transition metals
... 11.) Does the Qc for the formation of 1 mole of NO from its elements differ from the Qc of the decomposition of 1 mole of NO into its elements? Explain and give the relationship between the two Qc values. 12.) Balance the reaction and write the Qc a. ________NaHCO3 (s) ________Na2CO3 (s) + _______ ...
... 11.) Does the Qc for the formation of 1 mole of NO from its elements differ from the Qc of the decomposition of 1 mole of NO into its elements? Explain and give the relationship between the two Qc values. 12.) Balance the reaction and write the Qc a. ________NaHCO3 (s) ________Na2CO3 (s) + _______ ...
I PUC Chemistry Mock Paper
... 14. How is plaster of paris prepared from gypsum? Give equation 15. Write any two differences between diamond and graphite. 16. Explain Wurtz reaction with an example. 17. How do you convert benzene to hexachlorobenzene ? Give equation 18. Mention any two gases which are responsible for greenhouse e ...
... 14. How is plaster of paris prepared from gypsum? Give equation 15. Write any two differences between diamond and graphite. 16. Explain Wurtz reaction with an example. 17. How do you convert benzene to hexachlorobenzene ? Give equation 18. Mention any two gases which are responsible for greenhouse e ...
Writing And Balancing Equations
... molecule of calcium hydroxide plus one molecule of sulfuric acid yields one molecule of calcium sulfate plus two molecules of water. ...
... molecule of calcium hydroxide plus one molecule of sulfuric acid yields one molecule of calcium sulfate plus two molecules of water. ...
Exam 2 Review - Iowa State University
... 4. The sum of oxidation numbers of all atoms in a compound equal the charge of that compound 1. Identify the oxidation number of each individual atom in the following equation. a. Which atom is being oxidized? Reduced? Zn (s) + H2SO4 (aq) ZnSO4 (aq) + H2 (g) ...
... 4. The sum of oxidation numbers of all atoms in a compound equal the charge of that compound 1. Identify the oxidation number of each individual atom in the following equation. a. Which atom is being oxidized? Reduced? Zn (s) + H2SO4 (aq) ZnSO4 (aq) + H2 (g) ...
Exam practice answers 5
... Cl– and Br– is the same but the ionic radius of Cl– is smaller than that of Br– and therefore the attraction between the Mg2+ ion and the Cl– will be greater. or Charge density of Cl– is greater than the charge density of Br–, hence the lattice enthalpy MgCl2 is more negative than MgBr2 (d) Ther ...
... Cl– and Br– is the same but the ionic radius of Cl– is smaller than that of Br– and therefore the attraction between the Mg2+ ion and the Cl– will be greater. or Charge density of Cl– is greater than the charge density of Br–, hence the lattice enthalpy MgCl2 is more negative than MgBr2 (d) Ther ...
File
... Law of conservation of mass- mass is cannot be destroyed or created during chemical reaction. Law of definite proportions- When two or more elements combine to form a compound, their masses in that compound are in a fixed and definite ratio, (e.g. NaCl is always 60.66% of Na and 39.34% of Cl by mass ...
... Law of conservation of mass- mass is cannot be destroyed or created during chemical reaction. Law of definite proportions- When two or more elements combine to form a compound, their masses in that compound are in a fixed and definite ratio, (e.g. NaCl is always 60.66% of Na and 39.34% of Cl by mass ...
Unit 6 Moles and Stoichiometry Short Answer Review
... Base your answers to questions 1 through 3 on the information below. Rust on an automobile door contains Fe 2O3(s). The balanced equation representing one of the reactions between iron in the door of the automobile and oxygen in the atmosphere is given below. 4Fe(s) + 3O 2(g) 2Fe 2O3(s) 1. Write the ...
... Base your answers to questions 1 through 3 on the information below. Rust on an automobile door contains Fe 2O3(s). The balanced equation representing one of the reactions between iron in the door of the automobile and oxygen in the atmosphere is given below. 4Fe(s) + 3O 2(g) 2Fe 2O3(s) 1. Write the ...
AP Chemistry Ch. 3 Sections 3.7-3.8 Notes Chemical Equations
... In a chemical reaction, atoms are neither created nor destroyed. All atoms present in the reactants must be accounted for among the products. In other words, there must be the same number of each type of atom on the product side and on the reactant side of the arrow. Known as balancing a chemical eq ...
... In a chemical reaction, atoms are neither created nor destroyed. All atoms present in the reactants must be accounted for among the products. In other words, there must be the same number of each type of atom on the product side and on the reactant side of the arrow. Known as balancing a chemical eq ...
Chemical Equations
... • A + B ---> AB Reaction Types: Combustion •Combustion, at its most general, can mean the reaction of oxygen gas (O2) with anything. •However, we will understand combustion to mean the reaction of oxygen with a compound containing carbon and hydrogen. A common synonym for combustion is burn. •Writte ...
... • A + B ---> AB Reaction Types: Combustion •Combustion, at its most general, can mean the reaction of oxygen gas (O2) with anything. •However, we will understand combustion to mean the reaction of oxygen with a compound containing carbon and hydrogen. A common synonym for combustion is burn. •Writte ...
Physical Science Semester 2 Final Exam 2013 –STUDY GUIDE
... 17. Increasing the speed of an object ____ its potential energy. 18. The SI (metric) unit for energy is the ____. 19. You can calculate kinetic energy by using the equation ____. 20. You can calculate gravitational potential energy by using the equation ____. 21. According to the law of conservatio ...
... 17. Increasing the speed of an object ____ its potential energy. 18. The SI (metric) unit for energy is the ____. 19. You can calculate kinetic energy by using the equation ____. 20. You can calculate gravitational potential energy by using the equation ____. 21. According to the law of conservatio ...
Task - Science - Grade 6 - Chemical Reactions
... a single lump. A sugar cube has a specific surface area, but, when it is split or crushed, the surface area of all of the pieces combined is significantly higher than the original sugar cube. The sugar plant in Savannah, Georgia, had finite particles of sugar coating the floor, the machinery, and fl ...
... a single lump. A sugar cube has a specific surface area, but, when it is split or crushed, the surface area of all of the pieces combined is significantly higher than the original sugar cube. The sugar plant in Savannah, Georgia, had finite particles of sugar coating the floor, the machinery, and fl ...
Balancing Equations
... Chemical reactions occur when bonds (between the electrons of atoms) are formed or broken Chemical reactions involve changes in the chemical composition of matter the making of new materials with new properties energy changes: Bond breaking absorbs Energy (endothermic process) Bond making ...
... Chemical reactions occur when bonds (between the electrons of atoms) are formed or broken Chemical reactions involve changes in the chemical composition of matter the making of new materials with new properties energy changes: Bond breaking absorbs Energy (endothermic process) Bond making ...
use-2012_review_sheettest_form_c_reactions
... d) 3 Mg + 2 FeBr3 Fe2Mg3 + 3 Br2 e) N + H NH3 f) NaBr + CaF2 --> NaCa + FBr g) ...
... d) 3 Mg + 2 FeBr3 Fe2Mg3 + 3 Br2 e) N + H NH3 f) NaBr + CaF2 --> NaCa + FBr g) ...
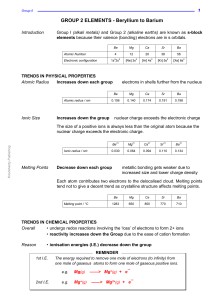
GROUP 2 ELEMENTS - Beryllium to Barium
... basic strength also increases down group this is because the solubility increases the metal ions get larger so charge density decreases there is a lower attraction between the OH¯ ions and larger dipositive ions the ions will split away from each other more easily there will be a greater concentrati ...
... basic strength also increases down group this is because the solubility increases the metal ions get larger so charge density decreases there is a lower attraction between the OH¯ ions and larger dipositive ions the ions will split away from each other more easily there will be a greater concentrati ...
Critical Thinking Questions 4
... How many C, H and O atoms are on the left of the arrow and how many C, H and O atoms are on the right of the arrow? ...
... How many C, H and O atoms are on the left of the arrow and how many C, H and O atoms are on the right of the arrow? ...
Chemistry Review2
... Naming and Ions: Two elements always end in “ide”, three or more many times end in “ite” or “ate” Ions are cations with a positive charge, mostly metals like Na+, Mg +2, Al +3, Fe +2 and Fe+3 and NH4+ Or anions with a negative charge, mostly non-metals. When writing a compound formula with cations a ...
... Naming and Ions: Two elements always end in “ide”, three or more many times end in “ite” or “ate” Ions are cations with a positive charge, mostly metals like Na+, Mg +2, Al +3, Fe +2 and Fe+3 and NH4+ Or anions with a negative charge, mostly non-metals. When writing a compound formula with cations a ...
Notes for powerpoint and worksheets PDF
... Polyatomic ions are ions made of many atoms and usually have parenthesis around them How are compound named? (Type I & II) all metals (cations) use the _______________________________________________________ all non metal ions (anions) use the ___________________________________________________ ...
... Polyatomic ions are ions made of many atoms and usually have parenthesis around them How are compound named? (Type I & II) all metals (cations) use the _______________________________________________________ all non metal ions (anions) use the ___________________________________________________ ...
Review
... Expression for the molar Gibbs free energy, the chemical potential, of a gas Calculation of the Equilibrium Constant from Gorxn or the reverse of this. Calculating the Temp dependence of the equilibrium constant Use of LeChatliers Principle Relationship between Kp, Kc write expression for K in acti ...
... Expression for the molar Gibbs free energy, the chemical potential, of a gas Calculation of the Equilibrium Constant from Gorxn or the reverse of this. Calculating the Temp dependence of the equilibrium constant Use of LeChatliers Principle Relationship between Kp, Kc write expression for K in acti ...
Unit 5 Study Guide
... Unit 5 Study Guide: Chemical Reactions 1. What are the 7 diatomic molecules? ...
... Unit 5 Study Guide: Chemical Reactions 1. What are the 7 diatomic molecules? ...
Document
... solution, but it cannot replace the ions of metal Z from solution. The order these metals should have in the activity series (from top to bottom) is A. x, y, z B. z, x, y C. y, x, z D. z, y, x ...
... solution, but it cannot replace the ions of metal Z from solution. The order these metals should have in the activity series (from top to bottom) is A. x, y, z B. z, x, y C. y, x, z D. z, y, x ...
Публикация доступна для обсуждения в рамках
... layer, capable to be formed in the conditions of treatment of semiconductor surface without and with electric field exposure at the different values of electrode potentials and рН. It is shown, that equilibrium with respect to the electronegative component (A) is shifted to the anode direction, ther ...
... layer, capable to be formed in the conditions of treatment of semiconductor surface without and with electric field exposure at the different values of electrode potentials and рН. It is shown, that equilibrium with respect to the electronegative component (A) is shifted to the anode direction, ther ...
Balancing Chemical Equations
... • Describe a chemical reaction by using a word equation and a formula equation. • Relate the conservation of mass to the rearrangement of atoms in a chemical reaction • Write and interpret a balanced chemical equation for a reaction, and relate conservation of mass to the balanced equation ...
... • Describe a chemical reaction by using a word equation and a formula equation. • Relate the conservation of mass to the rearrangement of atoms in a chemical reaction • Write and interpret a balanced chemical equation for a reaction, and relate conservation of mass to the balanced equation ...
Periodic Table, Bonding, Reactions, and Moles
... 8. Explain, in terms of valence electrons, why the bonding in magnesium oxide, MgO, is similar to the bonding in barium chloride, BaCl2. 9. Identify the type of bonding between the atoms in an oxygen molecule. ...
... 8. Explain, in terms of valence electrons, why the bonding in magnesium oxide, MgO, is similar to the bonding in barium chloride, BaCl2. 9. Identify the type of bonding between the atoms in an oxygen molecule. ...
Balancing ANY chemical Equation
... • Electrolytes: Substances that form ions when dissolved in solution. Electrolytes can be weak or strong. • Strong Electrolytes: Substances that completely separate into their component ions when dissolved. (All soluble ionic compounds and strong acids are strong electrolytes.) • Weak Electrolytes: ...
... • Electrolytes: Substances that form ions when dissolved in solution. Electrolytes can be weak or strong. • Strong Electrolytes: Substances that completely separate into their component ions when dissolved. (All soluble ionic compounds and strong acids are strong electrolytes.) • Weak Electrolytes: ...
Double layer forces

Double layer forces occur between charged objects across liquids, typically water. This force acts over distances that are comparable to the Debye length, which is on the order of one to a few tenths of nanometers. The strength of these forces increases with the magnitude of the surface charge density (or the electrical surface potential). For two similarly charged objects, this force is repulsive and decays exponentially at larger distances, see figure. For unequally charged objects and eventually at shorted distances, these forces may also be attractive. The theory due to Derjaguin, Landau, Verwey, and Overbeek (DLVO) combines such double layer forces together with Van der Waals forces in order to estimate the actual interaction potential between colloidal particles.An electrical double layer develops near charged surfaces (or another charged objects) in aqueous solutions. Within this double layer, the first layer corresponds to the charged surface. These charges may originate from tightly adsorbed ions, dissociated surface groups, or substituted ions within the crystal lattice. The second layer corresponds to the diffuse layer, which contains the neutralizing charge consisting of accumulated counterions and depleted coions. The resulting potential profile between these two objects leads to differences in the ionic concentrations within the gap between these objects with respect to the bulk solution. These differences generate an osmotic pressure, which generates a force between these objects.These forces are easily experienced when hands are washed with soap. Adsorbing soap molecules make the skin negatively charged, and the slippery feeling is caused by the strongly repulsive double layer forces. These forces are further relevant in many colloidal or biological systems, and may be responsible for their stability, formation of colloidal crystals, or their rheological properties.