Isotopes File - Northwest ISD Moodle
... Where has the cancer spread? And how can it be treated? In the case of one type of cancer, the answer to both questions may involve the isotope iodine-131. Iodine-131 is an artificial isotope of iodine. This isotope releases radiation that can kill cancer cells, especially those that originated in t ...
... Where has the cancer spread? And how can it be treated? In the case of one type of cancer, the answer to both questions may involve the isotope iodine-131. Iodine-131 is an artificial isotope of iodine. This isotope releases radiation that can kill cancer cells, especially those that originated in t ...
UC Irvine FOCUS! 5 E Lesson Plan Title: Marble Isotope Lab Grade
... nucleus of every atom is made up of neutrons and protons. While neutrons don’t have a charge they do important work such as helping to bind the positive charged protons together via the strong force. Isotopes can have different numbers of protons but the basic elemental structure remains the same. F ...
... nucleus of every atom is made up of neutrons and protons. While neutrons don’t have a charge they do important work such as helping to bind the positive charged protons together via the strong force. Isotopes can have different numbers of protons but the basic elemental structure remains the same. F ...
GOB 3ed Chapter 2 part 1
... B. Atomic Weight The atomic weight is the weighted average of the masses of the naturally occurring isotopes of a particular element reported in atomic mass units. From the periodic table: ...
... B. Atomic Weight The atomic weight is the weighted average of the masses of the naturally occurring isotopes of a particular element reported in atomic mass units. From the periodic table: ...
1 - Groupfusion.net
... 40. An ionic bond forms between what types of elements? A metal and a nonmetal An ionic bond is the attraction between positively charged metal cations and negatively charged anions. In an ionic bond, electrons are transferred from the metal (cation) to the nonmetal (anion). What is the structure of ...
... 40. An ionic bond forms between what types of elements? A metal and a nonmetal An ionic bond is the attraction between positively charged metal cations and negatively charged anions. In an ionic bond, electrons are transferred from the metal (cation) to the nonmetal (anion). What is the structure of ...
Name___________________________________ Physical
... B) Protons are positively charged and the lightest subatomic particle. C) The mass of a neutron nearly equals the mass of a proton. D) Electrons are negatively charged and are the heaviest subatomic particle. E) Neutrons have no charge and are the lightest subatomic particle. ...
... B) Protons are positively charged and the lightest subatomic particle. C) The mass of a neutron nearly equals the mass of a proton. D) Electrons are negatively charged and are the heaviest subatomic particle. E) Neutrons have no charge and are the lightest subatomic particle. ...
Practice problems for chapter 1, 3 and 5 1) A small amount of salt
... the isotopes has an atomic weight of 62.9 amu and constitutes 69.1% of the copper isotopes. The other isotope has an abundance of 30.9%. The atomic weight (amu) of the second isotope is __________ amu. A) 63.2 B) 63.8 C) 64.1 D) 64.8 E) 28.1 15) An unknown element is found to have three naturally oc ...
... the isotopes has an atomic weight of 62.9 amu and constitutes 69.1% of the copper isotopes. The other isotope has an abundance of 30.9%. The atomic weight (amu) of the second isotope is __________ amu. A) 63.2 B) 63.8 C) 64.1 D) 64.8 E) 28.1 15) An unknown element is found to have three naturally oc ...
periodic table elements
... the center of each atom lies the atomic __________________, which consists of _____________and__________. The atomic number refers to the number of ______________ in the nucleus of the atom. Atoms typically have the same number of electrons as the number of protons. All atoms of the same element hav ...
... the center of each atom lies the atomic __________________, which consists of _____________and__________. The atomic number refers to the number of ______________ in the nucleus of the atom. Atoms typically have the same number of electrons as the number of protons. All atoms of the same element hav ...
Atoms, Molecules and Ions
... 1. Atoms make up all matter. 2. The atoms of one element are different from the atoms of another element. 3. Atoms combine in definite ratios to make compounds. 4 Combinations of atoms in compounds can change ...
... 1. Atoms make up all matter. 2. The atoms of one element are different from the atoms of another element. 3. Atoms combine in definite ratios to make compounds. 4 Combinations of atoms in compounds can change ...
Reading Assignment Worksheet on Atoms - District 196 e
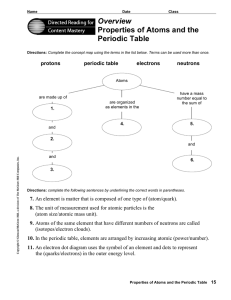
... Directions: Complete the concept map using the terms in the list below. Terms can be used more than once. ...
... Directions: Complete the concept map using the terms in the list below. Terms can be used more than once. ...
PHY–309 L. Solutions for homework set # 10. Textbook question Q
... T = 1 H — have slightly different chemical properties from the main hydrogen isotope 1 H. This difference is important in biochemistry, so drinking too much heavy water (D2 O instead of H2 O) would be bad for your health. But this mass effect is limited to hydrogen isotopes. For all the other elemen ...
... T = 1 H — have slightly different chemical properties from the main hydrogen isotope 1 H. This difference is important in biochemistry, so drinking too much heavy water (D2 O instead of H2 O) would be bad for your health. But this mass effect is limited to hydrogen isotopes. For all the other elemen ...
Chapter 2
... Since some particles were deflected at large angles, Thompson’s model could not be correct. The Nuclear Atom • Rutherford postulated a very small, dense nucleus with the electrons around the outside of the atom. • Most of the volume of the atom is empty space. Other Subatomic Particles • Protons wer ...
... Since some particles were deflected at large angles, Thompson’s model could not be correct. The Nuclear Atom • Rutherford postulated a very small, dense nucleus with the electrons around the outside of the atom. • Most of the volume of the atom is empty space. Other Subatomic Particles • Protons wer ...
File
... Station 5: Cl-Ev-R summary of Beanium Use the following format to create a lab summary report based on your Beanium Lab : How do the different beanium isotopes represent an isotope in real life ? Start your Claim based on what you know about Isotopes. This Report is due on Friday September 13, 2013 ...
... Station 5: Cl-Ev-R summary of Beanium Use the following format to create a lab summary report based on your Beanium Lab : How do the different beanium isotopes represent an isotope in real life ? Start your Claim based on what you know about Isotopes. This Report is due on Friday September 13, 2013 ...
I. The Atomic Concept:
... 2. What is background radiation? ________________________________________________________________ ...
... 2. What is background radiation? ________________________________________________________________ ...
atoms and elements
... An atom is the smallest particle into which an element can be divided and still maintain the properties of that element. All elements are made of atoms. So what’s an element? What makes one element different from another? Let’s find out! Vocabulary: First things first, let’s look at the structure of ...
... An atom is the smallest particle into which an element can be divided and still maintain the properties of that element. All elements are made of atoms. So what’s an element? What makes one element different from another? Let’s find out! Vocabulary: First things first, let’s look at the structure of ...
Atoms, Elements, and the Periodic Table Part 1: The Atomic Model
... 1. Atoms are tiny, invisible particles. ...
... 1. Atoms are tiny, invisible particles. ...
Atoms, Elements, and the Periodic Table Part 1: The Atomic Model
... 1. Atoms are tiny, invisible particles. ...
... 1. Atoms are tiny, invisible particles. ...
Practice problems for chapter 1, 2 and 3 1) A small amount of salt
... the isotopes has an atomic weight of 62.9 amu and constitutes 69.1% of the copper isotopes. The other isotope has an abundance of 30.9%. The atomic weight (amu) of the second isotope is __________ amu. A) 63.2 B) 63.8 C) 64.1 D) 64.8 E) 28.1 15) An unknown element is found to have three naturally oc ...
... the isotopes has an atomic weight of 62.9 amu and constitutes 69.1% of the copper isotopes. The other isotope has an abundance of 30.9%. The atomic weight (amu) of the second isotope is __________ amu. A) 63.2 B) 63.8 C) 64.1 D) 64.8 E) 28.1 15) An unknown element is found to have three naturally oc ...
Honors Chem: Atomic History-Isotopes
... In the nuclear symbol for deuterium H : a.Identify the atomic number and the mass number. b.Write its hyphen notation. Determine the number of protons, neutrons, and electrons in Co–59. How many protons, neutrons, and electrons are in an atom of Ac–221? How many electrons, neutrons, and protons are ...
... In the nuclear symbol for deuterium H : a.Identify the atomic number and the mass number. b.Write its hyphen notation. Determine the number of protons, neutrons, and electrons in Co–59. How many protons, neutrons, and electrons are in an atom of Ac–221? How many electrons, neutrons, and protons are ...
Protons
... Laura works as a consultant at a software company. The amount of her annual bonus is based upon the number of hours she works. Over summer vacation, Debbie has to read a novel for English class. She has decided to spend the same amount of time reading every day. The number of ...
... Laura works as a consultant at a software company. The amount of her annual bonus is based upon the number of hours she works. Over summer vacation, Debbie has to read a novel for English class. She has decided to spend the same amount of time reading every day. The number of ...
PRACTICE PROBLEMS EXAM 1,2 and 3 1311
... the isotopes has an atomic weight of 62.9 amu and constitutes 69.1% of the copper isotopes. The other isotope has an abundance of 30.9%. The atomic weight (amu) of the second isotope is __________ amu. A) 63.2 B) 63.8 C) 64.1 D) 64.8 E) 28.1 15) An unknown element is found to have three naturally oc ...
... the isotopes has an atomic weight of 62.9 amu and constitutes 69.1% of the copper isotopes. The other isotope has an abundance of 30.9%. The atomic weight (amu) of the second isotope is __________ amu. A) 63.2 B) 63.8 C) 64.1 D) 64.8 E) 28.1 15) An unknown element is found to have three naturally oc ...
Unit 3 - Princeton High School
... The half-life for this disintegration is approximately 30 years. This is the amount of time required for half the atoms in a sample to undergo decay. Assume that a 64-gram sample of Cs-137 is analyzed every 30 years for a 150 –year period. Calculate the grams of cesium and barium present each time t ...
... The half-life for this disintegration is approximately 30 years. This is the amount of time required for half the atoms in a sample to undergo decay. Assume that a 64-gram sample of Cs-137 is analyzed every 30 years for a 150 –year period. Calculate the grams of cesium and barium present each time t ...
Chemistry 11 – Course Review
... Element “X” is composed of the following naturally occurring isotopes: Isotope ...
... Element “X” is composed of the following naturally occurring isotopes: Isotope ...
Review: theory vs law the atomic theory contributions of early scientists
... Neutron Heavy (similar to nucleus protons) Oct 711:36 AM ...
... Neutron Heavy (similar to nucleus protons) Oct 711:36 AM ...
Lawrencium

Lawrencium is a synthetic chemical element with chemical symbol Lr (formerly Lw) and atomic number 103. It is named in honor of Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactive metal, lawrencium is the eleventh transuranic element and is also the final member of the actinide series. Like all elements with atomic number over 100, lawrencium can only be produced in particle accelerators by bombarding lighter elements with charged particles. Twelve isotopes of lawrencium are currently known; the most stable is 266Lr with a half-life of 11 hours, but the shorter-lived 260Lr (half-life 2.7 minutes) is most commonly used in chemistry because it can be produced on a larger scale.Chemistry experiments have confirmed that lawrencium indeed behaves as a heavier homolog to lutetium in the periodic table, and is a trivalent element. It thus could also be classified as the first of the 7th-period transition metals: however, its electron configuration is anomalous for its position in the periodic table, having an s2p configuration instead of the s2d configuration of its homolog lutetium. This means that lawrencium may be less volatile than expected for its position in the periodic table and have a volatility comparable to that of lead.In the 1950s, 1960s, and 1970s, many claims of the synthesis of lawrencium of varying quality were made from laboratories in the Soviet Union and the United States. The priority of the discovery and therefore the naming of the element was disputed between Soviet and American scientists, and while the International Union of Pure and Applied Chemistry (IUPAC) established lawrencium as the official name for the element and gave the American team credit for the discovery, this was reevaluated in 1997, giving both teams shared credit for the discovery but not changing the element's name.