Compounds Power point
... That atoms are made up of Protons, Neutrons, and Electrons The identity of an atom is determined by the number of protons in the nucleus ...
... That atoms are made up of Protons, Neutrons, and Electrons The identity of an atom is determined by the number of protons in the nucleus ...
Section 11.1 Assessment How many mole ratios can be written for
... • A mole ratio is a ratio between the numbers of moles of any two substances in a balanced equation. • The number of mole ratios that can be written for any equation is (n)(n – 1) where n is the number of species in the chemical reaction. ...
... • A mole ratio is a ratio between the numbers of moles of any two substances in a balanced equation. • The number of mole ratios that can be written for any equation is (n)(n – 1) where n is the number of species in the chemical reaction. ...
Document
... 34. Which of the following correctly represents an ion pair and the ionic compound the ions form? ...
... 34. Which of the following correctly represents an ion pair and the ionic compound the ions form? ...
Title - Iowa State University
... 6) Which of the following are insoluble? I.Ag2SO4 II. Ni3PO4 III. NaI a) I only b) II only c) III only d) I and II e)I, II and II 7) Aqueous sodium carbonate reacts with barium chloride Molecular equation: Ionic equation: Spectator ions: Net ionic equation: 1060 Hixson-Lied Student Success Center ...
... 6) Which of the following are insoluble? I.Ag2SO4 II. Ni3PO4 III. NaI a) I only b) II only c) III only d) I and II e)I, II and II 7) Aqueous sodium carbonate reacts with barium chloride Molecular equation: Ionic equation: Spectator ions: Net ionic equation: 1060 Hixson-Lied Student Success Center ...
Reaction Stoichiometry
... We cannot simply add the total moles of all the reactants to decide which reactant mixture makes the most product. We must always think about how much product can be formed by using what we are given, and the ratio in the balanced equation. ...
... We cannot simply add the total moles of all the reactants to decide which reactant mixture makes the most product. We must always think about how much product can be formed by using what we are given, and the ratio in the balanced equation. ...
Student Exploration Sheet: Growing Plants
... made of more than one kind of atom, such as water (H 2O) or table salt (NaCl). Question: How are chemical reactions classified? Directions – Reselect each equation below on the gizmo, study what is happening with the compounds and elements and write a definition to explain what is occurring within t ...
... made of more than one kind of atom, such as water (H 2O) or table salt (NaCl). Question: How are chemical reactions classified? Directions – Reselect each equation below on the gizmo, study what is happening with the compounds and elements and write a definition to explain what is occurring within t ...
Balancing a Chemical Equation
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
Balancing a Chemical Equation
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
Balancing a Chemical Equation
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
Conservation of Energy in chemical reactions, Hess`s Law
... high, or because the reactants are difficult to obtain or handle. Hess’s Law (named after the scientist who proposed it) helps us to calculate H for a reaction by using data from other reactions that are related to it. Hess’s Law states: If a chemical equation can be written as the ‘sum’ of several ...
... high, or because the reactants are difficult to obtain or handle. Hess’s Law (named after the scientist who proposed it) helps us to calculate H for a reaction by using data from other reactions that are related to it. Hess’s Law states: If a chemical equation can be written as the ‘sum’ of several ...
balancing eqns teacher
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
... Chemical Equations A chemical equation is written as an expression similar to a mathematic equation that can be compared to a recipe that a chemist follows in order to produce desired results. ...
Chapter 8
... Balanced equations are equations for a chemical reaction in which the number of atoms for each element in the reaction and the total charge are the same for both the reactants and the products . In other words…the mass and the charge are balanced on both sides of the reaction. ...
... Balanced equations are equations for a chemical reaction in which the number of atoms for each element in the reaction and the total charge are the same for both the reactants and the products . In other words…the mass and the charge are balanced on both sides of the reaction. ...
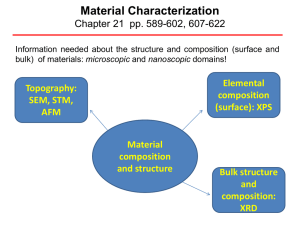
Material Characterization
... Constant Height Mode = By using a feedback loop the tip is vertically adjusted in such a way that the current always stays constant. As the current is proportional to the local density of states, the tip follows a contour of a constant density of states during scanning. A kind of a topographic image ...
... Constant Height Mode = By using a feedback loop the tip is vertically adjusted in such a way that the current always stays constant. As the current is proportional to the local density of states, the tip follows a contour of a constant density of states during scanning. A kind of a topographic image ...
Lecture 9
... include dipole-dipole interactions, dispersion forces, attraction between oppositely charged ions. Cohesive forces are largely temperature independent. e.g. magnets and gravity function the same way at different temperature. ...
... include dipole-dipole interactions, dispersion forces, attraction between oppositely charged ions. Cohesive forces are largely temperature independent. e.g. magnets and gravity function the same way at different temperature. ...
Key To T2 Review For Final Study Guide File - District 196 e
... 8. What is a limiting reactant? Why is this reactant so important? The limiting reactant is the reactant that runs out first in a chemical reaction, therefore determining the amount of product produced. 9. What is an excess reactant? The reactant that there is more than enough of to complete the lim ...
... 8. What is a limiting reactant? Why is this reactant so important? The limiting reactant is the reactant that runs out first in a chemical reaction, therefore determining the amount of product produced. 9. What is an excess reactant? The reactant that there is more than enough of to complete the lim ...
Chapter 10
... Predicting Products of Synthesis Reactions For Synthesis Reactions For metals that only form one cation, determine the charge on the ion of each element (metallic and nonmetallic) and form a compound from the two ions. If one of the elements forms more than one cation or 2 nonmetals are combined ...
... Predicting Products of Synthesis Reactions For Synthesis Reactions For metals that only form one cation, determine the charge on the ion of each element (metallic and nonmetallic) and form a compound from the two ions. If one of the elements forms more than one cation or 2 nonmetals are combined ...
Chemical Reactions Chemical Arithmetic
... reaction in which the oxidation numbers of elements change because of a loss or gain of electrons • Oxidation Number- A number that indicates the charge that an atom in a molecule or polyatomic ion would have if all bonds were ionic. – Fictitious- No actual charge of this magnitude actually exists w ...
... reaction in which the oxidation numbers of elements change because of a loss or gain of electrons • Oxidation Number- A number that indicates the charge that an atom in a molecule or polyatomic ion would have if all bonds were ionic. – Fictitious- No actual charge of this magnitude actually exists w ...
Final Exam Review – Free Response Section Name: 1. A sample of
... 1. A sample of chalk (Calcium carbonate) weighs 1.75 g. Calculate the mass of calcium in the sample of chalk in both grams and ounces (there are 28.35 g in 1 ounce). Calculate how many oxygen atoms are in the sample of chalk. ...
... 1. A sample of chalk (Calcium carbonate) weighs 1.75 g. Calculate the mass of calcium in the sample of chalk in both grams and ounces (there are 28.35 g in 1 ounce). Calculate how many oxygen atoms are in the sample of chalk. ...
7th Chemistry Unit Test Study Guide Test Date: Friday, Nov. 16
... reactions. They read that if you heat a small amount of calcium carbonate (CaCO3), it will produce calcium oxide (CaO) and carbon ...
... reactions. They read that if you heat a small amount of calcium carbonate (CaCO3), it will produce calcium oxide (CaO) and carbon ...
Oxidation-Reduction (Redox) Reactions
... b. Balance O by adding H2O to the appropriate side. c. Balance H by adding the appropriate number of H+ ions to the side that needs H. Add H2O to the other side if needed. d. Balance net charge by adding electrons. Step 3: Multiply the half-reactions by integers that will allow for cancellation of e ...
... b. Balance O by adding H2O to the appropriate side. c. Balance H by adding the appropriate number of H+ ions to the side that needs H. Add H2O to the other side if needed. d. Balance net charge by adding electrons. Step 3: Multiply the half-reactions by integers that will allow for cancellation of e ...
Science24-UnitA-Section3.4
... 1. Previously, you studied the neutralization reaction that occurs when you combine vinegar and baking soda. If you were able to measure the mass of the reactants and then the mass of products in this reaction, how do ...
... 1. Previously, you studied the neutralization reaction that occurs when you combine vinegar and baking soda. If you were able to measure the mass of the reactants and then the mass of products in this reaction, how do ...
5 · Chemical Reactions
... You will be given a periodic table and a solubility chart. No other resources are allowed. You have fifty (50) minutes to complete this test, unless other arrangements have been made. Please transfer your answers for questions in Sections 1 and 2 onto the Answer Document. Work for these questions wi ...
... You will be given a periodic table and a solubility chart. No other resources are allowed. You have fifty (50) minutes to complete this test, unless other arrangements have been made. Please transfer your answers for questions in Sections 1 and 2 onto the Answer Document. Work for these questions wi ...
Ionic Compounds 1. What is the formula for aluminum phosphate
... 3. A gas sample is held at constant pressure. The gas occupies 3.62 L of volume when the temperature is 21.6oC. Determine the temperature at which the volume of the gas is 3.46 L. 4. A balloon has a volume of 2.46 liters at 24.0oC. The balloon is heated to 48.0oC. Calculate the new volume of the bal ...
... 3. A gas sample is held at constant pressure. The gas occupies 3.62 L of volume when the temperature is 21.6oC. Determine the temperature at which the volume of the gas is 3.46 L. 4. A balloon has a volume of 2.46 liters at 24.0oC. The balloon is heated to 48.0oC. Calculate the new volume of the bal ...
document
... Short-hand way of describing a reaction. Provides information about the reaction. Formulas of reactants and products. States of reactants and products. Relative numbers of reactant and product molecules that are required. Can be used to determine masses of reactants used and products tha ...
... Short-hand way of describing a reaction. Provides information about the reaction. Formulas of reactants and products. States of reactants and products. Relative numbers of reactant and product molecules that are required. Can be used to determine masses of reactants used and products tha ...
Introduction to Chemical Equations
... Heat/light given off or heat absorbed Production of a gas Formation of a new solid (precipitate) A new color appears ...
... Heat/light given off or heat absorbed Production of a gas Formation of a new solid (precipitate) A new color appears ...
Double layer forces

Double layer forces occur between charged objects across liquids, typically water. This force acts over distances that are comparable to the Debye length, which is on the order of one to a few tenths of nanometers. The strength of these forces increases with the magnitude of the surface charge density (or the electrical surface potential). For two similarly charged objects, this force is repulsive and decays exponentially at larger distances, see figure. For unequally charged objects and eventually at shorted distances, these forces may also be attractive. The theory due to Derjaguin, Landau, Verwey, and Overbeek (DLVO) combines such double layer forces together with Van der Waals forces in order to estimate the actual interaction potential between colloidal particles.An electrical double layer develops near charged surfaces (or another charged objects) in aqueous solutions. Within this double layer, the first layer corresponds to the charged surface. These charges may originate from tightly adsorbed ions, dissociated surface groups, or substituted ions within the crystal lattice. The second layer corresponds to the diffuse layer, which contains the neutralizing charge consisting of accumulated counterions and depleted coions. The resulting potential profile between these two objects leads to differences in the ionic concentrations within the gap between these objects with respect to the bulk solution. These differences generate an osmotic pressure, which generates a force between these objects.These forces are easily experienced when hands are washed with soap. Adsorbing soap molecules make the skin negatively charged, and the slippery feeling is caused by the strongly repulsive double layer forces. These forces are further relevant in many colloidal or biological systems, and may be responsible for their stability, formation of colloidal crystals, or their rheological properties.