Fundamentals Diagnostic Quiz
... to yield copper(II)sulfate, water, and sulfur dioxide. *a) Cu + 2 H2SO4 -> CuSO4 + 2 H2O + SO2 b) 2 Cu + s H2S -> Cu2S + 3 H20 + S20 c) Cu + H2SO4 -> Cu2SO4 + H2O + SO2 d) 2 Cu + 2 H2SO4 -> 2 CuSO4 + 4 H2O + S2O e) Cu + H2S + 3O2 -> CuSO4 + H2O + SO2 ...
... to yield copper(II)sulfate, water, and sulfur dioxide. *a) Cu + 2 H2SO4 -> CuSO4 + 2 H2O + SO2 b) 2 Cu + s H2S -> Cu2S + 3 H20 + S20 c) Cu + H2SO4 -> Cu2SO4 + H2O + SO2 d) 2 Cu + 2 H2SO4 -> 2 CuSO4 + 4 H2O + S2O e) Cu + H2S + 3O2 -> CuSO4 + H2O + SO2 ...
H o - CashmereChemistry
... 1. Write the data in the form of equations 2. Rewrite the equations to give the desired species on the correct side of the equation. If the reaction must be reversed (perhaps because we require a species to be a reactant and not a product) then the sign of the H must also be ...
... 1. Write the data in the form of equations 2. Rewrite the equations to give the desired species on the correct side of the equation. If the reaction must be reversed (perhaps because we require a species to be a reactant and not a product) then the sign of the H must also be ...
AJR Ch6 Thermochemistry.docx Slide 1 Chapter 6
... To discuss the energy changes associated with chemical reactions we must first define the specific part of the universe that is of interest to us, which we call the “system”. System - what we study (e.g. a beaker or flask). Surroundings - the rest of the universe outside of our system (e.g. the room ...
... To discuss the energy changes associated with chemical reactions we must first define the specific part of the universe that is of interest to us, which we call the “system”. System - what we study (e.g. a beaker or flask). Surroundings - the rest of the universe outside of our system (e.g. the room ...
vce chemistry trial exam 1
... A is incorrect because the components have different areas under the peaks, which indicates they are present in different quantities. The second peak has approximately twice the area under the peak compared to the first peak, so is present in twice the amount of the other. C is incorrect because onl ...
... A is incorrect because the components have different areas under the peaks, which indicates they are present in different quantities. The second peak has approximately twice the area under the peak compared to the first peak, so is present in twice the amount of the other. C is incorrect because onl ...
5.1 questions - DrBravoChemistry
... Use this information to calculate the temperature at which this reaction becomes feasible. ...
... Use this information to calculate the temperature at which this reaction becomes feasible. ...
Table of Contents
... Some simple methods also exist for separating homogeneous mixtures. A solid dissolved in a liquid solution can be separated by letting it dry out in the process of ___________________. Mixtures are separated into pure _____________________. A pure substance always has the same composition. Pure subs ...
... Some simple methods also exist for separating homogeneous mixtures. A solid dissolved in a liquid solution can be separated by letting it dry out in the process of ___________________. Mixtures are separated into pure _____________________. A pure substance always has the same composition. Pure subs ...
Ch. 5
... - Temperature (always in K ; K = C +273) - Volume - amount (usually measured in moles) 3. The Ideal Gas Law a. PV = nRT , where : R = universal gas constant (.08206 L·atm/K·mol) - it is important to remember that the gas laws describe ideal gases, not real gases - real gas behavior approaches ideal ...
... - Temperature (always in K ; K = C +273) - Volume - amount (usually measured in moles) 3. The Ideal Gas Law a. PV = nRT , where : R = universal gas constant (.08206 L·atm/K·mol) - it is important to remember that the gas laws describe ideal gases, not real gases - real gas behavior approaches ideal ...
GROUP 13 ELEMENTS -THE BORON FAMILY -
... Gallium has the chemical symbol Ga and atomic number 31. It has the electron configuration [Ar]2s2 2p1 and +3 oxidation state. The melting point is 29.8º C and therefore melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert ...
... Gallium has the chemical symbol Ga and atomic number 31. It has the electron configuration [Ar]2s2 2p1 and +3 oxidation state. The melting point is 29.8º C and therefore melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert ...
The Complete Notes - Joliet Junior College
... remembering. An analogy would be this: you read all the books out there on the subject of golf, but don’t get round to swinging a club – what do you think happens when you tee off for the first time? ...
... remembering. An analogy would be this: you read all the books out there on the subject of golf, but don’t get round to swinging a club – what do you think happens when you tee off for the first time? ...
Higher Chemistry Specimen Question Paper
... (b) The label on a tub of margarine states that 100 g of the margarine contains 0·70 g of sodium. The sodium is present as sodium chloride. Calculate the mass of sodium chloride, in grams, present in a 10 g portion of the margarine. The mass of one mole of sodium chloride, NaCl, is 58·5 g. ...
... (b) The label on a tub of margarine states that 100 g of the margarine contains 0·70 g of sodium. The sodium is present as sodium chloride. Calculate the mass of sodium chloride, in grams, present in a 10 g portion of the margarine. The mass of one mole of sodium chloride, NaCl, is 58·5 g. ...
Note Sheets and Sample Problems
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
Section 5
... Smaller and highly charged cations (hard) like Al3+, Fe3+, and Ti3+ are better at pulling away electron density from water molecules than larger ions, thus these aquated ions would be expected to be quite acidic: [Al(H2O)6]3+(aq) + H2O(l) ⇌ H3O+(aq) + [Al(H2O)5(OH)]2+(aq) pKa = 5.0 [Ti(H2O)6]3+(aq) ...
... Smaller and highly charged cations (hard) like Al3+, Fe3+, and Ti3+ are better at pulling away electron density from water molecules than larger ions, thus these aquated ions would be expected to be quite acidic: [Al(H2O)6]3+(aq) + H2O(l) ⇌ H3O+(aq) + [Al(H2O)5(OH)]2+(aq) pKa = 5.0 [Ti(H2O)6]3+(aq) ...
Chemistry 101: The Complete Notes
... remembering. An analogy would be this: you read all the books out there on the subject of golf, but don‟t get round to swinging a club – what do you think happens when you tee off for the first time? ...
... remembering. An analogy would be this: you read all the books out there on the subject of golf, but don‟t get round to swinging a club – what do you think happens when you tee off for the first time? ...
Word - chemmybear.com
... 11. Ammonium chloride is placed inside a closed vessel where it comes into equilibrium at 400C according to the equation shown. Only these three substances are present inside the vessel. If Kp for the system at 400C is 0.640, what is the pressure inside the vessel? NH4Cl(s) NH3(g) + HCl(g) 12. Bro ...
... 11. Ammonium chloride is placed inside a closed vessel where it comes into equilibrium at 400C according to the equation shown. Only these three substances are present inside the vessel. If Kp for the system at 400C is 0.640, what is the pressure inside the vessel? NH4Cl(s) NH3(g) + HCl(g) 12. Bro ...
South Pasadena · AP Chemistry
... 11. Ammonium chloride is placed inside a closed vessel where it comes into equilibrium at 400C according to the equation shown. Only these three substances are present inside the vessel. If Kp for the system at 400C is 0.640, what is the pressure inside the vessel? NH4Cl(s) NH3(g) + HCl(g) 12. Bro ...
... 11. Ammonium chloride is placed inside a closed vessel where it comes into equilibrium at 400C according to the equation shown. Only these three substances are present inside the vessel. If Kp for the system at 400C is 0.640, what is the pressure inside the vessel? NH4Cl(s) NH3(g) + HCl(g) 12. Bro ...
Experimental determination of hydromagnesite precipitation rates
... under conditions prevalent in geothermal power plants (i.e. high temperatures and high flow rates) is still in its infancy. Here we present results from a study of silica precipitates that formed inside the heat exchanger unit used for the production of thermal energy at the Hellisheiði power statio ...
... under conditions prevalent in geothermal power plants (i.e. high temperatures and high flow rates) is still in its infancy. Here we present results from a study of silica precipitates that formed inside the heat exchanger unit used for the production of thermal energy at the Hellisheiði power statio ...
CHEM-1411 Final Practice Exam
... Since there are a total of four atoms plus lone pairs (four “electron domains”) around the central sulfur, the overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not s ...
... Since there are a total of four atoms plus lone pairs (four “electron domains”) around the central sulfur, the overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not s ...
1411FINALSAMPLE+KEY - Houston Community College
... Since there are a total of four atoms plus lone pairs (four “electron domains”) around the central sulfur, the overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not s ...
... Since there are a total of four atoms plus lone pairs (four “electron domains”) around the central sulfur, the overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not s ...
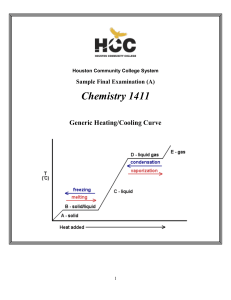
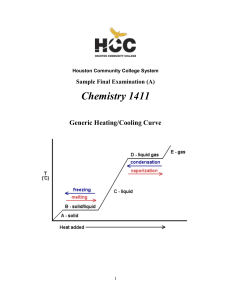
Thermochemistry 122
... the particles move further apart from each other, the substance expands. The liquid in the thermometer rises as the kinetic energy increases. Temperature is a measure of the average translational kinetic energy. When particles collide, a chemical reaction may occur. Chemical bonds are broken and new ...
... the particles move further apart from each other, the substance expands. The liquid in the thermometer rises as the kinetic energy increases. Temperature is a measure of the average translational kinetic energy. When particles collide, a chemical reaction may occur. Chemical bonds are broken and new ...
Chapter 17 - Cengage Learning
... The collision model says that in order for molecules to react with each other, they must first collide. Increases in the temperature and concentration of reactants bring about more collisions, and the rate of reaction increases. The collision model explains many observations about reactions. Not all ...
... The collision model says that in order for molecules to react with each other, they must first collide. Increases in the temperature and concentration of reactants bring about more collisions, and the rate of reaction increases. The collision model explains many observations about reactions. Not all ...
File
... the particles move further apart from each other, the substance expands. The liquid in the thermometer rises as the kinetic energy increases. Temperature is a measure of the average translational kinetic energy. When particles collide, a chemical reaction may occur. Chemical bonds are broken and new ...
... the particles move further apart from each other, the substance expands. The liquid in the thermometer rises as the kinetic energy increases. Temperature is a measure of the average translational kinetic energy. When particles collide, a chemical reaction may occur. Chemical bonds are broken and new ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.