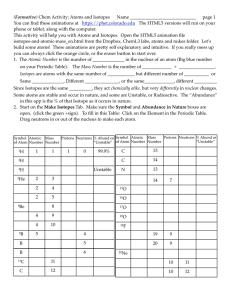
4.1 Experiencing Atoms at Tiburon 4.1 Experiencing Atoms
... • Elements on the left side are metals. They tend to lose electrons in their chemical changes. • Elements on the upper right side are nonmetals. They tend to gain electrons in their chemical changes. • Elements between the two are called metalloids. Atomic number: • The characteristic that defin ...
... • Elements on the left side are metals. They tend to lose electrons in their chemical changes. • Elements on the upper right side are nonmetals. They tend to gain electrons in their chemical changes. • Elements between the two are called metalloids. Atomic number: • The characteristic that defin ...
Chapter 4 Atoms and Elements
... • Elements on the left side are metals. They tend to lose electrons in their chemical changes. • Elements on the upper right side are nonmetals. They tend to gain electrons in their chemical changes. • Elements between the two are called metalloids. Atomic number: • The characteristic that defines a ...
... • Elements on the left side are metals. They tend to lose electrons in their chemical changes. • Elements on the upper right side are nonmetals. They tend to gain electrons in their chemical changes. • Elements between the two are called metalloids. Atomic number: • The characteristic that defines a ...
Unit 2 Complete 2016 2017
... 14)______________________ A positively charged particle. 15)______________________ Chemical compounds have the same elements in exactly the same ratios. 16)______________________ This English scientist is credited with discovering the neutron. 17)______________________ It represents the number of pr ...
... 14)______________________ A positively charged particle. 15)______________________ Chemical compounds have the same elements in exactly the same ratios. 16)______________________ This English scientist is credited with discovering the neutron. 17)______________________ It represents the number of pr ...
Chapter #4 Section Assessment #1 - 33
... i) All matter is composed of tiny, indivisible particles called “atoms”. Is everything he said here still believed to be true? ii) Atoms of the same element are identical. The atoms of any one element are different from those of another element. Is everything he said here still believed to be true? ...
... i) All matter is composed of tiny, indivisible particles called “atoms”. Is everything he said here still believed to be true? ii) Atoms of the same element are identical. The atoms of any one element are different from those of another element. Is everything he said here still believed to be true? ...
1.1 - cloudfront.net
... If we were to place a sample of carbon into a mass spectrometer and analyze its mass, we would find that some of the carbon atoms have a relative mass of 12, while other atoms have a relative mass of 13, and still others have a relative mass of 14. The mass spectrometer measures the percent abundanc ...
... If we were to place a sample of carbon into a mass spectrometer and analyze its mass, we would find that some of the carbon atoms have a relative mass of 12, while other atoms have a relative mass of 13, and still others have a relative mass of 14. The mass spectrometer measures the percent abundanc ...
Topic 2 Atomic Structure File
... 6. If the mass number of the atom of a given element is known, the number of neutrons in its nucleus can be calculated by subtracting the _______________________ from the _______________________. For example, if an atom of the element sodium, atomic number 11, has a mass of 23, the atom has ________ ...
... 6. If the mass number of the atom of a given element is known, the number of neutrons in its nucleus can be calculated by subtracting the _______________________ from the _______________________. For example, if an atom of the element sodium, atomic number 11, has a mass of 23, the atom has ________ ...
Atoms and nukes packet 2016
... Indiana Jones and Nicholas Cage are searching for buried treasures again. They think they may find something in your backyard! They’re searching for 30 million year old fish fossils, 2000 year old cups and billion year old rocks! Radioactive dating will help us estimate the age of some objects. But ...
... Indiana Jones and Nicholas Cage are searching for buried treasures again. They think they may find something in your backyard! They’re searching for 30 million year old fish fossils, 2000 year old cups and billion year old rocks! Radioactive dating will help us estimate the age of some objects. But ...
Ch 3 Sec 3 Highlighted
... Mass Number: the total number of protons and neutrons in the nucleus of an isotope. From the periodic table the Mass Number is the atomic mass rounded to the nearest whole number. SECTION 3-3 OBJECTIVE 3 Given the identity of a nuclide, determine its number of protons, neutrons, and electrons. ...
... Mass Number: the total number of protons and neutrons in the nucleus of an isotope. From the periodic table the Mass Number is the atomic mass rounded to the nearest whole number. SECTION 3-3 OBJECTIVE 3 Given the identity of a nuclide, determine its number of protons, neutrons, and electrons. ...
Nuts,Bolts and Isotopes- Average Atomic Mass Activity
... (for example carbon is composed of carbon atoms). However, not all of the atoms found in that element are the same. For example, carbon contains three different types of atoms (carbon-12, 13 and 14). Each atom has the same number of protons and electrons but differing numbers of neutrons. These are ...
... (for example carbon is composed of carbon atoms). However, not all of the atoms found in that element are the same. For example, carbon contains three different types of atoms (carbon-12, 13 and 14). Each atom has the same number of protons and electrons but differing numbers of neutrons. These are ...
Chapter 4 Practice Test
... d. Cathode rays were found to be made of protons. All atoms are ____. a. positively charged, with the number of protons exceeding the number of electrons b. negatively charged, with the number of electrons exceeding the number of protons c. neutral, with the number of protons equaling the number of ...
... d. Cathode rays were found to be made of protons. All atoms are ____. a. positively charged, with the number of protons exceeding the number of electrons b. negatively charged, with the number of electrons exceeding the number of protons c. neutral, with the number of protons equaling the number of ...
Chapter 3—Time and Geology
... atom (34): The smallest particle of matter that can exist as a chemical element. atomic mass (34): A quantity essentially equivalent to the number of neutrons plus the number of protons in an atomic nucleus. atomic number (34): The number of protons in the nuclei of atoms of a particular element. An ...
... atom (34): The smallest particle of matter that can exist as a chemical element. atomic mass (34): A quantity essentially equivalent to the number of neutrons plus the number of protons in an atomic nucleus. atomic number (34): The number of protons in the nuclei of atoms of a particular element. An ...
Chapter 2 - Chemistry
... Nuclear Model of Atom Ernest Rutherford (British Physicist - 1911) - proposed that metal atom he was studying must be almost entirely empty space with mass concentrated in tiny central core - showed that atoms consisted of a positively charged nucleus at the center of the atom, around which negative ...
... Nuclear Model of Atom Ernest Rutherford (British Physicist - 1911) - proposed that metal atom he was studying must be almost entirely empty space with mass concentrated in tiny central core - showed that atoms consisted of a positively charged nucleus at the center of the atom, around which negative ...
Flavors of the Atom
... All atoms of the same element, have the same number of protons. But may not have the same weight. Some hydrogen atoms weigh twice as much as other hydrogen atoms. The difference is in the number of neutrons. Atoms of the same element but different masses are called isotopes. Isotopes are defined by ...
... All atoms of the same element, have the same number of protons. But may not have the same weight. Some hydrogen atoms weigh twice as much as other hydrogen atoms. The difference is in the number of neutrons. Atoms of the same element but different masses are called isotopes. Isotopes are defined by ...
по темі “Atoms and Molecules. The Periodic Table”
... State University in 1863. In 1865 he became Doctor of Science for his dissertation "On the Combinations of Water with Alcohol". He achieved tenure in 1867, and by 1871 had transformed Saint Petersburg into an internationally recognized center for chemistry research. In 1876, he became obsessed with ...
... State University in 1863. In 1865 he became Doctor of Science for his dissertation "On the Combinations of Water with Alcohol". He achieved tenure in 1867, and by 1871 had transformed Saint Petersburg into an internationally recognized center for chemistry research. In 1876, he became obsessed with ...
Chapter 4 Atoms and Elements
... Barium is on the left side of the periodic table; it is a metal. Iodine is on the right side of the periodic table; it is a nonmetal. Oxygen is on the right side of the periodic table; it is a nonmetal. Tellurium is in the middle-right section of the periodic table, along the line that divides the m ...
... Barium is on the left side of the periodic table; it is a metal. Iodine is on the right side of the periodic table; it is a nonmetal. Oxygen is on the right side of the periodic table; it is a nonmetal. Tellurium is in the middle-right section of the periodic table, along the line that divides the m ...
Review Unit 5
... CHEMICALLY STABLE: Elements that are nonreactive because their last electron shell is completely filled with 8 electrons. (e.g. Neon, Argon, Krypton.) ISOTOPE: ...
... CHEMICALLY STABLE: Elements that are nonreactive because their last electron shell is completely filled with 8 electrons. (e.g. Neon, Argon, Krypton.) ISOTOPE: ...
Atomic Structure PowerPoint Presentation
... the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The atomic mass is calculated by adding the % of 1H mass foun ...
... the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The atomic mass is calculated by adding the % of 1H mass foun ...
Atomic Theory PPT
... Atomic Mass o The atomic mass of an element represents the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The a ...
... Atomic Mass o The atomic mass of an element represents the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The a ...
atom - West Ada
... element’s name. The first letter is always capitalized and any other letter is not. Aluminum (Al), Platinum (Pt) and Cadmium (Cd) are some examples. The origins of some elements are not as obvious as others. Gold (Au) refers to the Latin name for gold, aurum. Lead (Pb) comes from the Latin word plub ...
... element’s name. The first letter is always capitalized and any other letter is not. Aluminum (Al), Platinum (Pt) and Cadmium (Cd) are some examples. The origins of some elements are not as obvious as others. Gold (Au) refers to the Latin name for gold, aurum. Lead (Pb) comes from the Latin word plub ...
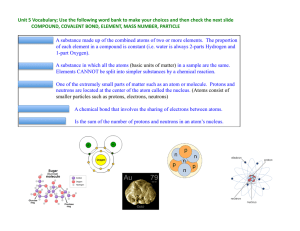
Elements, Compounds, and Mixtures
... A compound contains two or more different atoms joined together. A mixture contains two or more different substances that are only physically joined together, not chemically. A mixture can contain both elements and compounds. ...
... A compound contains two or more different atoms joined together. A mixture contains two or more different substances that are only physically joined together, not chemically. A mixture can contain both elements and compounds. ...
average atomic mass
... Isotopes have mass numbers Isotopes are atoms of the same element (the same number of p+ and e-) but with different masses due to having different numbers of n0. ...
... Isotopes have mass numbers Isotopes are atoms of the same element (the same number of p+ and e-) but with different masses due to having different numbers of n0. ...
Chapter 4 ppt.
... groups based on a set of repeating properties The periodic table allows you to easily compare the properties of one element to another ...
... groups based on a set of repeating properties The periodic table allows you to easily compare the properties of one element to another ...
Document
... Any given element can have more than one isotope. To distinguish between the different isotopes of an atom, the element is named with its mass number, for example, lithium-7. Remember that the mass number is the number of protons and neutrons added together. When symbols are used to represent an iso ...
... Any given element can have more than one isotope. To distinguish between the different isotopes of an atom, the element is named with its mass number, for example, lithium-7. Remember that the mass number is the number of protons and neutrons added together. When symbols are used to represent an iso ...
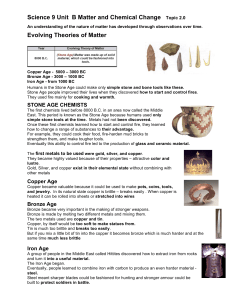
An understanding of the nature of matter has developed
... the mass of a carbon atom (which is 12.0). Atomic mass is measured by atomic mass unit (amu). In 1864, the English chemist John Newlands recognized a pattern when elements were listed by increasing atomic mass. He noticed that properties of elements seemed to repeat through this list at regular inte ...
... the mass of a carbon atom (which is 12.0). Atomic mass is measured by atomic mass unit (amu). In 1864, the English chemist John Newlands recognized a pattern when elements were listed by increasing atomic mass. He noticed that properties of elements seemed to repeat through this list at regular inte ...
IT IS ELEMENTARY - the OLLI at UCI Blog
... plant or animal origin—Sulfur was the exception • 1914-1918 A “golden age” for the military use of chemicals—the first weapons of mass destruction • Since the 1930s Chemical weapons have been used against defenseless peoples in colonial or civil wars ...
... plant or animal origin—Sulfur was the exception • 1914-1918 A “golden age” for the military use of chemicals—the first weapons of mass destruction • Since the 1930s Chemical weapons have been used against defenseless peoples in colonial or civil wars ...
Lawrencium

Lawrencium is a synthetic chemical element with chemical symbol Lr (formerly Lw) and atomic number 103. It is named in honor of Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactive metal, lawrencium is the eleventh transuranic element and is also the final member of the actinide series. Like all elements with atomic number over 100, lawrencium can only be produced in particle accelerators by bombarding lighter elements with charged particles. Twelve isotopes of lawrencium are currently known; the most stable is 266Lr with a half-life of 11 hours, but the shorter-lived 260Lr (half-life 2.7 minutes) is most commonly used in chemistry because it can be produced on a larger scale.Chemistry experiments have confirmed that lawrencium indeed behaves as a heavier homolog to lutetium in the periodic table, and is a trivalent element. It thus could also be classified as the first of the 7th-period transition metals: however, its electron configuration is anomalous for its position in the periodic table, having an s2p configuration instead of the s2d configuration of its homolog lutetium. This means that lawrencium may be less volatile than expected for its position in the periodic table and have a volatility comparable to that of lead.In the 1950s, 1960s, and 1970s, many claims of the synthesis of lawrencium of varying quality were made from laboratories in the Soviet Union and the United States. The priority of the discovery and therefore the naming of the element was disputed between Soviet and American scientists, and while the International Union of Pure and Applied Chemistry (IUPAC) established lawrencium as the official name for the element and gave the American team credit for the discovery, this was reevaluated in 1997, giving both teams shared credit for the discovery but not changing the element's name.