Ozone Writeup - International Research Institute for Climate and
... played a part in depleting the ozone layer. Many factors play into ozone depletion and formation and will be discussed in this paper: location of ozone depletion, the role of greenhouse gases, chemistry, sunlight, and temperature. The Arctic and Antarctic vary slightly concerning their abundance of ...
... played a part in depleting the ozone layer. Many factors play into ozone depletion and formation and will be discussed in this paper: location of ozone depletion, the role of greenhouse gases, chemistry, sunlight, and temperature. The Arctic and Antarctic vary slightly concerning their abundance of ...
Determination of the Vapor Pressure of Triacetone Triperoxide (TATP)
... The ambient TATP vapor pressure ~ 0.05 mm Hg or ~6 ng per 10 µL of air is an amount readily observable with modern laboratory instrumentation. Computing heat of sublimation as above, we find )Hsub equal to 109 kJ/mol. Among military explosives TNT is quite volatile, making its detection relatively e ...
... The ambient TATP vapor pressure ~ 0.05 mm Hg or ~6 ng per 10 µL of air is an amount readily observable with modern laboratory instrumentation. Computing heat of sublimation as above, we find )Hsub equal to 109 kJ/mol. Among military explosives TNT is quite volatile, making its detection relatively e ...
Rutile titanium dioxide nanoparticles and ordered acicular
... shaped rutile TiO2 nanoparticles produced according to Example 1 and shown in FIG. 1. FIG. 6 is an SEM image of the shaped rutile TiO2 nanopar ticles shown in FIG. 1 after calcining at 550° C. for 6 hours. FIG. 7 is an enlarged SEM image of the shaped rutile TiO2 nanoparticles shown in FIG. 6. FIG. ...
... shaped rutile TiO2 nanoparticles produced according to Example 1 and shown in FIG. 1. FIG. 6 is an SEM image of the shaped rutile TiO2 nanopar ticles shown in FIG. 1 after calcining at 550° C. for 6 hours. FIG. 7 is an enlarged SEM image of the shaped rutile TiO2 nanoparticles shown in FIG. 6. FIG. ...
Chapter 8: Balances on Nonreactive Processes
... Hydrogen produced by steam-methane reforming exits at a pressure of 1.6 MPa and a temperature of 855°C. The hydrogen is stored in a metal hydride tank with a volume of 70 m3 and must be cooled down to a temperature of 30°C before being fed to a proton exchange membrane fuel cell. Determine the amoun ...
... Hydrogen produced by steam-methane reforming exits at a pressure of 1.6 MPa and a temperature of 855°C. The hydrogen is stored in a metal hydride tank with a volume of 70 m3 and must be cooled down to a temperature of 30°C before being fed to a proton exchange membrane fuel cell. Determine the amoun ...
Chapter 15 Chemical Equilibrium
... concentrations are known, calculate the change in concentration that occurs as the system reaches equilibrium. 3. Use the stoichiometry of the reaction (that is, use the coefficients in the balanced chemical equation) to calculate the changes in concentration of all the other species in the equilibr ...
... concentrations are known, calculate the change in concentration that occurs as the system reaches equilibrium. 3. Use the stoichiometry of the reaction (that is, use the coefficients in the balanced chemical equation) to calculate the changes in concentration of all the other species in the equilibr ...
LABORATORY MANUAL FOR CHEMISTRY 102
... to the sum of x + y + z +... .) The term k is known as the rate constant for the reaction. Usually, when a reaction is initiated, the rate (known as the initial rate) is found to be at its maximum value. As the reaction progresses, reactants are consumed (lowering their concentrations) and the rate ...
... to the sum of x + y + z +... .) The term k is known as the rate constant for the reaction. Usually, when a reaction is initiated, the rate (known as the initial rate) is found to be at its maximum value. As the reaction progresses, reactants are consumed (lowering their concentrations) and the rate ...
equilibrium - eVirtualGuru
... The double half arrows indicate that the processes in both the directions are going on simultaneously. The mixture of reactants and products in the equilibrium state is called an equilibrium mixture. Equilibrium can be established for both physical processes and chemical reactions. The reaction may ...
... The double half arrows indicate that the processes in both the directions are going on simultaneously. The mixture of reactants and products in the equilibrium state is called an equilibrium mixture. Equilibrium can be established for both physical processes and chemical reactions. The reaction may ...
Chemical Equilibrium - 2012 Book Archive
... Consider the following reaction occurring in a closed container (so that no material can go in or out): H2 + I2 → 2HI This is simply the reaction between elemental hydrogen and elemental iodine to make hydrogen iodide. The way the equation is written, we are led to believe that the reaction goes to ...
... Consider the following reaction occurring in a closed container (so that no material can go in or out): H2 + I2 → 2HI This is simply the reaction between elemental hydrogen and elemental iodine to make hydrogen iodide. The way the equation is written, we are led to believe that the reaction goes to ...
Chemical Equilibrium - local.brookings.k12.sd.us
... reaction results in the almost ________ complete conversion of ________ reactants to ________, products the 1N2 (g) + 3H2(g) 2NH3(g) ________ reaction is said to go to completion but _____ most _________ reactions __________, 1N2 (g) + 3H2(g) 2NH3(g) ___ do ____ not go to __________, completion most ...
... reaction results in the almost ________ complete conversion of ________ reactants to ________, products the 1N2 (g) + 3H2(g) 2NH3(g) ________ reaction is said to go to completion but _____ most _________ reactions __________, 1N2 (g) + 3H2(g) 2NH3(g) ___ do ____ not go to __________, completion most ...
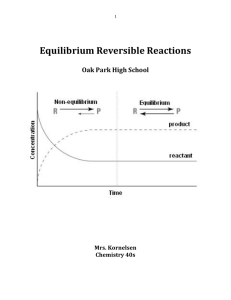
Equilibrium Reversible Reactions
... progress of a reaction. For reactions involving a solid or a liquid, while the amounts of the solid or liquid will change during a reaction, their concentrations (much like their densities) will not change during the reaction. • Instead, their values will remain constant. Because they are constant, ...
... progress of a reaction. For reactions involving a solid or a liquid, while the amounts of the solid or liquid will change during a reaction, their concentrations (much like their densities) will not change during the reaction. • Instead, their values will remain constant. Because they are constant, ...
Chapter 15: Chemical Equilibrium
... The equilibrium concentrations of ammonia and hydrogen chloride do not depend on the amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporate ...
... The equilibrium concentrations of ammonia and hydrogen chloride do not depend on the amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporate ...
Chapter 18 pdf
... The dynamic nature of equilibrium Equilibrium is a state of action, not inaction. For example, consider this analogy. The Golden Gate Bridge, shown in Figure 18-3, connects two California cities, San Francisco and Sausalito. Suppose that all roads leading into and out of the two cities are closed fo ...
... The dynamic nature of equilibrium Equilibrium is a state of action, not inaction. For example, consider this analogy. The Golden Gate Bridge, shown in Figure 18-3, connects two California cities, San Francisco and Sausalito. Suppose that all roads leading into and out of the two cities are closed fo ...
10. Solution Guide to Supplementary Exercises
... The direction in which a net reaction will proceed to achieve equilibrium can be predicted by constant (Kc). ...
... The direction in which a net reaction will proceed to achieve equilibrium can be predicted by constant (Kc). ...
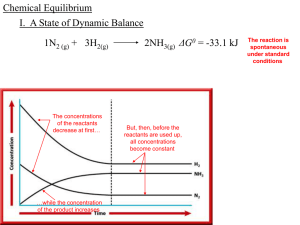
Chapter 18: Chemical Equilibrium
... time. The reactants, H2 and N2, are consumed in the reaction, so their concentrations gradually decrease. After a period of time, however, the concentrations of H2, N2, and NH3 no longer change. All concentrations become constant, as shown by the horizontal lines on the right side of the diagram. Th ...
... time. The reactants, H2 and N2, are consumed in the reaction, so their concentrations gradually decrease. After a period of time, however, the concentrations of H2, N2, and NH3 no longer change. All concentrations become constant, as shown by the horizontal lines on the right side of the diagram. Th ...
Solutions for Chapter 8 End-of-Chapter Problems
... within the dried fruit. (d) You get thirsty and your skin wrinkles when you swim in the ocean, because the salty ocean water is more concentrated than your body fluids and water is transferred by osmosis through the semipermeable cell membranes in your skin into the ocean. The loss of water from the ...
... within the dried fruit. (d) You get thirsty and your skin wrinkles when you swim in the ocean, because the salty ocean water is more concentrated than your body fluids and water is transferred by osmosis through the semipermeable cell membranes in your skin into the ocean. The loss of water from the ...
CHEM 1411 – STUDY-GUIDE-for-TEST-2
... 31. Which of the following statements is consistent with Boyle's Law concerning an ideal gas? A) At constant temperature and moles, a plot of volume versus pressure is linear. B) At constant pressure and volume, a plot of temperature versus moles is linear. C) At constant pressure and moles, a plot ...
... 31. Which of the following statements is consistent with Boyle's Law concerning an ideal gas? A) At constant temperature and moles, a plot of volume versus pressure is linear. B) At constant pressure and volume, a plot of temperature versus moles is linear. C) At constant pressure and moles, a plot ...
Synthesis and thermal decarbonylation of W(CO)5 complexes
... the complexes, that showed a distinct glass transition with a Tg that depended on the metal content and also on the temperature range chosen for the running cycles (Table 1). It has been established that the Tg of a polyphosphazene bearing a complex should be higher than that of the corresponding po ...
... the complexes, that showed a distinct glass transition with a Tg that depended on the metal content and also on the temperature range chosen for the running cycles (Table 1). It has been established that the Tg of a polyphosphazene bearing a complex should be higher than that of the corresponding po ...
wiley_ch6_Chemical_Equilibrium
... disturbance (Q ≠ K) will shift to offset stress System said to “shift to right” when forward reaction is dominant (Q < K) System said to “shift to left” when reverse direction is dominant (Q > K) Jespersen/Brady/Hyslop ...
... disturbance (Q ≠ K) will shift to offset stress System said to “shift to right” when forward reaction is dominant (Q < K) System said to “shift to left” when reverse direction is dominant (Q > K) Jespersen/Brady/Hyslop ...
High Performance Plastics for the Chemical Processing Industry
... molecules (crystallinity). As stated, the degree of branching between the molecular chains and the length of the side chains significantly influence the level of crystallinity and density of the polyethylene. An example of the effects of these differences in terms of physical properties can be seen ...
... molecules (crystallinity). As stated, the degree of branching between the molecular chains and the length of the side chains significantly influence the level of crystallinity and density of the polyethylene. An example of the effects of these differences in terms of physical properties can be seen ...
Chemical Engineering Thermodynamics II
... temperature, and the object that receives energy is at a lower temperature. We would be able to observe that the electrical resistance of the warmer object decreases with time, and that of the colder block increases with time; eventually there would be no change in the electrical resistances of thes ...
... temperature, and the object that receives energy is at a lower temperature. We would be able to observe that the electrical resistance of the warmer object decreases with time, and that of the colder block increases with time; eventually there would be no change in the electrical resistances of thes ...
Li−Fe−P−O2 Phase Diagram from First Principles Calculations
... using ab initio methods. The ground-state energies of all known compounds in the Li-Fe-P-O2 system were calculated using the generalized gradient approximation (GGA) approximation to density functional theory (DFT) and the DFT+U extension to it. Considering only the entropy of gaseous phases, the ph ...
... using ab initio methods. The ground-state energies of all known compounds in the Li-Fe-P-O2 system were calculated using the generalized gradient approximation (GGA) approximation to density functional theory (DFT) and the DFT+U extension to it. Considering only the entropy of gaseous phases, the ph ...
The Process of Chemical Reactions
... absorb energy overall as it takes place. If this energy comes from the motion (kinetic energy) of the reactants, the particles in the system will be moving more slowly after the reaction than before. The system will have lower kinetic energy, and the temperature will decrease. Because the system is ...
... absorb energy overall as it takes place. If this energy comes from the motion (kinetic energy) of the reactants, the particles in the system will be moving more slowly after the reaction than before. The system will have lower kinetic energy, and the temperature will decrease. Because the system is ...
Covert Chemical... 2_Couvertures English chimie 4
... Chemical Reactions 2: Equilibrium and Oxidation-reduction is the third of the three Learning Guides for the Secondary V Chemistry program, which comprises the following three courses: Gases Chemical Reactions 1: Energy and Chemical Dynamics Chemical Reactions 2: Equilibrium and Oxidation-reduction ...
... Chemical Reactions 2: Equilibrium and Oxidation-reduction is the third of the three Learning Guides for the Secondary V Chemistry program, which comprises the following three courses: Gases Chemical Reactions 1: Energy and Chemical Dynamics Chemical Reactions 2: Equilibrium and Oxidation-reduction ...
The Process of Chemical Reactions
... absorb energy overall as it takes place. If this energy comes from the motion (kinetic energy) of the reactants, the particles in the system will be moving more slowly after the reaction than before. The system will have lower kinetic energy, and the temperature will decrease. Because the system is ...
... absorb energy overall as it takes place. If this energy comes from the motion (kinetic energy) of the reactants, the particles in the system will be moving more slowly after the reaction than before. The system will have lower kinetic energy, and the temperature will decrease. Because the system is ...