CH 5-7 Chapter 5-7 review wkey
... 35. Consider the thermal energy transfer during a chemical process. When heat is transferred to the system, the process is said to be _______ and the sign of H is ________. a) exothermic, positive b) endothermic, negative c) exothermic, negative d) endothermic, positive ...
... 35. Consider the thermal energy transfer during a chemical process. When heat is transferred to the system, the process is said to be _______ and the sign of H is ________. a) exothermic, positive b) endothermic, negative c) exothermic, negative d) endothermic, positive ...
Final review packet
... 1. Compare the parts of an atom based on location, charge and mass: - proton - neutron - electron 2. Define: - isotope - ion - atomic number - mass number - atomic mass unit 3. How many neutrons does U-238 have? 4. Write isotope notation for the particle that contains 17 neutrons and 15 protons. 5. ...
... 1. Compare the parts of an atom based on location, charge and mass: - proton - neutron - electron 2. Define: - isotope - ion - atomic number - mass number - atomic mass unit 3. How many neutrons does U-238 have? 4. Write isotope notation for the particle that contains 17 neutrons and 15 protons. 5. ...
Thursday, March 27, 2008
... A 100.00-gram sample of naturally occurring boron contains 19.78 grams of boron-10 (atomic mass = 10.01 atomic mass units) and 80.22 grams of boron-11 (atomic mass = 11.01 atomic mass units). Which numerical setup can be used to determine the atomic mass of naturally occurring boron? ...
... A 100.00-gram sample of naturally occurring boron contains 19.78 grams of boron-10 (atomic mass = 10.01 atomic mass units) and 80.22 grams of boron-11 (atomic mass = 11.01 atomic mass units). Which numerical setup can be used to determine the atomic mass of naturally occurring boron? ...
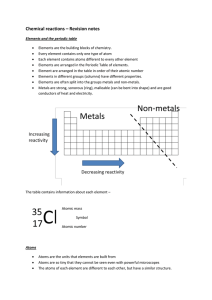
Chemical reactions revision
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
Name: Northwest Vista College Chem 1311
... 41. (6 pts) What mass of Na2SO4 is needed to prepare 350. mL of a solution having a sodium ion concentration of 0.125 M? NOTE that there are 2 Na+ ions for every 1 Na2SO4 ...
... 41. (6 pts) What mass of Na2SO4 is needed to prepare 350. mL of a solution having a sodium ion concentration of 0.125 M? NOTE that there are 2 Na+ ions for every 1 Na2SO4 ...
MSTA WOW Chemistry
... 1. Mix some water and food coloring in a plastic cup 2. If you lamp oil is green like the rubbing alcohol, then you need to add a different shade of food coloring to a small amount in a plastic cup. 3. Pour about 10 mL of Honey down the center of the graduated cylinder and try not to let it run down ...
... 1. Mix some water and food coloring in a plastic cup 2. If you lamp oil is green like the rubbing alcohol, then you need to add a different shade of food coloring to a small amount in a plastic cup. 3. Pour about 10 mL of Honey down the center of the graduated cylinder and try not to let it run down ...
makeup6
... If the concentration of ammonia gas is tripled, the value of the equilibrium constant will (A) triple (B) increase, buy by more than a factor of three (C) decrease to one-third its value (D) remain the same 24. The weak base ionization constant (Kb) for hydroxylamine, HONH2, is 1.1 x 10¯8. Which of ...
... If the concentration of ammonia gas is tripled, the value of the equilibrium constant will (A) triple (B) increase, buy by more than a factor of three (C) decrease to one-third its value (D) remain the same 24. The weak base ionization constant (Kb) for hydroxylamine, HONH2, is 1.1 x 10¯8. Which of ...
Al + Ag+ Al3+ + Ag
... last fall and "half" of a reaction cannot occur. Remind yourself that if the reaction above is read left to right it is called oxidation. Each metal will have a unique equilibrium with its ions that will result in a slight positive or negative charge build-up on the electrode. This tendency cannot b ...
... last fall and "half" of a reaction cannot occur. Remind yourself that if the reaction above is read left to right it is called oxidation. Each metal will have a unique equilibrium with its ions that will result in a slight positive or negative charge build-up on the electrode. This tendency cannot b ...
Biochemistry I (CHE 418 / 5418)
... • In the equation above, C is oxidized and oxygen is reduced. • Many antiseptics are oxidizing agents. See p. 152 ...
... • In the equation above, C is oxidized and oxygen is reduced. • Many antiseptics are oxidizing agents. See p. 152 ...
AP Chemistry Unit 3 Test Review Topics Covered: Gases Liquids
... 17) The best explanation for the lower pressure in container 4 is that SO2 molecules (A) have a larger average speed than the other three gases (B) occupy a larger portion of the container volume than the other three gases (C) have stronger intermolecular attractions than the other three gases (D) c ...
... 17) The best explanation for the lower pressure in container 4 is that SO2 molecules (A) have a larger average speed than the other three gases (B) occupy a larger portion of the container volume than the other three gases (C) have stronger intermolecular attractions than the other three gases (D) c ...
Chapter 4: Chemical Quantities and Aqueous Reactions
... • A titration may be based on any of the previously discussed types of reactions … A measured portion of acid solution is placed in the flask, and an indicator is added. Base solution of known concentration is slowly added from the buret. ...
... • A titration may be based on any of the previously discussed types of reactions … A measured portion of acid solution is placed in the flask, and an indicator is added. Base solution of known concentration is slowly added from the buret. ...
Review Sheet: Unit 6 Name__________________ CHEMISTRY: A
... ____________. ____________ are used to show the number of each ion in the formula. The ____________ system of nomenclature is used for compounds of metals that have more than one charge. Ions made up of more than one element are called ____________ ions, and the ending is NOT changed when naming the ...
... ____________. ____________ are used to show the number of each ion in the formula. The ____________ system of nomenclature is used for compounds of metals that have more than one charge. Ions made up of more than one element are called ____________ ions, and the ending is NOT changed when naming the ...
AS Paper 1 Practice Paper 12 - A
... Sodium hydroxide can be obtained as a monohydrate (NaOH.H2O). When heated, the water of crystallisation is lost, leaving anhydrous sodium hydroxide (NaOH). A chemist weighed a clean, dry crucible. The chemist transferred 1.10 g of NaOH.H2O to the crucible. The crucible and its contents were heated u ...
... Sodium hydroxide can be obtained as a monohydrate (NaOH.H2O). When heated, the water of crystallisation is lost, leaving anhydrous sodium hydroxide (NaOH). A chemist weighed a clean, dry crucible. The chemist transferred 1.10 g of NaOH.H2O to the crucible. The crucible and its contents were heated u ...
Review Part 2
... properties of the ingredients of solutions such as dissolving salt in water or adding lemon juice to water. • A solution is a special kind of mixture in which the particles in one ingredient spread out and mix evenly among the particles in the other ingredient. • For example, when sugar dissolves in ...
... properties of the ingredients of solutions such as dissolving salt in water or adding lemon juice to water. • A solution is a special kind of mixture in which the particles in one ingredient spread out and mix evenly among the particles in the other ingredient. • For example, when sugar dissolves in ...
Solutions, Acids, and Bases
... Solutions of acids and bases pH scale (page 247) A numerical scale developed to measure the relative strengths of acids and bases Ranges from 0 to 14 0 is the strongest acid 14 is the strongest base. ...
... Solutions of acids and bases pH scale (page 247) A numerical scale developed to measure the relative strengths of acids and bases Ranges from 0 to 14 0 is the strongest acid 14 is the strongest base. ...
South Pasadena • AP Chemistry
... obtained another solid and a gas, each of which was a pure substance. From this information which of the following statements is ALWAYS a correct conclusion? a) The original solid is not an element. b) Both products are elements. c) The original solid is a compound and the gas is an element. d) The ...
... obtained another solid and a gas, each of which was a pure substance. From this information which of the following statements is ALWAYS a correct conclusion? a) The original solid is not an element. b) Both products are elements. c) The original solid is a compound and the gas is an element. d) The ...
Sustainable Hydrogen and Electrical Energy Storage 5 Interested in
... - or the temperature is below the ‘inversion temperature’. The latter is a result of the increased distance between the interacting molecules → increased potential energy → reduced kinetic energy → reduced temperature. Hydrogen has a maximum inversion temperature of 205 K. For this reason pre-coolin ...
... - or the temperature is below the ‘inversion temperature’. The latter is a result of the increased distance between the interacting molecules → increased potential energy → reduced kinetic energy → reduced temperature. Hydrogen has a maximum inversion temperature of 205 K. For this reason pre-coolin ...
Unit 1: Matter and Energy HW Packet
... 5. __________ Exothermic Reaction e. releases heat to the surroundings 6. __________ Endothermic Reaction f. substances produced in a chemical reaction (on the right) 7. __________ Law of Conservation g. process in which one or more substances change into Of Mass new substances ...
... 5. __________ Exothermic Reaction e. releases heat to the surroundings 6. __________ Endothermic Reaction f. substances produced in a chemical reaction (on the right) 7. __________ Law of Conservation g. process in which one or more substances change into Of Mass new substances ...
Chem 30A Final Exam
... b. an acid – base neutralization c. an oxidation – reduction (redox) reaction not involving oxygen. ...
... b. an acid – base neutralization c. an oxidation – reduction (redox) reaction not involving oxygen. ...
AP Chemistry Unit 3 Test Review Topics Covered: Gases Liquids
... (D) XeF6 (E) XeF8 7) A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constant temperature? (A) The volume of the gas increase. (B) The pressure of the gas decreases. (C) The average speed of the gas molecules remains th ...
... (D) XeF6 (E) XeF8 7) A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constant temperature? (A) The volume of the gas increase. (B) The pressure of the gas decreases. (C) The average speed of the gas molecules remains th ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.