File - IGCSE STUDY BANK
... The phrase ‘rate of reaction’ means ‘how fast is the reaction’. It can be measured as the 'rate of formation of product' or the 'rate of disappearance of reactant'. o Rusting is a ‘slow’ reaction, you hardly see any change looking at it!, o weathering of rocks is a very slow reaction, o fermentation ...
... The phrase ‘rate of reaction’ means ‘how fast is the reaction’. It can be measured as the 'rate of formation of product' or the 'rate of disappearance of reactant'. o Rusting is a ‘slow’ reaction, you hardly see any change looking at it!, o weathering of rocks is a very slow reaction, o fermentation ...
8th Grade: First Semester Final Review
... 1. Sample answer: The individual components of a heterogeneous mixture can be seen; the individual components of a homogeneous mixture cannot be seen. The individual components of a homogeneous mixture are evenly mixed; the individual components of a heterogeneous mixture are not evenly mixed. 2. Sa ...
... 1. Sample answer: The individual components of a heterogeneous mixture can be seen; the individual components of a homogeneous mixture cannot be seen. The individual components of a homogeneous mixture are evenly mixed; the individual components of a heterogeneous mixture are not evenly mixed. 2. Sa ...
Chapter 9 Notes - Get a Clue with Mrs. Perdue
... breaking down molecules = __digestive enzymes______ ...
... breaking down molecules = __digestive enzymes______ ...
Chem 2A Final Review
... 66. Chlorophyll, essential to plants for photosynthesis, contains one magnesium atom, three oxygen atoms, four nitrogen atoms, thirty three carbon atoms and twenty nine hydrogen atoms. What is the chemical formula for chlorophyll? 1) FeO3N4Ca33Hy29 2) Mn3O4N33C29H 3) NaO3Ni4Ca33H29 4) MgO3N4C33H29 5 ...
... 66. Chlorophyll, essential to plants for photosynthesis, contains one magnesium atom, three oxygen atoms, four nitrogen atoms, thirty three carbon atoms and twenty nine hydrogen atoms. What is the chemical formula for chlorophyll? 1) FeO3N4Ca33Hy29 2) Mn3O4N33C29H 3) NaO3Ni4Ca33H29 4) MgO3N4C33H29 5 ...
CLASS NOTES- Balancing Chemical Equations.pptx
... 1. Write the correct formula for the reactants and the products ...
... 1. Write the correct formula for the reactants and the products ...
Chemistry 1. The Periodic Table displays the
... between electrons and protons and between atoms and molecules. As a basis for understanding this concept students know: a. atoms combine to form molecules by sharing electrons to form covalent or metallic bonds, or by exchanging electrons to form ionic bonds. b. chemical bonds between atoms in molec ...
... between electrons and protons and between atoms and molecules. As a basis for understanding this concept students know: a. atoms combine to form molecules by sharing electrons to form covalent or metallic bonds, or by exchanging electrons to form ionic bonds. b. chemical bonds between atoms in molec ...
Chemistry Notes for the Whole Year Powerpoint
... of electrons in that orbital. For example, 1s1, the first number means energy level one, s is the orbital, and the superscript number means that there is one electron in this orbital. • Start at hydrogen and fill electrons from left to right starting from there. Count how many electrons are in these ...
... of electrons in that orbital. For example, 1s1, the first number means energy level one, s is the orbital, and the superscript number means that there is one electron in this orbital. • Start at hydrogen and fill electrons from left to right starting from there. Count how many electrons are in these ...
CH 5-7 Chapter 5-7 review wkey
... One of the gases shown below is NOT usually formed in a double replacement reaction. Which one? a) N2 d) NH3 b) CO2 e) H2S c) SO2 ...
... One of the gases shown below is NOT usually formed in a double replacement reaction. Which one? a) N2 d) NH3 b) CO2 e) H2S c) SO2 ...
Expt 8B Equation Writing and Products
... language that is simple and clear. A chemical equation uses formulas and symbols to describe the substances involved in a reaction, the physical state of the substance, the use of a catalyst (a substance that speeds up a reaction without itself being consumed), and relative proportions., The general ...
... language that is simple and clear. A chemical equation uses formulas and symbols to describe the substances involved in a reaction, the physical state of the substance, the use of a catalyst (a substance that speeds up a reaction without itself being consumed), and relative proportions., The general ...
CHEM 20 FINAL EXAM: STUDY HEADINGS Jan 2012
... In an experiment an aqueous solution of Ca(NO3)2 reacts with an aqueous solution of Na3PO4 in a replacement reaction. Indicate the formula for the resulting precipitate: a) Ca2(PO4)3 b) Ca3(PO4)2 c) NaNO3 d) Na2NO3 ...
... In an experiment an aqueous solution of Ca(NO3)2 reacts with an aqueous solution of Na3PO4 in a replacement reaction. Indicate the formula for the resulting precipitate: a) Ca2(PO4)3 b) Ca3(PO4)2 c) NaNO3 d) Na2NO3 ...
Enzymes
... - The ETC is present in inner mitochondrial membrane & is the final common pathway by which electrons derived from different fuels of the body flow to O2 Electron transport & ATP synthesis by oxphos proceed continuously in all tissues that contain mitochondria 1. Structure of mitochondria Components ...
... - The ETC is present in inner mitochondrial membrane & is the final common pathway by which electrons derived from different fuels of the body flow to O2 Electron transport & ATP synthesis by oxphos proceed continuously in all tissues that contain mitochondria 1. Structure of mitochondria Components ...
Introductory Chemistry, 2nd Edition Nivaldo Tro
... Additional aspects of Free Energy • Even though a reaction has a negative G it may occur too slowly to be observed (i.e. combustion). • Thermodynamics gives us the direction of a spontaneous process, it does not give us the rate of the process. • A nonspontaneous process can be driven if coupled w ...
... Additional aspects of Free Energy • Even though a reaction has a negative G it may occur too slowly to be observed (i.e. combustion). • Thermodynamics gives us the direction of a spontaneous process, it does not give us the rate of the process. • A nonspontaneous process can be driven if coupled w ...
Chemical Reactions
... 9. Give the students the reaction and ask them to identify the element oxidized, the element reduced, the oxidizing reactant and the reducing reactant Observations: As the yeast combines with the sugar solution bubbling will occur. Let your students know that bubbling is a sign of a chemical reactio ...
... 9. Give the students the reaction and ask them to identify the element oxidized, the element reduced, the oxidizing reactant and the reducing reactant Observations: As the yeast combines with the sugar solution bubbling will occur. Let your students know that bubbling is a sign of a chemical reactio ...
Question 1. Phosgene was used during the World War - IQ
... Both were connected, one by one, with a half-cell of standard hydrogen electrode and the values recorded were: Half-cell A: Cd2+ + 2 e- = Cd ...
... Both were connected, one by one, with a half-cell of standard hydrogen electrode and the values recorded were: Half-cell A: Cd2+ + 2 e- = Cd ...
Vocabulary CHEM121
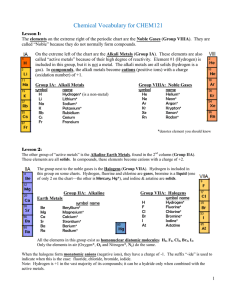
... Note the heavy stair-step line drawn between elements B & Al, etc. This line separates the metals (lower left) from the non-metals (upper right). Metals can only form cations. Non-metals form anions when combined with metals. Elements that touch the line are called metalloids (except Al, which ...
... Note the heavy stair-step line drawn between elements B & Al, etc. This line separates the metals (lower left) from the non-metals (upper right). Metals can only form cations. Non-metals form anions when combined with metals. Elements that touch the line are called metalloids (except Al, which ...
Chemistry - NIC Karnataka
... Concept of oxidation and reduction: classical idea–oxidation (addition of oxygen/ electronegative element or removal of hydrogen/ electropositive element, example for each), reduction - (removal of oxygen/electronegative element or addition of hydrogen/ electropositive element, example for each). Re ...
... Concept of oxidation and reduction: classical idea–oxidation (addition of oxygen/ electronegative element or removal of hydrogen/ electropositive element, example for each), reduction - (removal of oxygen/electronegative element or addition of hydrogen/ electropositive element, example for each). Re ...
Chapter 2 - My Teacher Site
... This reversibility of weak bonding is advantageous because 2 molecules can come together, respond to one another in some way, and then separate ...
... This reversibility of weak bonding is advantageous because 2 molecules can come together, respond to one another in some way, and then separate ...
File
... particles have translational motion (place-to-place) particle arrangement ____________________ - a high-temperature physical state of matter in which atoms lose most of their electrons, particles that make up atoms. Chemical Properties and Chemical Changes ...
... particles have translational motion (place-to-place) particle arrangement ____________________ - a high-temperature physical state of matter in which atoms lose most of their electrons, particles that make up atoms. Chemical Properties and Chemical Changes ...
Chemistry for BIOS 302
... if more protons than electrons, and negative charge if more electrons than protons. • Cations are positively charged: Na+ and Mg2+ are examples. • Anions are negatively charged: Cl- for example. – The prefix “an-” means “not” or “negative”: anonymous = no name; anarchy = no government; anhydrous = n ...
... if more protons than electrons, and negative charge if more electrons than protons. • Cations are positively charged: Na+ and Mg2+ are examples. • Anions are negatively charged: Cl- for example. – The prefix “an-” means “not” or “negative”: anonymous = no name; anarchy = no government; anhydrous = n ...
Practice Exam #2 with Answers
... BaCl2(s) + H2SO4(l)--------------> BaSO4(s) + 2 HCl (g) A mixture of 2.65 g of BaCl2 and 6.78 g of H2SO4 are allowed to react. ...
... BaCl2(s) + H2SO4(l)--------------> BaSO4(s) + 2 HCl (g) A mixture of 2.65 g of BaCl2 and 6.78 g of H2SO4 are allowed to react. ...
Atoms and Elements: Are they Related?
... A burning match is a good example of a chemical reaction. Application of a spark to the chemicals on the match head start the chemical reaction. Signs of a chemical change – heat given off, ...
... A burning match is a good example of a chemical reaction. Application of a spark to the chemicals on the match head start the chemical reaction. Signs of a chemical change – heat given off, ...
Chemistry Syllabus
... mass numbers, and write electron configurations of elements and ions following the Aufbau principle. (DOK 1) 3b. Analyze patterns and trends in the organization of elements in the periodic table and compare their relationship to position in the periodic table. (DOK 2) Atomic number, atomic mass, m ...
... mass numbers, and write electron configurations of elements and ions following the Aufbau principle. (DOK 1) 3b. Analyze patterns and trends in the organization of elements in the periodic table and compare their relationship to position in the periodic table. (DOK 2) Atomic number, atomic mass, m ...
Chemistry Syllabus - Madison County Schools
... boiling points, and vapor pressure when comparing differences in properties of pure substances. (DOK 1) 2g. Develop a three-dimensional model of molecular structure. (DOK 2) Lewis dot structures for simple molecules and ionic compounds Valence shell electron pair repulsion theory (VSEPR) ...
... boiling points, and vapor pressure when comparing differences in properties of pure substances. (DOK 1) 2g. Develop a three-dimensional model of molecular structure. (DOK 2) Lewis dot structures for simple molecules and ionic compounds Valence shell electron pair repulsion theory (VSEPR) ...
An object is thrown from a cliff.
... Reference: Ties sentences together A ball fell from a cliff. The ball weighs 10 N. ...
... Reference: Ties sentences together A ball fell from a cliff. The ball weighs 10 N. ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.