Atomic Structure and Periodic Table Quick Notes
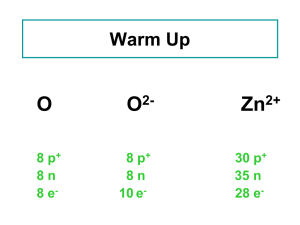
... If an atom loses an electron, it becomes a Positive Ion If an atom gains an electron, it becomes a Negative Ion Ex: when Na reacts with Cl to form NaCl, the Na atom loses an electron, making it a Positive Ion: Na+ The Cl, which gains the electron, now has more negative particles than positive, b ...
... If an atom loses an electron, it becomes a Positive Ion If an atom gains an electron, it becomes a Negative Ion Ex: when Na reacts with Cl to form NaCl, the Na atom loses an electron, making it a Positive Ion: Na+ The Cl, which gains the electron, now has more negative particles than positive, b ...
Ch. 5 Outline Notes
... A. Early Models of the Atom 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________________ Atomic Theory 1. All matter is made of ________________ 2. At ...
... A. Early Models of the Atom 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________________ Atomic Theory 1. All matter is made of ________________ 2. At ...
Chapter 10 Test A
... ____ 19. When an unstable isotope undergoes alpha decay, it is gives off: a. an electron. b. two protons and two neutrons. c. high energy electromagnetic radiation. d. a hydrogen atom. ____ 20. The half-life is best described as the time it takes for: a. an atom to rotate halfway around. b. 50 perce ...
... ____ 19. When an unstable isotope undergoes alpha decay, it is gives off: a. an electron. b. two protons and two neutrons. c. high energy electromagnetic radiation. d. a hydrogen atom. ____ 20. The half-life is best described as the time it takes for: a. an atom to rotate halfway around. b. 50 perce ...
Structure of the Atom JJ Thomson- discovered the electron in late
... as protons are found to be at the center of this nucleus. James Chadwick- discovers the NEUTRON in 1932. The neutron is located in the nucleus and has NO CHARGE. The following table summarizes the subatomic particles listed in order of discovery: ...
... as protons are found to be at the center of this nucleus. James Chadwick- discovers the NEUTRON in 1932. The neutron is located in the nucleus and has NO CHARGE. The following table summarizes the subatomic particles listed in order of discovery: ...
Chapter 3: Atomic Structure
... Application of Nuclear Chemistry • Use of half life + Radioactive Dating • Nuclear Bombardment – Reactions • Create radioactive isotopes used in medicine • Power Generation • Fission – Limerick Generating Plant • Fusion – “research” ...
... Application of Nuclear Chemistry • Use of half life + Radioactive Dating • Nuclear Bombardment – Reactions • Create radioactive isotopes used in medicine • Power Generation • Fission – Limerick Generating Plant • Fusion – “research” ...
Atoms
... National Science Education Standards NSES B1a. Matter is made of minute particles called atoms, and atoms are composed of even smaller components. These components have measurable properties, such as mass and electrical charge. Each atom has a positively charged nucleus surrounded by negatively char ...
... National Science Education Standards NSES B1a. Matter is made of minute particles called atoms, and atoms are composed of even smaller components. These components have measurable properties, such as mass and electrical charge. Each atom has a positively charged nucleus surrounded by negatively char ...
Topic 2.1- The Nuclear Atom
... Isotopes • same element but differ in their number of neutrons • the atomic mass on periodic table is the WEIGHTED AVERAGE MASS of “all” the isotopes of that element – this is based on an isotope’s natural abundance • the percentage of each isotope of an element that occurs in nature ...
... Isotopes • same element but differ in their number of neutrons • the atomic mass on periodic table is the WEIGHTED AVERAGE MASS of “all” the isotopes of that element – this is based on an isotope’s natural abundance • the percentage of each isotope of an element that occurs in nature ...
Chapter 2 - profpaz.com
... (different mass numbers) and are called isotopes. Most elements have several isotopes, which are indicated by its chemical symbol, followed by a dash and the mass number of isotope. For example, the 3 isotopes of neon are shown below: ...
... (different mass numbers) and are called isotopes. Most elements have several isotopes, which are indicated by its chemical symbol, followed by a dash and the mass number of isotope. For example, the 3 isotopes of neon are shown below: ...
ISOSTOPE NOTES - Mr. Collier`s 9th Grade Physical Science
... electrons • The number of neutrons can vary from one atom of an element to another. –These variations are called ISOTOPES. ...
... electrons • The number of neutrons can vary from one atom of an element to another. –These variations are called ISOTOPES. ...
Structure-Prop of Matter session
... Carbon-14 and Carbon-13 atoms’ are not as stable as carbon-12 and easily break down. If an isotope has too many or too few neutrons compared to the number of protons, it is unstable and will undergo radioactive decay. These radioactive isotopes become different elements in an effort to become more s ...
... Carbon-14 and Carbon-13 atoms’ are not as stable as carbon-12 and easily break down. If an isotope has too many or too few neutrons compared to the number of protons, it is unstable and will undergo radioactive decay. These radioactive isotopes become different elements in an effort to become more s ...
atomic number
... Dalton’s Atomic Theory – Atoms of different elements can be distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed bu ...
... Dalton’s Atomic Theory – Atoms of different elements can be distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed bu ...
No Slide Title
... Dalton’s Atomic Theory – Atoms of different elements can be distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed bu ...
... Dalton’s Atomic Theory – Atoms of different elements can be distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed bu ...
Unit 3 - Princeton High School
... very small, indivisible particles, each of which was called a(n) _____________. The theory that such particles existed was supported much later, by _____________ who proposed, in his law of _______________ _____ __________, that matter could not be created or destroyed. Then ___________ proposed, in ...
... very small, indivisible particles, each of which was called a(n) _____________. The theory that such particles existed was supported much later, by _____________ who proposed, in his law of _______________ _____ __________, that matter could not be created or destroyed. Then ___________ proposed, in ...
Law of Physics
... • .009 is the mass that is lost. It goes to energy and is called the nuclear binding energy. With this absence, the nucleolus will not fly apart. ...
... • .009 is the mass that is lost. It goes to energy and is called the nuclear binding energy. With this absence, the nucleolus will not fly apart. ...
Isotopes-Chemistry
... Same Element Different AtomIsotopes All atoms of a particular element are not exactly alike. Some elements have atoms with different masses (isotopes) ...
... Same Element Different AtomIsotopes All atoms of a particular element are not exactly alike. Some elements have atoms with different masses (isotopes) ...
Isotopes
... • To name isotopes use the name of the element followed by the isotope’s mass number. ...
... • To name isotopes use the name of the element followed by the isotope’s mass number. ...
Periodic Scavenger Hunt - bates
... 8. The atomic mass of an element is a combination of the number of protons and neutrons. Because the same element does not always have the same number of neutrons, the atomic mass is an average mass of the element as it occurs in nature. What is the atomic mass of fluorine? ...
... 8. The atomic mass of an element is a combination of the number of protons and neutrons. Because the same element does not always have the same number of neutrons, the atomic mass is an average mass of the element as it occurs in nature. What is the atomic mass of fluorine? ...
2 C Atomic Number Mass Number Atomic Mass and Isotopes
... Atoms have no overall electrical charge so, an atom must have as many electrons as there are ...
... Atoms have no overall electrical charge so, an atom must have as many electrons as there are ...
Notes on Atomic Structure Structure of Atoms Atoms are composed
... The periodic table is a list of the elements that make up matter. It is organized by increasing atomic number. The Atomic Number shows the number of protons in the nucleus of an atom. It identifies the type of atom/element. The atomic number also equals the number of electrons whenever the atom is n ...
... The periodic table is a list of the elements that make up matter. It is organized by increasing atomic number. The Atomic Number shows the number of protons in the nucleus of an atom. It identifies the type of atom/element. The atomic number also equals the number of electrons whenever the atom is n ...
What is an isotope?
... neutrons are called isotopes of each other. All atoms in existence are isotopes! Some isotopes are just more common than others. ...
... neutrons are called isotopes of each other. All atoms in existence are isotopes! Some isotopes are just more common than others. ...
elements and isotopes - vocabulary
... A species of atom; each atom of a particular isotope has a specific number of protons and a specific number of neutrons in the nucleus which are the same for all atoms of the isotope, but are not necessarily equal to each other. atomic number The number of protons in the nucleus of one atom; the ato ...
... A species of atom; each atom of a particular isotope has a specific number of protons and a specific number of neutrons in the nucleus which are the same for all atoms of the isotope, but are not necessarily equal to each other. atomic number The number of protons in the nucleus of one atom; the ato ...
Atom, Ion, Isotope Notes from 10/5 and 10/6
... a good estimation for finding the most common stable isotope of an atom. HOWEVER, it is not a perfect method. Look at Ag for example. It’s atomic mass is 107.87 amu, which would round to 108 amu. This is actually NOT a stable isotope of Ag (only 107 amu and 109 amu are). If you really wanted to know ...
... a good estimation for finding the most common stable isotope of an atom. HOWEVER, it is not a perfect method. Look at Ag for example. It’s atomic mass is 107.87 amu, which would round to 108 amu. This is actually NOT a stable isotope of Ag (only 107 amu and 109 amu are). If you really wanted to know ...
Topic 2.1 The Nuclear Atom
... • this is NOT IB material until indicated • it is very interesting from a geeky-science stand point • it will help you understand and appreciate the structure of the atom • you are not responsible for knowing the information from all thescientists ...
... • this is NOT IB material until indicated • it is very interesting from a geeky-science stand point • it will help you understand and appreciate the structure of the atom • you are not responsible for knowing the information from all thescientists ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.