Unit: Corrosion Science Important Questions with Hints
... 16. Pure copper is immune to stress corrosion than Brass. Why? Hints: Brass is an alloy of copper and zinc. So, in corroding environment containing traces of ammonia or amines, copper and zinc present in brass form complexes [Cu(NH3)4]2+ and [Zn(NH3)4]2+ . This results in dissolution of brass leadin ...
... 16. Pure copper is immune to stress corrosion than Brass. Why? Hints: Brass is an alloy of copper and zinc. So, in corroding environment containing traces of ammonia or amines, copper and zinc present in brass form complexes [Cu(NH3)4]2+ and [Zn(NH3)4]2+ . This results in dissolution of brass leadin ...
equilibrium questions - Southington Public Schools
... (i) Which of the two acids is stronger, HOCl or HOBr? Justify your answer in terms of Ka. (ii) Draw a complete Lewis electron-dot diagram for the acid that you identified in part (a)(i). (iii) Hypoiodous acid has the formula HOI. Predict whether HOI is a stronger acid or a weaker acid than the acid ...
... (i) Which of the two acids is stronger, HOCl or HOBr? Justify your answer in terms of Ka. (ii) Draw a complete Lewis electron-dot diagram for the acid that you identified in part (a)(i). (iii) Hypoiodous acid has the formula HOI. Predict whether HOI is a stronger acid or a weaker acid than the acid ...
Chapter 16.1
... • example: the Hf0 of carbon dioxide is –393.5 kJ per mol of gas produced. • Elements in their standard states are defined as having Hf0 = 0. • This indicates that carbon dioxide is more stable than the elements from which it was formed. ...
... • example: the Hf0 of carbon dioxide is –393.5 kJ per mol of gas produced. • Elements in their standard states are defined as having Hf0 = 0. • This indicates that carbon dioxide is more stable than the elements from which it was formed. ...
Chapter 1 Review Questions
... The percent by volume concentration of the dimethicone solution is 0.57%. 25. a. Because like charges repel each other, the slightly negative charge on the surface of the hair repels the negative charge on the head of the anionic surfactant. Since the oily layer of sebum and other debris is attached ...
... The percent by volume concentration of the dimethicone solution is 0.57%. 25. a. Because like charges repel each other, the slightly negative charge on the surface of the hair repels the negative charge on the head of the anionic surfactant. Since the oily layer of sebum and other debris is attached ...
1 Mole
... Standard 3a: describing chemical reactions An equation describes a chemical reaction or ...
... Standard 3a: describing chemical reactions An equation describes a chemical reaction or ...
AP Chemistry - Problem Drill 03: Matter and Energy Question No. 1
... Question No. 4 of 10 Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 4. What type of statement is the following: “The Alkaseltzer will react with the water faster ...
... Question No. 4 of 10 Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 4. What type of statement is the following: “The Alkaseltzer will react with the water faster ...
Building the sense of math in physics activities
... B.2 If the viscosity of air is about 10-3 kg/m-s, find the value of the Reynolds number for the coffee filter falling in air. B.3 From your calculation, which force do you expect to dominate for a falling coffee filter? 3. Different cells have many of the same components but they often have differen ...
... B.2 If the viscosity of air is about 10-3 kg/m-s, find the value of the Reynolds number for the coffee filter falling in air. B.3 From your calculation, which force do you expect to dominate for a falling coffee filter? 3. Different cells have many of the same components but they often have differen ...
UNIVERSITY OF TARTU THE GIFTED AND
... 5. Precisely half a litre of a mixture of gases (at standard conditions) comprised of two substances: A and B. A is an acyclic alkane and there is 8,93 mmol of A in the mixture. B does not contain hydrogen. The mixture was ignited after adding 1,857 g of oxygen. When the reaction was complete, only ...
... 5. Precisely half a litre of a mixture of gases (at standard conditions) comprised of two substances: A and B. A is an acyclic alkane and there is 8,93 mmol of A in the mixture. B does not contain hydrogen. The mixture was ignited after adding 1,857 g of oxygen. When the reaction was complete, only ...
Laboratory Practices from Physical Chemistry
... temperature Tk. Only the end of the flask neck (1–2 cm) has to project from the bath. During sample evaporation, all air from the flask escapes and the flask contains just sample vapor. Sample evaporation process can be monitored using a small piece of filter paper set to the flask neck. When vapor ...
... temperature Tk. Only the end of the flask neck (1–2 cm) has to project from the bath. During sample evaporation, all air from the flask escapes and the flask contains just sample vapor. Sample evaporation process can be monitored using a small piece of filter paper set to the flask neck. When vapor ...
Redox
... 2H (aq) + 2e → H2 (g) What happened to chloride? It didn’t change (Cl- on both sides of the equation). Ions that don’t change in a reaction are called spectator ions. ...
... 2H (aq) + 2e → H2 (g) What happened to chloride? It didn’t change (Cl- on both sides of the equation). Ions that don’t change in a reaction are called spectator ions. ...
OCR Document
... - titration by Winkler's method - spectrophotometry of oxidized dyes - electrochemically by Makareth O2 electrode – Some important terms: - TOC: total organic carbon; determined by oxidizing all organic C to CO2 and analyzing with gas chromatography - COD: chemical oxygen demand; react with Na2Cr2O7 ...
... - titration by Winkler's method - spectrophotometry of oxidized dyes - electrochemically by Makareth O2 electrode – Some important terms: - TOC: total organic carbon; determined by oxidizing all organic C to CO2 and analyzing with gas chromatography - COD: chemical oxygen demand; react with Na2Cr2O7 ...
Supplementary Exercise 1B Topic 5
... Option B –Iron(II) sulphate solution is the reducing agent. Iron(II) ions are oxidized to iron(III) ions. Option C –The oxidation number of potassium remains the same. Option D –The reaction mixture appears yellow due to the presence of iron(III) ions. ...
... Option B –Iron(II) sulphate solution is the reducing agent. Iron(II) ions are oxidized to iron(III) ions. Option C –The oxidation number of potassium remains the same. Option D –The reaction mixture appears yellow due to the presence of iron(III) ions. ...
C. 3.5 g
... exposed to sunlight for half an hour and then inverted over water. The volume of gas remained in the tube will be about ...
... exposed to sunlight for half an hour and then inverted over water. The volume of gas remained in the tube will be about ...
Syllabus of Medical / Dental Colleges Entrance Test 2016
... Explain the terms pH; Ka; pKa; Kw and use them in calculations ...
... Explain the terms pH; Ka; pKa; Kw and use them in calculations ...
How to Use Reaction Stoichiometry
... Figure 4.6 (a) When an octane molecule undergoes complete combustion, it forms carbon dioxide and water: one CO2 molecule is formed for each carbon atom present (yellow arrows). (b) However, in a limited supply of oxygen, some of the carbon atoms end up as carbon monoxide molecules, CO, so the yiel ...
... Figure 4.6 (a) When an octane molecule undergoes complete combustion, it forms carbon dioxide and water: one CO2 molecule is formed for each carbon atom present (yellow arrows). (b) However, in a limited supply of oxygen, some of the carbon atoms end up as carbon monoxide molecules, CO, so the yiel ...
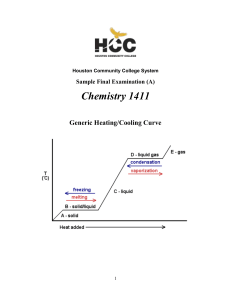
1411FINALSAMPLEs and Key
... Since there are a total of four atoms plus lone pairs (four “electron domains”) around the central sulfur, the overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not s ...
... Since there are a total of four atoms plus lone pairs (four “electron domains”) around the central sulfur, the overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not s ...
1 of 52
... The following questions represent the baseline knowledge you will need to succeed in AP Chemistry. The goal of this assignment is twofold. First, it will tell you what you are up against in the upcoming school year. Second, this assignment covers curriculum that is basic and will allow us the time t ...
... The following questions represent the baseline knowledge you will need to succeed in AP Chemistry. The goal of this assignment is twofold. First, it will tell you what you are up against in the upcoming school year. Second, this assignment covers curriculum that is basic and will allow us the time t ...
OCR Document
... - titration by Winkler's method - spectrophotometry of oxidized dyes - electrochemically by Makareth O2 electrode – Some important terms: - TOC: total organic carbon; determined by oxidizing all organic C to CO2 and analyzing with gas chromatography - COD: chemical oxygen demand; react with Na2Cr2O7 ...
... - titration by Winkler's method - spectrophotometry of oxidized dyes - electrochemically by Makareth O2 electrode – Some important terms: - TOC: total organic carbon; determined by oxidizing all organic C to CO2 and analyzing with gas chromatography - COD: chemical oxygen demand; react with Na2Cr2O7 ...
111 Exam III OUTLINE TRO 1-3-11
... When equilibrium between substances involve two or more phases it is called Heterogeneous Equilibria. The concentration of a pure solid or a pure liquid in their standard states is constant (at constant T° and P). Therefore, the concentrations of solids or liquids involved in a heterogeneous equilib ...
... When equilibrium between substances involve two or more phases it is called Heterogeneous Equilibria. The concentration of a pure solid or a pure liquid in their standard states is constant (at constant T° and P). Therefore, the concentrations of solids or liquids involved in a heterogeneous equilib ...
Stoichiometry
... E.x: Calculate the number of carbon atoms and the number of hydrogen atoms in 600g of propane, C3 H8 C = 12 , H = 1. MW = ( 3 x 12 ) + ( 8 x 1 ) = 44 amu Number of moles of propane = 600 g x 1 mol = 13.63 mol ...
... E.x: Calculate the number of carbon atoms and the number of hydrogen atoms in 600g of propane, C3 H8 C = 12 , H = 1. MW = ( 3 x 12 ) + ( 8 x 1 ) = 44 amu Number of moles of propane = 600 g x 1 mol = 13.63 mol ...
PDF - ACS Publications - American Chemical Society
... Successful penetration of lithium−sulfur (Li−S) technology into the rechargeable battery market could serve the thirst for higher energy density battery systems in the electric-transport sector. These batteries that utilize lithium metal as the negative electrode and sulfur as positive electrode ben ...
... Successful penetration of lithium−sulfur (Li−S) technology into the rechargeable battery market could serve the thirst for higher energy density battery systems in the electric-transport sector. These batteries that utilize lithium metal as the negative electrode and sulfur as positive electrode ben ...
Absorption of Flue-Gas Components by Ionic Liquids
... The total absorption capacity for SO2 (1500 ppm in N2) for the 20 wt% [TMGH]ClSiO2 SILP absorber at the given conditions is determined by integration of the area of absorption within the curve from 10 to ca. 80 min to be 0.05 mole fraction SO2 in the IL for the dry gas and reduced by ca. 35% in the ...
... The total absorption capacity for SO2 (1500 ppm in N2) for the 20 wt% [TMGH]ClSiO2 SILP absorber at the given conditions is determined by integration of the area of absorption within the curve from 10 to ca. 80 min to be 0.05 mole fraction SO2 in the IL for the dry gas and reduced by ca. 35% in the ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.