purdue university - IUPUI ScholarWorks
... I certify that in the preparation of this thesis, I have observed the provisions of Purdue University Teaching, Research, and Outreach Policy on Research Misconduct (VIII.3.1), October 1, 2008.* Further, I certify that this work is free of plagiarism and all materials appearing in this thesis/disser ...
... I certify that in the preparation of this thesis, I have observed the provisions of Purdue University Teaching, Research, and Outreach Policy on Research Misconduct (VIII.3.1), October 1, 2008.* Further, I certify that this work is free of plagiarism and all materials appearing in this thesis/disser ...
Physiological and phylogenetic studies of thermophilic
... The present study is based on two earlier projects. One, supervised by Ólafur Friðjónsson at Prokaria ehf, was based on the study of three mesophilic hydrogen oxidizing (HOX) bacteria, Wautersia eutropha, Hydrogenomonas pseudoflava and Acidovorax facilis. This research emphasised on genetic engineer ...
... The present study is based on two earlier projects. One, supervised by Ólafur Friðjónsson at Prokaria ehf, was based on the study of three mesophilic hydrogen oxidizing (HOX) bacteria, Wautersia eutropha, Hydrogenomonas pseudoflava and Acidovorax facilis. This research emphasised on genetic engineer ...
Kinetic Assay of Human Pepsin with Albumin
... comparisons with results from other investigative centers. In general, research groups have tended to establish their own reference ranges for the given experiment, with the result that the reported values often do not have universal applicability. Moreover, no pure, stable human pepsin is yet avail ...
... comparisons with results from other investigative centers. In general, research groups have tended to establish their own reference ranges for the given experiment, with the result that the reported values often do not have universal applicability. Moreover, no pure, stable human pepsin is yet avail ...
File
... Changes in Concentration and Keq Now, as you know, changing the concentration of one of the reactants or products will cause the reaction to shift right or left. But this does not change the value for Keq as long as the temperature remains constant. Consider the reaction: ...
... Changes in Concentration and Keq Now, as you know, changing the concentration of one of the reactants or products will cause the reaction to shift right or left. But this does not change the value for Keq as long as the temperature remains constant. Consider the reaction: ...
Determination of Equilibrium Constants for Reactions between Nitric
... and the dissociation constant of ammonium, the cobalt(II) ammonia system can be established. A detailed calculation scheme can be found in the SI. Of the complexes with different coordination numbers in the cobalt(II) ammonia system, only penta- and hexaamminecobalt(II) ions were referred to as react ...
... and the dissociation constant of ammonium, the cobalt(II) ammonia system can be established. A detailed calculation scheme can be found in the SI. Of the complexes with different coordination numbers in the cobalt(II) ammonia system, only penta- and hexaamminecobalt(II) ions were referred to as react ...
Massachusetts Tests for Educator Licensure (MTEL )
... because degradation rates provided by the manufacturer may be biased and also because the degradation rates of the two types of plastic were likely measured using different environmental conditions. D is incorrect because the experiment in this option only involves one type of plastic. ...
... because degradation rates provided by the manufacturer may be biased and also because the degradation rates of the two types of plastic were likely measured using different environmental conditions. D is incorrect because the experiment in this option only involves one type of plastic. ...
Types of Chemical Reactions
... electrolytes as separate independent ions. This is a more accurate representation of the way electrolytes behave in solution. – A complete ionic equation is a chemical equation in which strong electrolytes (such as soluble ionic compounds, strong acids/bases) are written as separate ions in solution ...
... electrolytes as separate independent ions. This is a more accurate representation of the way electrolytes behave in solution. – A complete ionic equation is a chemical equation in which strong electrolytes (such as soluble ionic compounds, strong acids/bases) are written as separate ions in solution ...
Date: 16 / 01 / 2014 - Qatar University QSpace
... series of experiments were carried out to study the variation of preparation parameters such as support type, temperature, ‘ion-exchange’ time and the concentration of the (precursor) salt. ...
... series of experiments were carried out to study the variation of preparation parameters such as support type, temperature, ‘ion-exchange’ time and the concentration of the (precursor) salt. ...
Removal of hydrogen fluoride from gas streams
... exchange resins, and carbon based materials. Further the study can be classified as a thermogravimetric study as this was the principle analytical tool used in this investigation. This study also aims to address some of the controversy surrounding the removal of HF by calcium salts. ...
... exchange resins, and carbon based materials. Further the study can be classified as a thermogravimetric study as this was the principle analytical tool used in this investigation. This study also aims to address some of the controversy surrounding the removal of HF by calcium salts. ...
Catalytic decomposition of N2O over Rh/Zn–Al2O3 catalysts
... TEM and Rh dispersion data later. Rh/ZnO-800 shows very sharp peaks corresponding to ZnO, implying that the ZnO support is highly crystalline and has a low surface area. Again, no Rh or Rh2O3 can be seen on the XRD pattern of Rh/ZnO-800. Fig. S1 in the ESI† depicts the XRD patterns in the 2q ¼ 30–40 ...
... TEM and Rh dispersion data later. Rh/ZnO-800 shows very sharp peaks corresponding to ZnO, implying that the ZnO support is highly crystalline and has a low surface area. Again, no Rh or Rh2O3 can be seen on the XRD pattern of Rh/ZnO-800. Fig. S1 in the ESI† depicts the XRD patterns in the 2q ¼ 30–40 ...
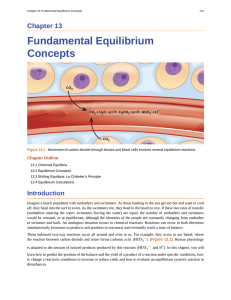
Fundamental Equilibrium Concepts
... Imagine a beach populated with sunbathers and swimmers. As those basking in the sun get too hot and want to cool off, they head into the surf to swim. As the swimmers tire, they head to the beach to rest. If these two rates of transfer (sunbathers entering the water, swimmers leaving the water) are ...
... Imagine a beach populated with sunbathers and swimmers. As those basking in the sun get too hot and want to cool off, they head into the surf to swim. As the swimmers tire, they head to the beach to rest. If these two rates of transfer (sunbathers entering the water, swimmers leaving the water) are ...
Chem Course Desc2. New
... However, the ability to understand and manipulate algebraic equations remains the most essential component for student success in chemistry. For this reason, a great deal of emphasis is placed on reinforcing these math skills. Chemistry is a subject in which concepts scaffold and build on each other ...
... However, the ability to understand and manipulate algebraic equations remains the most essential component for student success in chemistry. For this reason, a great deal of emphasis is placed on reinforcing these math skills. Chemistry is a subject in which concepts scaffold and build on each other ...
Author final version (postprint) - DTU Orbit
... agreement was observed between the results obtained using the two different systems. In the context of the PhD project, it was also important to investigate the spectral properties of major combustion species such as carbon dioxide and carbon monoxide in the infrared range at high temperatures to pr ...
... agreement was observed between the results obtained using the two different systems. In the context of the PhD project, it was also important to investigate the spectral properties of major combustion species such as carbon dioxide and carbon monoxide in the infrared range at high temperatures to pr ...
SQA Advanced Higher Chemistry Unit 2 Principles of Chemical
... 1. Calcium carbonate reacts with hydrochloric acid to produce calcium chloride solution, water and carbon dioxide. 2. One formula unit of calcium carbonate reacts with two formula units of hydrochloric acid to produce one formula unit each of calcium chloride, water and carbon dioxide. 3. One mole o ...
... 1. Calcium carbonate reacts with hydrochloric acid to produce calcium chloride solution, water and carbon dioxide. 2. One formula unit of calcium carbonate reacts with two formula units of hydrochloric acid to produce one formula unit each of calcium chloride, water and carbon dioxide. 3. One mole o ...
Separation of 2 (3), 9 (10), 16 (17), 23 (24)
... decreases with increasing temperature as assumed. Both phthalocyanines 3 and 12 show a minimum of the T1 time between 330 and 335 K without aggregation (1 mg of 3 or 12 in 0.5 mL of benzene, see Tables 1 and 2). At higher and lower temperatures, the relaxation time T1 increases as predicted. If aggr ...
... decreases with increasing temperature as assumed. Both phthalocyanines 3 and 12 show a minimum of the T1 time between 330 and 335 K without aggregation (1 mg of 3 or 12 in 0.5 mL of benzene, see Tables 1 and 2). At higher and lower temperatures, the relaxation time T1 increases as predicted. If aggr ...
ELECTROCHEMICAL STUDY OF CORROSION PROCESSES IN
... Figure 5.2: polarization curves for Type 304 SS in 0.01 M HCl (purged by hydrogen) at various temperatures .......................................................... 103 Figure 5.3: polarization curves for Ti in 0.01 M HCl (purged by hydrogen) at various temperatures………………………………………………………. 104 Figure ...
... Figure 5.2: polarization curves for Type 304 SS in 0.01 M HCl (purged by hydrogen) at various temperatures .......................................................... 103 Figure 5.3: polarization curves for Ti in 0.01 M HCl (purged by hydrogen) at various temperatures………………………………………………………. 104 Figure ...
Fate of Fuel-Bound Nitrogen and Sulfur in Biomass-Fired
... deposit formation and may also accelerate superheater corrosion. Deposit formation on heat transfer surfaces reduces the boiler efficiency and may result in unplanned shutdowns of the boiler. Sulfation of alkali chlorides was observed in the co-firing case with bark, sludge, and solid recovered fuel ...
... deposit formation and may also accelerate superheater corrosion. Deposit formation on heat transfer surfaces reduces the boiler efficiency and may result in unplanned shutdowns of the boiler. Sulfation of alkali chlorides was observed in the co-firing case with bark, sludge, and solid recovered fuel ...
GCE Chemistry SAMs 2009 onwards pdf
... water, ΔT is the temperature rise and n is the number of moles of ethanol burned and he assumes that c = 4.18 J g-1 K-1 and Mr ethanol = 46.1 (i) ...
... water, ΔT is the temperature rise and n is the number of moles of ethanol burned and he assumes that c = 4.18 J g-1 K-1 and Mr ethanol = 46.1 (i) ...
EIT Review S2012 Part 2 Dr. J. Mack CSUS Department of Chemistry
... The rate of effusion of a gas is proportional to its uRMS. over time, the mixture becomes homogeneous. ...
... The rate of effusion of a gas is proportional to its uRMS. over time, the mixture becomes homogeneous. ...
Physical Chemistry
... The predictive power of chemical thermodynamics is based on the characteristics of thermodynamic properties namely internal energy (U), enthalpy (H), entropy (S) and free energy functions (A and G) and their variations with variables like temperature, pressure, volume and amount. The changes in thes ...
... The predictive power of chemical thermodynamics is based on the characteristics of thermodynamic properties namely internal energy (U), enthalpy (H), entropy (S) and free energy functions (A and G) and their variations with variables like temperature, pressure, volume and amount. The changes in thes ...
Entropy and Free Energy
... listing. The units of entropy are J/K ∙ mol. We use joules rather than kilojoules because entropy values typically are quite small. The entropies of substances (elements and compounds) are always positive (i.e., S > 0), even for elements in their standard states. (Recall that the standard enthalpy o ...
... listing. The units of entropy are J/K ∙ mol. We use joules rather than kilojoules because entropy values typically are quite small. The entropies of substances (elements and compounds) are always positive (i.e., S > 0), even for elements in their standard states. (Recall that the standard enthalpy o ...
W. M. White Geochemistry Chapter 9: Stable Isotopes Chapter 9
... The elements interest in stable isotope geochemistry are H, Li, B, C, N, O, Si, S, and Cl. Of these, O, H, C and S are of the greatest interest. Most of these elements have several common characteristics: (1) They have low atomic mass. (2) The relative mass difference between their isotopes is large ...
... The elements interest in stable isotope geochemistry are H, Li, B, C, N, O, Si, S, and Cl. Of these, O, H, C and S are of the greatest interest. Most of these elements have several common characteristics: (1) They have low atomic mass. (2) The relative mass difference between their isotopes is large ...
Influence of Temperature on Electrical
... steam, condensate, etc.) and for certain operational conditions (start-up, normal operation). As conductivity values are dependent on temperature, the limits for standard conditions are set at 25 °C and 101.325 kPa (1 atm). With internal cooling water, the samples can usually be cooled to below 50 ° ...
... steam, condensate, etc.) and for certain operational conditions (start-up, normal operation). As conductivity values are dependent on temperature, the limits for standard conditions are set at 25 °C and 101.325 kPa (1 atm). With internal cooling water, the samples can usually be cooled to below 50 ° ...
Document
... reaction is started with [H2 ]0 = 0.76 M, [N2]0 = 0.60 M and [NH3]0= 0.48 M. Which of the following is correct as the reaction comes to equilibrium? A) The concentration of N2will increase B) The concentration of H2will decrease C) The concentration of NH3will decrease D) The concentration of both N ...
... reaction is started with [H2 ]0 = 0.76 M, [N2]0 = 0.60 M and [NH3]0= 0.48 M. Which of the following is correct as the reaction comes to equilibrium? A) The concentration of N2will increase B) The concentration of H2will decrease C) The concentration of NH3will decrease D) The concentration of both N ...
Experiment 7: Determination of the concentration of a solution of an
... In order to assist you in developing good and safe laboratory techniques, a set of Laboratory Rules and Regulations is attached. You are required to read these and to acknowledge that you have read and understood them. Additionally, in the laboratory manuals/books and/ ...
... In order to assist you in developing good and safe laboratory techniques, a set of Laboratory Rules and Regulations is attached. You are required to read these and to acknowledge that you have read and understood them. Additionally, in the laboratory manuals/books and/ ...