Chemistry 121: Topic 2 - From Atoms to Stoichiometry Topic 2
... ¾ HCl, Hydrogen Chloride as a molecule becomes hydrochloric acid Oxyacids are acids that contain Hydrogen, oxygen and another element (which is the central element. To write the molecular formula, H first, then central element, then Oxygen ie., HNO3 → Nitric Acid; H2CO3 → Carbonic Acid; H2SO4 → Sulp ...
... ¾ HCl, Hydrogen Chloride as a molecule becomes hydrochloric acid Oxyacids are acids that contain Hydrogen, oxygen and another element (which is the central element. To write the molecular formula, H first, then central element, then Oxygen ie., HNO3 → Nitric Acid; H2CO3 → Carbonic Acid; H2SO4 → Sulp ...
sch103manual - university of nairobi staff profiles
... The above equation is the mathematical expression of Avogadro’s Law, which states that at constant pressure and temperature, the volume of a gas is directly proportional to the number of moles of the gas present. One mole of any gas contains the same number of molecules ( Avogadro’s number = 6.022 ...
... The above equation is the mathematical expression of Avogadro’s Law, which states that at constant pressure and temperature, the volume of a gas is directly proportional to the number of moles of the gas present. One mole of any gas contains the same number of molecules ( Avogadro’s number = 6.022 ...
Part II - American Chemical Society
... d. No. There is so little N2O4 (about 10 parts per billion of the total NO2!) that even if it all dissociates, it will have a negligible effect on the [NO2], which will remain 30 ppb (all the gases in the atmosphere change volume with temperature to approximately the same extent). e. While the decom ...
... d. No. There is so little N2O4 (about 10 parts per billion of the total NO2!) that even if it all dissociates, it will have a negligible effect on the [NO2], which will remain 30 ppb (all the gases in the atmosphere change volume with temperature to approximately the same extent). e. While the decom ...
Chemistry 11 Final Examination Review
... a) Electrons can absorb or emit energy only in whole numbers of photons. b) Atoms have a central positively charged nucleus. c) Electrons move around the nucleus as planets orbit the sun. d) Most of the volume of an atom is empty space. 10. Which of the following orbitals is spherical in shape? a) 3 ...
... a) Electrons can absorb or emit energy only in whole numbers of photons. b) Atoms have a central positively charged nucleus. c) Electrons move around the nucleus as planets orbit the sun. d) Most of the volume of an atom is empty space. 10. Which of the following orbitals is spherical in shape? a) 3 ...
mass
... 6.022X1023 methane molecules and 6.022X1023 water molecules 6.022X1023 to 6.022X1023 is also a 1 to 1 ratio between CH4 and H2O Realizing that 6.022X1023 molecules is also 1 mole leads to the idea that 1 mole methane to 1 mole water is also a 1 to 1 ratio. 0.5 mole to 0.5 mole is also a 1 to 1 ratio ...
... 6.022X1023 methane molecules and 6.022X1023 water molecules 6.022X1023 to 6.022X1023 is also a 1 to 1 ratio between CH4 and H2O Realizing that 6.022X1023 molecules is also 1 mole leads to the idea that 1 mole methane to 1 mole water is also a 1 to 1 ratio. 0.5 mole to 0.5 mole is also a 1 to 1 ratio ...
1.09 MB / 64 pages
... network of hydrogen bonds. In Chapter 1, we found that there are, on the average, fewer than four H bonds per water molecule in liquid water. The three H bonds formed by ethanol fits right into this structure and the rather small ethyl group, –CH2CH3, probably does not perturb the structure very muc ...
... network of hydrogen bonds. In Chapter 1, we found that there are, on the average, fewer than four H bonds per water molecule in liquid water. The three H bonds formed by ethanol fits right into this structure and the rather small ethyl group, –CH2CH3, probably does not perturb the structure very muc ...
BARIUM NITRATE
... Barium sulfate has many commercial applications. It is used as natural barite, or precipitated BaSO4. The precipitated salt in combination with equimolar amount of co-precipitated zinc sulfide formerly was used as a white protective coating pigment, known as lithophone. Similarly, in combination wit ...
... Barium sulfate has many commercial applications. It is used as natural barite, or precipitated BaSO4. The precipitated salt in combination with equimolar amount of co-precipitated zinc sulfide formerly was used as a white protective coating pigment, known as lithophone. Similarly, in combination wit ...
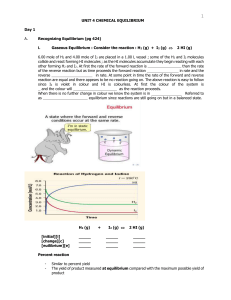
Unit 4 - Chemical Equilibrium
... Recognizing Equilibrium (pg 424) Gaseous Equilibrium : Consider the reaction : H2 (g) + I2 (g) ...
... Recognizing Equilibrium (pg 424) Gaseous Equilibrium : Consider the reaction : H2 (g) + I2 (g) ...
Industrial Zinc Plating Processes
... The metal deposit distribution is affected by the variation of the cathode efficiency with current density. In plating solutions in which the cathode efficiencies decrease rapidly as current density increases, excess deposits will plate on edges and corners. This phenomenon is coined throwing power. ...
... The metal deposit distribution is affected by the variation of the cathode efficiency with current density. In plating solutions in which the cathode efficiencies decrease rapidly as current density increases, excess deposits will plate on edges and corners. This phenomenon is coined throwing power. ...
Massachusetts Tests for Educator Licensure (MTEL )
... Correct Response: D. The combination of chemicals is that of a weak acid and a strong base. This conclusion can be drawn because the equivalence point on the graph corresponds to a pH greater than 7. It is clear that a weak acid is being titrated with a strong base (instead of a strong base being ti ...
... Correct Response: D. The combination of chemicals is that of a weak acid and a strong base. This conclusion can be drawn because the equivalence point on the graph corresponds to a pH greater than 7. It is clear that a weak acid is being titrated with a strong base (instead of a strong base being ti ...
Chapter 1 – Reaction Kinetics Answer Key
... 3. The concentrations of pure solids and liquids are fixed. That is they do not change (appreciably for the liquid if it is the solvent and at all for the solid) during a chemical reaction. ...
... 3. The concentrations of pure solids and liquids are fixed. That is they do not change (appreciably for the liquid if it is the solvent and at all for the solid) during a chemical reaction. ...
Presentation 3
... This is almost the case when either pH = 6.4 or pH = 10.3 because at either pH, {106.4 10-pH + 1 + 10-10.3 10pH} is only slightly greater than 2, e.g., ...
... This is almost the case when either pH = 6.4 or pH = 10.3 because at either pH, {106.4 10-pH + 1 + 10-10.3 10pH} is only slightly greater than 2, e.g., ...
2003 AP Chemistry Form B Scoring Guidelines - AP Central
... (d) On the graph above, make a sketch that shows how the concentration of H2(g) changes as a function of time. From the graph, [H2]eq is 0.10 M The curve should have the following characteristics: - start at 0 M; - increase to 0.1 M; - reach equilibrium at the same time [HI] reaches equilibrium ...
... (d) On the graph above, make a sketch that shows how the concentration of H2(g) changes as a function of time. From the graph, [H2]eq is 0.10 M The curve should have the following characteristics: - start at 0 M; - increase to 0.1 M; - reach equilibrium at the same time [HI] reaches equilibrium ...
Preparatory Problems of the 40th IChO - IChO-2016
... Find the equation which gives the dependence of the density on y, assuming that only the iron content varies. Find a similar equation for the case when only the S ...
... Find the equation which gives the dependence of the density on y, assuming that only the iron content varies. Find a similar equation for the case when only the S ...
reaction rate - davis.k12.ut.us
... • Key factors that influence the rate of chemical reactions include reactivity, concentration, surface area, temperature, and catalysts. • Raising the temperature of a reaction generally increases the rate of the reaction by increasing the collision frequency and the number of collisions that ...
... • Key factors that influence the rate of chemical reactions include reactivity, concentration, surface area, temperature, and catalysts. • Raising the temperature of a reaction generally increases the rate of the reaction by increasing the collision frequency and the number of collisions that ...
ch17
... If equilibrium quantities are given, we simply substitute these into the expression for Kc to calculate its value. If only some equilibrium quantities are given, we use a reaction table to calculate them and find Kc. ...
... If equilibrium quantities are given, we simply substitute these into the expression for Kc to calculate its value. If only some equilibrium quantities are given, we use a reaction table to calculate them and find Kc. ...
topic: chemical formula, chemical equations and stoichiometry
... and the relations between molar mass, molar volume 3. State the formula or relationship. and Avogadro constant, NA 4. Use information given in 2. Analysis the numerical problem. formula to solve problem. 5. Checking the answer ...
... and the relations between molar mass, molar volume 3. State the formula or relationship. and Avogadro constant, NA 4. Use information given in 2. Analysis the numerical problem. formula to solve problem. 5. Checking the answer ...
1. A pharmacy analyst supervises the state of a refractometer. For its
... sulfuric acid: at dissolving of substance in sulphuric acid and following addition of water a dark-red colour with a greenish-brown fluorescence develops, On adding of chloroform an lower layer becomes yellow and upper layer is coloured into green. 29. An analytical laboratory received a sample of a ...
... sulfuric acid: at dissolving of substance in sulphuric acid and following addition of water a dark-red colour with a greenish-brown fluorescence develops, On adding of chloroform an lower layer becomes yellow and upper layer is coloured into green. 29. An analytical laboratory received a sample of a ...
Specification and sample assessment material - Edexcel
... colleges. It is part of a suite of International GCSEs offered by Edexcel. The course gives students the opportunity to experience chemistry within the context of their general education. The course design provides a basis for progression to further study in GCE Advanced Subsidiary and Advanced Leve ...
... colleges. It is part of a suite of International GCSEs offered by Edexcel. The course gives students the opportunity to experience chemistry within the context of their general education. The course design provides a basis for progression to further study in GCE Advanced Subsidiary and Advanced Leve ...
Proton Transfers at the Air
... 1. How do anions mediate proton transfers at the air-water interface? 2. What is the basis for the negative surface potential at the air-water interface? 3. What is the mechanism for catalysis ‘on-water’? In addition to our experiments with the ESIMS, we applied quantum mechanics and molecular dynam ...
... 1. How do anions mediate proton transfers at the air-water interface? 2. What is the basis for the negative surface potential at the air-water interface? 3. What is the mechanism for catalysis ‘on-water’? In addition to our experiments with the ESIMS, we applied quantum mechanics and molecular dynam ...
unit iv – stoichiometry 1
... * Chemical Formulas - a series of symbols and numbers used to represent the composition of an element or a compound I. Formulas for Compounds A. Ionic Compounds - represents ratio of cations to anions * remember, ionic compounds don’t form molecules as we think of them * we call “molecules” formula ...
... * Chemical Formulas - a series of symbols and numbers used to represent the composition of an element or a compound I. Formulas for Compounds A. Ionic Compounds - represents ratio of cations to anions * remember, ionic compounds don’t form molecules as we think of them * we call “molecules” formula ...
Chem Course Desc2. New
... to the gasoline you put in your cars. All of these products share in common that a chemist has manipulated all their molecules to make your life more comfortable. Chemistry is needed to understand many processes in a variety of industries: pharmaceuticals, cosmetics, petroleum, plastics, food techno ...
... to the gasoline you put in your cars. All of these products share in common that a chemist has manipulated all their molecules to make your life more comfortable. Chemistry is needed to understand many processes in a variety of industries: pharmaceuticals, cosmetics, petroleum, plastics, food techno ...
Ammonia destruction in the reaction furnace
... only requires that one-third of the total H2S be oxidised to SO2, which leaves the remaining two-thirds to act as a heat sink in the flame. Bypassing clean AAG around the front zone burner flame increases the flame temperature by reducing the amount of non-combusting gas in the flame that is acting ...
... only requires that one-third of the total H2S be oxidised to SO2, which leaves the remaining two-thirds to act as a heat sink in the flame. Bypassing clean AAG around the front zone burner flame increases the flame temperature by reducing the amount of non-combusting gas in the flame that is acting ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.