Active Learning Questions
... What type of solution forms when a nonmetal oxide dissolves in water? Give an example of such an oxide. What type of solution forms when a metal oxide dissolves in water? Give an example of such an oxide. ...
... What type of solution forms when a nonmetal oxide dissolves in water? Give an example of such an oxide. What type of solution forms when a metal oxide dissolves in water? Give an example of such an oxide. ...
Amines - ncert
... The lower aliphatic amines are gases with fishy odour. Primary amines with three or more carbon atoms are liquid and still higher ones are solid. Aniline and other arylamines are usually colourless but get coloured on storage due to atmospheric oxidation. Lower aliphatic amines are soluble in water ...
... The lower aliphatic amines are gases with fishy odour. Primary amines with three or more carbon atoms are liquid and still higher ones are solid. Aniline and other arylamines are usually colourless but get coloured on storage due to atmospheric oxidation. Lower aliphatic amines are soluble in water ...
Competing Intramolecular vs. Intermolecular Hydrogen Bonds in
... “The hydrogen bond is an attractive interaction between a hydrogen atom from a molecule or a molecular fragment X–H in which X is more electronegative than H, and an atom or a group of atoms in the same or a different molecule, in which there is evidence of bond formation. A typical hydrogen bond ma ...
... “The hydrogen bond is an attractive interaction between a hydrogen atom from a molecule or a molecular fragment X–H in which X is more electronegative than H, and an atom or a group of atoms in the same or a different molecule, in which there is evidence of bond formation. A typical hydrogen bond ma ...
U6B _13-14
... pH is always = 7 The titration curve graph shows the pH of the equivalence point. Take the vertical region and cut the length in half and then look to what pH value aligns to that point. ...
... pH is always = 7 The titration curve graph shows the pH of the equivalence point. Take the vertical region and cut the length in half and then look to what pH value aligns to that point. ...
Basic chemistry help is available here for high school or college
... In many high schools and colleges the basic chemistry course is the one that causes most concern among students. With everything going right, chemistry can be a fun but challenging course. Under poor conditions, your first chemistry course can be a real spittin’, cussin’ nightmare. The study of chem ...
... In many high schools and colleges the basic chemistry course is the one that causes most concern among students. With everything going right, chemistry can be a fun but challenging course. Under poor conditions, your first chemistry course can be a real spittin’, cussin’ nightmare. The study of chem ...
how do water molecules probe and control
... as well as on water at the same temperature. The isotopic substitution was applied to the water hydrogen atoms. Diffraction data were analyzed by independent MD simulations and using the empirical potential structure refinement method (EPSR). Both approaches revealed the presence of aggregates of st ...
... as well as on water at the same temperature. The isotopic substitution was applied to the water hydrogen atoms. Diffraction data were analyzed by independent MD simulations and using the empirical potential structure refinement method (EPSR). Both approaches revealed the presence of aggregates of st ...
No Slide Title
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
Chapter 5: Calculations and the Chemical Equation
... 2. The products which are formed by the reaction. 3. The amounts (moles) of each substance used and each substance produced. The Numbers in a Chemical Equation: 1. Subscripts: The small numbers to the lower right of chemical symbols. Subscripts represent the number of atoms of each element in the mo ...
... 2. The products which are formed by the reaction. 3. The amounts (moles) of each substance used and each substance produced. The Numbers in a Chemical Equation: 1. Subscripts: The small numbers to the lower right of chemical symbols. Subscripts represent the number of atoms of each element in the mo ...
Organic Chemistry Organic Chemistry
... evaporate easily. In fact, they are often gases at room temperature. (b) Polar substances, with strong forces of attraction among the molecules, require considerable energy to evaporate. ...
... evaporate easily. In fact, they are often gases at room temperature. (b) Polar substances, with strong forces of attraction among the molecules, require considerable energy to evaporate. ...
Physical and Chemical equilibrium
... (iv)The value of the equilibrium constant of a particular reaction is always constant depending upon the temperature of the reaction and is independent of the direction from which the equilibrium is attained (v)Equilibrium constant is not changed by the inert material added to the system (v) The val ...
... (iv)The value of the equilibrium constant of a particular reaction is always constant depending upon the temperature of the reaction and is independent of the direction from which the equilibrium is attained (v)Equilibrium constant is not changed by the inert material added to the system (v) The val ...
EIT Review S2012 Part 2 Dr. J. Mack CSUS Department of Chemistry
... The rate of effusion of a gas is proportional to its uRMS. over time, the mixture becomes homogeneous. ...
... The rate of effusion of a gas is proportional to its uRMS. over time, the mixture becomes homogeneous. ...
GCSE Chemistry Specification Specification for exams from 2014 2014
... Hypotheses can then be used to make predictions that can be tested. Data from testing a prediction can support or refute the hypothesis or lead to a new hypothesis. If the hypotheses and models we have available to us do not completely match our data or observations, we need to check the validity of ...
... Hypotheses can then be used to make predictions that can be tested. Data from testing a prediction can support or refute the hypothesis or lead to a new hypothesis. If the hypotheses and models we have available to us do not completely match our data or observations, we need to check the validity of ...
Document
... Complex ion, or coordination compound, is the name given to any metal atom or ion that is bonded to more than one atom or molecule. Some of the most interesting ions have a metal ion surrounded by a number of ligands. Ligands are molecules, such as ammonia, NH3, or anions, such as cyanide, CN −, tha ...
... Complex ion, or coordination compound, is the name given to any metal atom or ion that is bonded to more than one atom or molecule. Some of the most interesting ions have a metal ion surrounded by a number of ligands. Ligands are molecules, such as ammonia, NH3, or anions, such as cyanide, CN −, tha ...
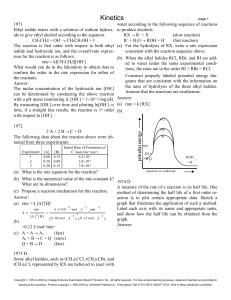
Kinetics
... The following data give the value of the rate constant at various temperatures for the gas phase reaction above. 1981 B T (K) k (litre/mol sec) A(aq) + 2 B(aq) 3 C(aq) + D(aq) ...
... The following data give the value of the rate constant at various temperatures for the gas phase reaction above. 1981 B T (K) k (litre/mol sec) A(aq) + 2 B(aq) 3 C(aq) + D(aq) ...
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical
... substance by 1 degree Celsius. Water has a specific heat capacity of about 4.2 J/g•K, meaning that 1 gram of water at 15 degrees C, to which 4.2 J of energy is added, will have a temperature of 16 degrees C. (or 14 degrees C—if 4.2 J of energy is removed). (d) State function—Any parameter which is d ...
... substance by 1 degree Celsius. Water has a specific heat capacity of about 4.2 J/g•K, meaning that 1 gram of water at 15 degrees C, to which 4.2 J of energy is added, will have a temperature of 16 degrees C. (or 14 degrees C—if 4.2 J of energy is removed). (d) State function—Any parameter which is d ...
Continued on Next page
... ∆H = −53.4 kJ/mol volume and concentration of the reactants: 55.0 mL, 1.30 mol/L density of both solutions: 1.00 g/mL c = 4.184 J/g·˚C Ti = 21.4˚C Plan Your Strategy Step 1 Determine the total volume by adding the volumes of the two solutions. Determine the mass of the reaction mixture using the giv ...
... ∆H = −53.4 kJ/mol volume and concentration of the reactants: 55.0 mL, 1.30 mol/L density of both solutions: 1.00 g/mL c = 4.184 J/g·˚C Ti = 21.4˚C Plan Your Strategy Step 1 Determine the total volume by adding the volumes of the two solutions. Determine the mass of the reaction mixture using the giv ...
Ni recovery using KOH, NaOH, and NH4OH in the presence of
... very different horizons – the Merensky Reef and the Upper Group 2 (UG2) chromitite, which together can be traced on surface for 300 km in two separate arcs, and the Platreef, which extends for over 30 km (Cawthorn, 1999). Throughout the BC, PGMs are strongly associated with the sulphide minerals cha ...
... very different horizons – the Merensky Reef and the Upper Group 2 (UG2) chromitite, which together can be traced on surface for 300 km in two separate arcs, and the Platreef, which extends for over 30 km (Cawthorn, 1999). Throughout the BC, PGMs are strongly associated with the sulphide minerals cha ...
Chemistry (Revised)
... (b) Chloromethane can be produced by the reaction of methane with chlorine. CH4(g) + Cl2(g) → CH3Cl(g) + HCl(g) Using bond enthalpies from the data booklet, calculate the enthalpy change, in kJ mol−1, for this reaction. ...
... (b) Chloromethane can be produced by the reaction of methane with chlorine. CH4(g) + Cl2(g) → CH3Cl(g) + HCl(g) Using bond enthalpies from the data booklet, calculate the enthalpy change, in kJ mol−1, for this reaction. ...
Information on measuring ammonia in water
... - in the food sector, e. g. for wine and beer, - in laboratories. The measurement can be made directly in the liquid medium. This means that it is not necessary to carry out time-consuming and complicated sample preparation. Colored or cloudy samples can also be measured. If the pH value is less tha ...
... - in the food sector, e. g. for wine and beer, - in laboratories. The measurement can be made directly in the liquid medium. This means that it is not necessary to carry out time-consuming and complicated sample preparation. Colored or cloudy samples can also be measured. If the pH value is less tha ...
Chemical Quantities
... the amounts of reactants needed to give a certain amount of product or to predict how much product we can make from a given quantity of reactants. To illustrate how this idea works with a chemistry example, consider the reaction between gaseous carbon monoxide and hydrogen to produce liquid methanol ...
... the amounts of reactants needed to give a certain amount of product or to predict how much product we can make from a given quantity of reactants. To illustrate how this idea works with a chemistry example, consider the reaction between gaseous carbon monoxide and hydrogen to produce liquid methanol ...
The Mole
... Finding the Amount of Excess By calculating the amount of the excess reactant needed to completely react with the limiting reactant, we can subtract that amount from the given amount to find the amount of excess. Can we find the amount of excess potassium in the previous problem? ...
... Finding the Amount of Excess By calculating the amount of the excess reactant needed to completely react with the limiting reactant, we can subtract that amount from the given amount to find the amount of excess. Can we find the amount of excess potassium in the previous problem? ...
Chapter 3: Stoichiometry
... Decomposition Reactions A single reactant breaks into two or more products ...
... Decomposition Reactions A single reactant breaks into two or more products ...
1. Blood cholesterol levels are generally expressed as milligrams of
... ANSWER: Solution :Lets assume we have 1 mole of the each gas in the ballon at the STP conditions at STP condition 1 mole gas = 22.4 L so both balloons will have volume 22.4 L but the denities of the gases are different because mass of 1 mol N2 = 28.014 g per mol and molar mass of He = 4.0026 g per m ...
... ANSWER: Solution :Lets assume we have 1 mole of the each gas in the ballon at the STP conditions at STP condition 1 mole gas = 22.4 L so both balloons will have volume 22.4 L but the denities of the gases are different because mass of 1 mol N2 = 28.014 g per mol and molar mass of He = 4.0026 g per m ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.