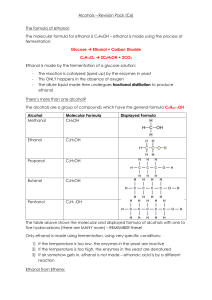
Precipitation Reactions
... The rules you just learned assume that the redox reaction is taking place under acidic conditions. (You are, after all, either producing or consuming H+ ions.) There are slightly different rules for basic conditions: 1. Balance the reaction (using your method of choice) as if it were under acidic co ...
... The rules you just learned assume that the redox reaction is taking place under acidic conditions. (You are, after all, either producing or consuming H+ ions.) There are slightly different rules for basic conditions: 1. Balance the reaction (using your method of choice) as if it were under acidic co ...
C6 Revision Guide - West Derby School
... If you combine these two equations, you get 2O3 + 3O2. STEP 3 – A possible termination reaction (that ends the depletion) is: Cl + Cl Cl2 It is very common for CFCs to last for between 20 and 50 years before they are completely broken down by UV radiation. For this reason, CFCs will continue to de ...
... If you combine these two equations, you get 2O3 + 3O2. STEP 3 – A possible termination reaction (that ends the depletion) is: Cl + Cl Cl2 It is very common for CFCs to last for between 20 and 50 years before they are completely broken down by UV radiation. For this reason, CFCs will continue to de ...
II. Acids and Bases
... equilibrium. 9. The weakest acids have the smallest Ka values because their solutions have the lowest concentrations of ions and the highest concentrations of unionized acid molecules. ...
... equilibrium. 9. The weakest acids have the smallest Ka values because their solutions have the lowest concentrations of ions and the highest concentrations of unionized acid molecules. ...
Ionic Compounds 1. What is the formula for aluminum phosphate
... 2. A 87.2-g sample of SrCl2 is dissolved in 112.5 mL of solution. Calculate the molarity of this solution. 3. How many grams of NaCl are contained in 350. mL of a 0.171 M solution of sodium chloride? 4. What mass of calcium chloride, CaCl2, is in 3.576 L of a 1.56 M solution? 5. Which of the followi ...
... 2. A 87.2-g sample of SrCl2 is dissolved in 112.5 mL of solution. Calculate the molarity of this solution. 3. How many grams of NaCl are contained in 350. mL of a 0.171 M solution of sodium chloride? 4. What mass of calcium chloride, CaCl2, is in 3.576 L of a 1.56 M solution? 5. Which of the followi ...
Chapter 23 (Section 3) Pregnancy, Birth, and Childhood (Pages 735
... *4. As the totally _________ water is heated by the surrounding ___ __ of the room, its temperature will _____ continue to ________, and as more ____________ is added the temperature of the water will rise _________ 0o C and continue rising until it reaches ___ __ causing __________________, a speci ...
... *4. As the totally _________ water is heated by the surrounding ___ __ of the room, its temperature will _____ continue to ________, and as more ____________ is added the temperature of the water will rise _________ 0o C and continue rising until it reaches ___ __ causing __________________, a speci ...
Atoms in Combination: The Chemical Bond
... regular pattern of alternating sodium and chloride ions. ...
... regular pattern of alternating sodium and chloride ions. ...
Chapter 12 Chemical Quantities
... Stoichiometry is the study of quantitative relationships between amounts of reactants used and products formed by a chemical reaction. ...
... Stoichiometry is the study of quantitative relationships between amounts of reactants used and products formed by a chemical reaction. ...
i principi di base - Structural Biology
... boiling temperature. The water forms ideal hydrogen bonds in the ice state where the water molecules are strongly ordered and a partial order is also maintained in solution. This implies that water molecules are partially ordered in the liquid phase. The radial distribution function of water is repo ...
... boiling temperature. The water forms ideal hydrogen bonds in the ice state where the water molecules are strongly ordered and a partial order is also maintained in solution. This implies that water molecules are partially ordered in the liquid phase. The radial distribution function of water is repo ...
Precipitation and Redox Reactions
... Early chemists saw “oxidation” reactions only as the combination of a material with oxygen to produce an oxide. For example, when gasoline burns in air, it oxidizes and forms oxides of carbon and hydrogen (oxides are compounds containing Oxygen, duh) 2 C8H18 + 25 O2 16 CO2 + 18 H2O ...
... Early chemists saw “oxidation” reactions only as the combination of a material with oxygen to produce an oxide. For example, when gasoline burns in air, it oxidizes and forms oxides of carbon and hydrogen (oxides are compounds containing Oxygen, duh) 2 C8H18 + 25 O2 16 CO2 + 18 H2O ...
Unit Two Objectives
... 2. Heat is the total energy of all of the particles in the sample; it is the form of energy that flows between two bodies when the bodies are at different temperatures; heat will flow from the hotter body to the cooler body. a. Endothermic Change: occurs when matter ABSORBS energy (in=endo) b. Exoth ...
... 2. Heat is the total energy of all of the particles in the sample; it is the form of energy that flows between two bodies when the bodies are at different temperatures; heat will flow from the hotter body to the cooler body. a. Endothermic Change: occurs when matter ABSORBS energy (in=endo) b. Exoth ...
Oxygen Reduction Reaction with the Rotating Ring Disk Electrode
... when compared with a literature value of 1.4 x 10–5 cm2 s–1. This property is sensitive to experimental parameters such as the concentration of O2 in the electrolyte, which is likely to be the most significant source of uncertainty in this measurement. Table 1. System variables for the oxygen reduct ...
... when compared with a literature value of 1.4 x 10–5 cm2 s–1. This property is sensitive to experimental parameters such as the concentration of O2 in the electrolyte, which is likely to be the most significant source of uncertainty in this measurement. Table 1. System variables for the oxygen reduct ...
File
... “Exothermic” means that heat is released during the reaction. This often results in the reaction container feeling warm to the touch (heat is given off). Reactants Products + HEAT (heat on product side because released) “Endothermic” means that heat is absorbed during the reaction. This often resu ...
... “Exothermic” means that heat is released during the reaction. This often results in the reaction container feeling warm to the touch (heat is given off). Reactants Products + HEAT (heat on product side because released) “Endothermic” means that heat is absorbed during the reaction. This often resu ...
Chapter 14…Kinetic Theory
... 7. Indicate the relationship between the variables in each of the equations above. 8. Indicate what each variable stands for in each of the equations above. 9. Temperature is in what unit for gas laws? 10. STP stands for: 11. Standard temperature = __________ 12. Standard pressure = __________ atm, ...
... 7. Indicate the relationship between the variables in each of the equations above. 8. Indicate what each variable stands for in each of the equations above. 9. Temperature is in what unit for gas laws? 10. STP stands for: 11. Standard temperature = __________ 12. Standard pressure = __________ atm, ...
PHYSICAL SETTING CHEMISTRY
... A separate answer sheet for Part A and Part B–1 has been provided to you. Follow the instructions from the proctor for completing the student information on your answer sheet. Record your answers to the Part A and Part B–1 multiple-choice questions on this separate answer sheet. Record your answers ...
... A separate answer sheet for Part A and Part B–1 has been provided to you. Follow the instructions from the proctor for completing the student information on your answer sheet. Record your answers to the Part A and Part B–1 multiple-choice questions on this separate answer sheet. Record your answers ...
Solution FRQs Practice
... (b) When table salt (NaCl) and sugar (C12H22O11) are dissolved in water, it is observed that (i) both solution have higher boiling points than pure water, and (ii) the boiling point of 0.10 M NaCl(aq) is higher than that of 0.10 M C12H22O11(aq). (c) Methane gas does not behave as an ideal gas at low ...
... (b) When table salt (NaCl) and sugar (C12H22O11) are dissolved in water, it is observed that (i) both solution have higher boiling points than pure water, and (ii) the boiling point of 0.10 M NaCl(aq) is higher than that of 0.10 M C12H22O11(aq). (c) Methane gas does not behave as an ideal gas at low ...
Word - chemmybear.com
... electrode (where K+ is attracted) and have it react with water to form H2 and OH-. K+ + e- K 2K° + 2H2O 2K+ + 2OH- + H2 The combination of these two reactions is exactly what happens when water is reduced at the cathode. 8. (Trick #2) When CuSO4(aq) is electrolyzed, you know that Cu° metal is go ...
... electrode (where K+ is attracted) and have it react with water to form H2 and OH-. K+ + e- K 2K° + 2H2O 2K+ + 2OH- + H2 The combination of these two reactions is exactly what happens when water is reduced at the cathode. 8. (Trick #2) When CuSO4(aq) is electrolyzed, you know that Cu° metal is go ...
File
... (a) A 0.7549 g sample of the compound burns in O2(g) to produce 1.9061 g of CO2(g) and 0.3370 g of H2O(g). (i) Calculate the individual masses of C, H, and O in the 0.7549 g sample. (ii) Determine the empirical formula for the compound. (b) A 0.5246 g sample of the compound was dissolved in 10.0012 ...
... (a) A 0.7549 g sample of the compound burns in O2(g) to produce 1.9061 g of CO2(g) and 0.3370 g of H2O(g). (i) Calculate the individual masses of C, H, and O in the 0.7549 g sample. (ii) Determine the empirical formula for the compound. (b) A 0.5246 g sample of the compound was dissolved in 10.0012 ...
Part a
... ◦ Common between dipoles such as water ◦ Also act as intramolecular bonds, holding a large molecule in a three-dimensional shape ...
... ◦ Common between dipoles such as water ◦ Also act as intramolecular bonds, holding a large molecule in a three-dimensional shape ...
2010 - SAASTA
... action of bacteria on organic matter. Natural gas is a gaseous fossil fuel also formed by the action of bacteria on organic matter. All fossil fuels can be burned with air or with oxygen derived from the air to provide heat. This heat may be employed directly, as in the case of a home furnace, or ut ...
... action of bacteria on organic matter. Natural gas is a gaseous fossil fuel also formed by the action of bacteria on organic matter. All fossil fuels can be burned with air or with oxygen derived from the air to provide heat. This heat may be employed directly, as in the case of a home furnace, or ut ...
Chapter 13…States of Matter
... 4. Calculate the amount of energy required to heat a 150 g chunk of aluminum from 20C to 40C. (Cp of aluminum = 0.220 cal/gC) H=mCpT (150g)(.22)(20) = 660 cal Chapters 17& 18…Reaction Rates & Equilibrium Define: 1. Equilibrium: the reaction occurs simultaneously in both directions. 2. Activate ...
... 4. Calculate the amount of energy required to heat a 150 g chunk of aluminum from 20C to 40C. (Cp of aluminum = 0.220 cal/gC) H=mCpT (150g)(.22)(20) = 660 cal Chapters 17& 18…Reaction Rates & Equilibrium Define: 1. Equilibrium: the reaction occurs simultaneously in both directions. 2. Activate ...
File
... The color change when NO is exposed to air The formation of steam from burning H2 and O2 The solidification of vegetable oil at low temperatures (E) The odor of NH3 when NH4Cl is rubbed together with Ca(OH)2 powder ...
... The color change when NO is exposed to air The formation of steam from burning H2 and O2 The solidification of vegetable oil at low temperatures (E) The odor of NH3 when NH4Cl is rubbed together with Ca(OH)2 powder ...
Acrobat - chemmybear.com
... 5 • Reactions in Aqueous Solution STUDY QUESTIONS AND PROBLEMS 1. Classify each of the following solutes as a strong electrolyte, weak electrolyte, or nonelectrolyte: sugar sodium hydroxide common salt (NaCl) hydrochloric acid alcohol copper sulfate acetic acid carbonic acid 2. Predict the solubilit ...
... 5 • Reactions in Aqueous Solution STUDY QUESTIONS AND PROBLEMS 1. Classify each of the following solutes as a strong electrolyte, weak electrolyte, or nonelectrolyte: sugar sodium hydroxide common salt (NaCl) hydrochloric acid alcohol copper sulfate acetic acid carbonic acid 2. Predict the solubilit ...
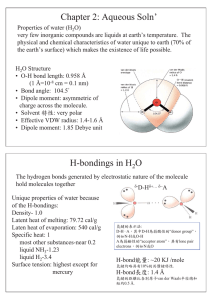
Chapter 2: Aqueous Soln` H
... Representation of the transport of CO2 from the tissues to the blood with delivery of O2 to the tissues. The opposite process occurs when O2 is taken up from the alveoli of the lungs and the CO2 is expelled. All of the processes of the transport of CO2 and O2 are not shown such as the formation and ...
... Representation of the transport of CO2 from the tissues to the blood with delivery of O2 to the tissues. The opposite process occurs when O2 is taken up from the alveoli of the lungs and the CO2 is expelled. All of the processes of the transport of CO2 and O2 are not shown such as the formation and ...
AQA Additional Sci C2 Revision Guide
... braces. It can also be used in spectacle frames because if they become bent by e.g. being sat on they can easily be reshaped by heating them. ...
... braces. It can also be used in spectacle frames because if they become bent by e.g. being sat on they can easily be reshaped by heating them. ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.