In chemistry, a salt is an ionic compound that
... Salts of strong acids and strong bases ("strong salts") are non-volatile and odorless, whereas salts of either weak acids or weak bases ("weak salts") may smell after the conjugate acid (e.g., acetates like acetic acid (vinegar) and cyanides like hydrogen cyanide (almonds)) or the conjugate base (e. ...
... Salts of strong acids and strong bases ("strong salts") are non-volatile and odorless, whereas salts of either weak acids or weak bases ("weak salts") may smell after the conjugate acid (e.g., acetates like acetic acid (vinegar) and cyanides like hydrogen cyanide (almonds)) or the conjugate base (e. ...
chemistry
... Gasoline is a mixture composed primarily of hydrocarbons such as isooctane, which is also known as 2,2,4-trimethylpentane. Gasoline is assigned a number called an octane rating. Gasoline with an octane rating of 87 performs the same as a mixture that consists of 87% isooctane and 13% heptane. An alt ...
... Gasoline is a mixture composed primarily of hydrocarbons such as isooctane, which is also known as 2,2,4-trimethylpentane. Gasoline is assigned a number called an octane rating. Gasoline with an octane rating of 87 performs the same as a mixture that consists of 87% isooctane and 13% heptane. An alt ...
The Gibbs Function of a Chemical Reaction*
... The ice-water equilibrium is a type of phase equilibrium which is established when the molar Gibbs functions of the two phases are equal. At higher temperatures the Gibbs function of ice is higher than that of water and the spontaneous change from higher to lower Gibbs function is associated with th ...
... The ice-water equilibrium is a type of phase equilibrium which is established when the molar Gibbs functions of the two phases are equal. At higher temperatures the Gibbs function of ice is higher than that of water and the spontaneous change from higher to lower Gibbs function is associated with th ...
Chapter 5 Thermochemistry
... Thermodynamics is the study of energy and its transformations. All chemical changes involve a transfer of energy, be it into the reaction or out of the reaction. Transformed energy in a chemical reaction comes from or forms chemical bonds and is exchanged with the surroundings as heat and/or work. W ...
... Thermodynamics is the study of energy and its transformations. All chemical changes involve a transfer of energy, be it into the reaction or out of the reaction. Transformed energy in a chemical reaction comes from or forms chemical bonds and is exchanged with the surroundings as heat and/or work. W ...
COURSE GUIDE CHM 292 Practical Chemistry IV
... easily prepared by the oxidation of 2-propanol with acidic dichromate, a reaction that you will carry out in this lab. The acetone product will be purified using a distillation. Physical properties Because they contain the polar carbonyl group, aldehydes and ketones are polar compounds. However, the ...
... easily prepared by the oxidation of 2-propanol with acidic dichromate, a reaction that you will carry out in this lab. The acetone product will be purified using a distillation. Physical properties Because they contain the polar carbonyl group, aldehydes and ketones are polar compounds. However, the ...
A matter of Equilibrium
... Such a state is a dynamical equilibrium – both the forward and backward reactions are proceeding simultaneously but the rates of each balance one another. In other words if we imagine following a particular H atom we would see that it spends some of its time bound in ammonia molecules and some time ...
... Such a state is a dynamical equilibrium – both the forward and backward reactions are proceeding simultaneously but the rates of each balance one another. In other words if we imagine following a particular H atom we would see that it spends some of its time bound in ammonia molecules and some time ...
Mole-Volume Conversion Assignment
... Yesterday’s calculations we found out that when we use 50mL of 5% acetic acid solutions, we require 3.5g of sodium bicarbonate to completely react. Trial 1: use 1.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left over: NaHCO3 all used Trial 2: use 2.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left ...
... Yesterday’s calculations we found out that when we use 50mL of 5% acetic acid solutions, we require 3.5g of sodium bicarbonate to completely react. Trial 1: use 1.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left over: NaHCO3 all used Trial 2: use 2.0g of NaHCO3 and 50mL CH3COOH: extra vinegar left ...
Thermochemistry
... • Chemical energy - The energy stored within the structural units of chemical substances; its quantity is determined by the type and arrangement of constituent atoms. – When substances participate in chemical reactions, chemical energy is released, stored, or converted to other forms of energy. – C ...
... • Chemical energy - The energy stored within the structural units of chemical substances; its quantity is determined by the type and arrangement of constituent atoms. – When substances participate in chemical reactions, chemical energy is released, stored, or converted to other forms of energy. – C ...
Science SOL CH
... The Science Vertical Team has revised the Chemistry Curriculum Matrix for 2009-2010. In addition to the necessary correlation to the Virginia Science Standards of Learning, the Chemistry content is organized by both concepts and topics. We encourage you to utilize this document while planning for in ...
... The Science Vertical Team has revised the Chemistry Curriculum Matrix for 2009-2010. In addition to the necessary correlation to the Virginia Science Standards of Learning, the Chemistry content is organized by both concepts and topics. We encourage you to utilize this document while planning for in ...
STOICHIOMETRY REVIEW WORKSHEET
... (a) How many moles of water formed? (b) How many moles of butane burned? (c) How many grams of butane burned? (d) How much oxygen was used up in moles? (e) How much oxygen was used up in grams? 2) Using the following equation: NaOH + ...
... (a) How many moles of water formed? (b) How many moles of butane burned? (c) How many grams of butane burned? (d) How much oxygen was used up in moles? (e) How much oxygen was used up in grams? 2) Using the following equation: NaOH + ...
support material
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
Chemistry Olympiad Support Booklet
... relatively easy recall of information which students will have met before, but instead on thinking and trying to work out answers to unfamiliar questions – this is much more difficult. The recent Round 1 questions, which are contained in this booklet, are typical; they may seem ‘impossible’ at first ...
... relatively easy recall of information which students will have met before, but instead on thinking and trying to work out answers to unfamiliar questions – this is much more difficult. The recent Round 1 questions, which are contained in this booklet, are typical; they may seem ‘impossible’ at first ...
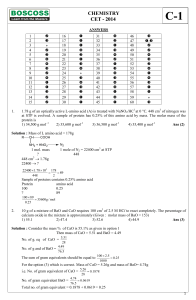
CHEMISTRY CET
... An incorrect statement with respect to SN1 and SN2 mechanisms for alkyl halide is (1) Competing reaction for an SN2 reaction is rearrangement. (2) A weak nucleophile and a protic solvent increases the rate or favours SN1 reaction. (3) A strong nucleophile in an aprotic solvent increases the rate or ...
... An incorrect statement with respect to SN1 and SN2 mechanisms for alkyl halide is (1) Competing reaction for an SN2 reaction is rearrangement. (2) A weak nucleophile and a protic solvent increases the rate or favours SN1 reaction. (3) A strong nucleophile in an aprotic solvent increases the rate or ...
Key Words Questions and Problems
... For many years chloroform (CHCl3) was used as an inhalation anesthetic in spite of the fact that it is also a toxic substance that may cause severe liver, kidney, and heart damage. Calculate the percent composition by mass of this compound. Cinnamic alcohol is used mainly in perfumery, particularly ...
... For many years chloroform (CHCl3) was used as an inhalation anesthetic in spite of the fact that it is also a toxic substance that may cause severe liver, kidney, and heart damage. Calculate the percent composition by mass of this compound. Cinnamic alcohol is used mainly in perfumery, particularly ...
Chapter 5: Thermochemistry
... more rapidly and its temperature increases. There are three factors that control the magnitude of a temperature change for an object: the amount of heat energy added to the object, the mass of the object, and the material the object is made of. Consider lighting a match and using it to heat a large ...
... more rapidly and its temperature increases. There are three factors that control the magnitude of a temperature change for an object: the amount of heat energy added to the object, the mass of the object, and the material the object is made of. Consider lighting a match and using it to heat a large ...
Final Exam - KFUPM Faculty List
... B) The change in enthalpy, ΔH, for a process is equal to the amount of heat absorbed at constant pressure, qP correct: ΔH = qP C) A bomb calorimeter measures ΔH directly false: by definition V is constant in a bomb calorimeter and not P, thus it measures ΔE directly not ΔH D) If a process gives off ...
... B) The change in enthalpy, ΔH, for a process is equal to the amount of heat absorbed at constant pressure, qP correct: ΔH = qP C) A bomb calorimeter measures ΔH directly false: by definition V is constant in a bomb calorimeter and not P, thus it measures ΔE directly not ΔH D) If a process gives off ...
Form 3 Chem. Term 1 Notes.FINAL.
... Avagadro’s constant. The amount of any substance that contains Avagadro’s number of particles is called a mole. A mole is the standard counting unit or SI unit of the amount of a substance. The mass in grams of one mole of a substance is called molar mass. For elements and compounds the molar mass o ...
... Avagadro’s constant. The amount of any substance that contains Avagadro’s number of particles is called a mole. A mole is the standard counting unit or SI unit of the amount of a substance. The mass in grams of one mole of a substance is called molar mass. For elements and compounds the molar mass o ...
Hydrogen dissociation under equilibrium and non
... The hydrogen dissociation reaction is important in many instances, such as for the hydrogen society; as pure hydrogen is needed for fuel cells in electric vehicles. Additionally, as it is a simple reaction, it is also well suited as a model system for chemical reactions. The phase diagram of hydroge ...
... The hydrogen dissociation reaction is important in many instances, such as for the hydrogen society; as pure hydrogen is needed for fuel cells in electric vehicles. Additionally, as it is a simple reaction, it is also well suited as a model system for chemical reactions. The phase diagram of hydroge ...
Document
... The boiling point is the temperature at which the (equilibrium) vapor pressure of a liquid is equal to the external pressure. (normal B.P. is at 1 atm) Heat of vaporization is the energy required to convert one mole of a substance from liquid to gas. ...
... The boiling point is the temperature at which the (equilibrium) vapor pressure of a liquid is equal to the external pressure. (normal B.P. is at 1 atm) Heat of vaporization is the energy required to convert one mole of a substance from liquid to gas. ...
Holt Modern Chemistry Workbook: ch 11
... flat feet. The same force applied to a smaller area results in greater pressure. Atmospheric Pressure The atmosphere, which is the air that surrounds Earth, exerts pressure on Earth’s surface. This pressure is equivalent to a 1.03 kg mass sitting on every square centimeter of Earth. The resulting pr ...
... flat feet. The same force applied to a smaller area results in greater pressure. Atmospheric Pressure The atmosphere, which is the air that surrounds Earth, exerts pressure on Earth’s surface. This pressure is equivalent to a 1.03 kg mass sitting on every square centimeter of Earth. The resulting pr ...
Multiple Pathways To Success Quarter 3 Learning Module
... a. List the formulas and names for the two compounds. b. What is different about the spelling of the prefix meaning “one” in these two names? 11. Identify any remaining names of compounds in Model 2 where the prefixes that do not exactly match the spelling shown in the prefix table. 12. Use your ans ...
... a. List the formulas and names for the two compounds. b. What is different about the spelling of the prefix meaning “one” in these two names? 11. Identify any remaining names of compounds in Model 2 where the prefixes that do not exactly match the spelling shown in the prefix table. 12. Use your ans ...
Document
... happening under standard conditions. All reactions will be carried out under standard conditions. In the common chemistry laboratory, this reaction cannot be examined directly. This is because, regardless of the low enthalpy, the reaction requires a very, very high activation energy to get the react ...
... happening under standard conditions. All reactions will be carried out under standard conditions. In the common chemistry laboratory, this reaction cannot be examined directly. This is because, regardless of the low enthalpy, the reaction requires a very, very high activation energy to get the react ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.


![[SiO4] tetrahedra form hydrogen bonds with the oxygens of the](http://s1.studyres.com/store/data/022276039_1-293e6a385c9abee7367767b263cc0a85-300x300.png)