrium is sub ...
... Ans. 1.If Qc
Ch 10 Practice Problems 1. Consider the process A(l) A(s). Which
... For the reaction A + B C + D, H° = +40 kJ and S° = +50 J/K. Therefore, the reaction under standard conditions is A) spontaneous at temperatures less than 10 K. B) spontaneous at temperatures greater than 800 K. C) spontaneous only at temperatures between 10 K and 800 K. D) spontaneous at all tem ...
... For the reaction A + B C + D, H° = +40 kJ and S° = +50 J/K. Therefore, the reaction under standard conditions is A) spontaneous at temperatures less than 10 K. B) spontaneous at temperatures greater than 800 K. C) spontaneous only at temperatures between 10 K and 800 K. D) spontaneous at all tem ...
(Monel) Thermal Spray Wire
... plasma spray, provide superior protection against acidic and alkaline solutions, particularly when sealed. However, caution should be exercised in applications were mechanical fatigue may be a factor. Such products include Metco 101, Metco 6200, Metco 6203, Amdry 187 and Amdry 6208. nn Coatings h ...
... plasma spray, provide superior protection against acidic and alkaline solutions, particularly when sealed. However, caution should be exercised in applications were mechanical fatigue may be a factor. Such products include Metco 101, Metco 6200, Metco 6203, Amdry 187 and Amdry 6208. nn Coatings h ...
Burning a Candle in a Vessel, a Simple Experiment
... that after the flame goes out there is enough oxygen in the enclosed volume to allow a mouse to breathe without difficulties (Birk and Lawson 1999), an experiment also reported in the times of Lavoisier by Priestley. The candle experiment cannot be used to demonstrate that the oxygen content of air ...
... that after the flame goes out there is enough oxygen in the enclosed volume to allow a mouse to breathe without difficulties (Birk and Lawson 1999), an experiment also reported in the times of Lavoisier by Priestley. The candle experiment cannot be used to demonstrate that the oxygen content of air ...
Chapter 14
... Because there are standard ways of find the change in entropy for a pure substance as we change the temperature of the substance at constant pressure, the third law of thermodynamics allows us to assign values for entropy for pure substances at any temperature. Standard molar entropy (S) – The valu ...
... Because there are standard ways of find the change in entropy for a pure substance as we change the temperature of the substance at constant pressure, the third law of thermodynamics allows us to assign values for entropy for pure substances at any temperature. Standard molar entropy (S) – The valu ...
Thermochemistry Exam Review Questions
... 3. The reaction: 2S(s) + 3O2 (g) ↔ 2SO3(g) + 800 kJ has reached the equilibrium in a closed container. Which change will shift the equilibrium to the right? A. adding a catalyst without changing the temperature or pressure B. increasing the pressure by reducing the volume C. increasing the temperatu ...
... 3. The reaction: 2S(s) + 3O2 (g) ↔ 2SO3(g) + 800 kJ has reached the equilibrium in a closed container. Which change will shift the equilibrium to the right? A. adding a catalyst without changing the temperature or pressure B. increasing the pressure by reducing the volume C. increasing the temperatu ...
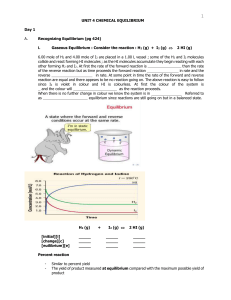
Unit 4 - Chemical Equilibrium
... When the _ _ _ _ _ in a chemical reaction looks like this ⇌ , it shows that the reaction is _ _ _ _ _ _ _ _ _ _. This means the products can react together and turn back into the _ _ _ _ _ _ _ _ reactants. In other words, the reaction can go _ _ _ _ ways. When a reversible reaction is set up in a _ ...
... When the _ _ _ _ _ in a chemical reaction looks like this ⇌ , it shows that the reaction is _ _ _ _ _ _ _ _ _ _. This means the products can react together and turn back into the _ _ _ _ _ _ _ _ reactants. In other words, the reaction can go _ _ _ _ ways. When a reversible reaction is set up in a _ ...
Answer
... The 1st reaction is doubled so the original equilibrium constant is squared. The 2nd reaction is reversed so the reciprocal of the equilibrium constant is used. The two reactions are then combined and the overall equilibrium constant is then the product: Kc(3) = Kc(1) × Kc(2) = (49.0)2 × (1/(1.075 × ...
... The 1st reaction is doubled so the original equilibrium constant is squared. The 2nd reaction is reversed so the reciprocal of the equilibrium constant is used. The two reactions are then combined and the overall equilibrium constant is then the product: Kc(3) = Kc(1) × Kc(2) = (49.0)2 × (1/(1.075 × ...
TiO2奈米光觸媒濾網應用於空調系統分解甲醛氣體之研究
... distance between pores of 1 mm (0.00328ft), was cleaned using ethanol and DI-water. b. A 1 wt. % solution of water-soluble polymer binder (QFnano Co., Ltd, Taiwan) was prepared, and evenly applied by spray onto the stainless steel metallic filters, which were then baked at 50°C (122°F) for 10 min to ...
... distance between pores of 1 mm (0.00328ft), was cleaned using ethanol and DI-water. b. A 1 wt. % solution of water-soluble polymer binder (QFnano Co., Ltd, Taiwan) was prepared, and evenly applied by spray onto the stainless steel metallic filters, which were then baked at 50°C (122°F) for 10 min to ...
PDF of Chapter 6 Foundations of Chemistry
... Have you ever seen a rock like the one in Figure 1? Why are different parts of the rock different in color? Why might some parts of the rock feel harder than other parts? The parts of the rock look and feel different because they are made of different types of matter. Matter is anything that has mas ...
... Have you ever seen a rock like the one in Figure 1? Why are different parts of the rock different in color? Why might some parts of the rock feel harder than other parts? The parts of the rock look and feel different because they are made of different types of matter. Matter is anything that has mas ...
A Low-Fluorine Solution with the F/Ba Mole Ratio of 2 for the
... spectra (ATR-FT-IR) were taken with a PerkinCElmer Spectrum GX FTIR system to detect the presence of BaCO3 . This sensitive technique allows the detection of BaCO3 at a level as low as 2 wt% [22]. Differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) were carried out using a N ...
... spectra (ATR-FT-IR) were taken with a PerkinCElmer Spectrum GX FTIR system to detect the presence of BaCO3 . This sensitive technique allows the detection of BaCO3 at a level as low as 2 wt% [22]. Differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) were carried out using a N ...
Equilibrium
... It may be tempting to think that once equilibrium has been reached, the reaction stops. However, chemical equilibrium is a dynamic process. The forward and reverse reactions continue to occur even after equilibrium has been reached, but, because the rates of the two reactions are equal, there is no ...
... It may be tempting to think that once equilibrium has been reached, the reaction stops. However, chemical equilibrium is a dynamic process. The forward and reverse reactions continue to occur even after equilibrium has been reached, but, because the rates of the two reactions are equal, there is no ...
General Concepts of Chemical Equilibrium
... common ion is usually a product ion which means that reactants concentration will increase on the expense of products. Let us look at the following example: ...
... common ion is usually a product ion which means that reactants concentration will increase on the expense of products. Let us look at the following example: ...
CHEM 1412. Chapter 15. Chemical Equilibrium (Homework)
... (Homework) Ky35 Key 1. Which is the correct equilibrium constant expression for the following reaction? Fe2O3 (s) + 3H2 (g) 2Fe (s) + 3H2O (g) A. Kc = [Fe2O3] [H2]3 / [Fe]2[H2O]3 B. Kc = [H2] / [H2O] C. Kc = [H2O]3 / [H2]3 D. Kc = [Fe]2[H2O]3 / [Fe2O3] [H2]3 E. Kc = [Fe] [H2O] / [Fe2O3] [H2] ...
... (Homework) Ky35 Key 1. Which is the correct equilibrium constant expression for the following reaction? Fe2O3 (s) + 3H2 (g) 2Fe (s) + 3H2O (g) A. Kc = [Fe2O3] [H2]3 / [Fe]2[H2O]3 B. Kc = [H2] / [H2O] C. Kc = [H2O]3 / [H2]3 D. Kc = [Fe]2[H2O]3 / [Fe2O3] [H2]3 E. Kc = [Fe] [H2O] / [Fe2O3] [H2] ...
Document
... come to rest in the center of the bowl. The ball has reached static equilibrium. Static equilibrium is a state in which nothing changes. Chemical equilibrium is different from static equilibrium because it is dynamic. In a dynamic equilibrium, there is no net change in the system. Two opposite chang ...
... come to rest in the center of the bowl. The ball has reached static equilibrium. Static equilibrium is a state in which nothing changes. Chemical equilibrium is different from static equilibrium because it is dynamic. In a dynamic equilibrium, there is no net change in the system. Two opposite chang ...
CHE 1402 Lab Manual
... 4. Write the ideal-gas equation and give the units for each term when R = 0.0821 L-atm /mol-K. 5. Show by mathematical equations how one can determine the molar mass of a volatile liquid by measurement of the pressure, volume, temperature, and weight of the liquid. 6. If 0.75 g of a gas occupies 300 ...
... 4. Write the ideal-gas equation and give the units for each term when R = 0.0821 L-atm /mol-K. 5. Show by mathematical equations how one can determine the molar mass of a volatile liquid by measurement of the pressure, volume, temperature, and weight of the liquid. 6. If 0.75 g of a gas occupies 300 ...
國 立 交 通 大 學
... below nanometer technology. The requirements for shallow trench isolation (STI) include voids free gap filling, better conformal step coverage, low wet etch rate and metallic contamination. It is found that HDP is increasingly finding it difficult to provide a good gap-fill solution for the more agg ...
... below nanometer technology. The requirements for shallow trench isolation (STI) include voids free gap filling, better conformal step coverage, low wet etch rate and metallic contamination. It is found that HDP is increasingly finding it difficult to provide a good gap-fill solution for the more agg ...
Equilibrium Notes - Chemistry Teaching Resources
... reactions by the same amount and there is no change in the equilibrium concentrations. The position of the equilibrium is therefore unaltered and the value of K at a specific temperature remains unchanged. All a catalyst does is to speed up the rate at which equilibrium is established. K and the pos ...
... reactions by the same amount and there is no change in the equilibrium concentrations. The position of the equilibrium is therefore unaltered and the value of K at a specific temperature remains unchanged. All a catalyst does is to speed up the rate at which equilibrium is established. K and the pos ...
Chapter 14 Review
... B. Increasing the temperature shifts the equilibrium to the right. C. A catalyst speeds up the approach to equilibrium and shifts the position of equilibrium to the right. D. Decreasing the total pressure of the system shifts the equilibrium to the right. E. Increasing the temperature causes the equ ...
... B. Increasing the temperature shifts the equilibrium to the right. C. A catalyst speeds up the approach to equilibrium and shifts the position of equilibrium to the right. D. Decreasing the total pressure of the system shifts the equilibrium to the right. E. Increasing the temperature causes the equ ...

![(K c ) [A] - Knockhardy](http://s1.studyres.com/store/data/011755527_1-914ea907d1ff7656ef398ad87316c94c-300x300.png)