enthalpy changes
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
No Slide Title
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
DELTAHPP
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
Empirical Formula, Molecular Formula, Percent Composition
... Compare both of your reactant amounts to the same product in this case Al2(SO4)3. Then find out how much products will be produced from each individual reactant. Whichever reactant yields the least amount of product that is your limiting reactant. 4 moles Al x 1 mole Al2(SO4)3 / 2 moles Al= 2 moles ...
... Compare both of your reactant amounts to the same product in this case Al2(SO4)3. Then find out how much products will be produced from each individual reactant. Whichever reactant yields the least amount of product that is your limiting reactant. 4 moles Al x 1 mole Al2(SO4)3 / 2 moles Al= 2 moles ...
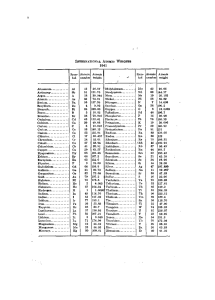
INTEKNATIONAL ATOMIC WEIGHTS Aluminum... Antimony..., Argon
... of the student. When an experiment is too short, the student will find interesting things to do under "Suggestions for Further Work"; when it is too long, the instructor will select parts of the "Procedure" which may be omitted. Several of the procedures have been expanded and the directions made mo ...
... of the student. When an experiment is too short, the student will find interesting things to do under "Suggestions for Further Work"; when it is too long, the instructor will select parts of the "Procedure" which may be omitted. Several of the procedures have been expanded and the directions made mo ...
Unit 2: Matter as Solutions and Gases
... 1. All Na+, NH4+, NO3−, ClO3−, ClO4− and CH3COO− salts are soluble (except RbClO4, CsClO4, AgCH3COO, Hg2(CH3COO)2). 2. Most F− are soluble (except with Li+, Mg2+, Ca2+, Sr2+, Ba2+ and Fe2+ Hg22+ and Pb2+). 3. Most Cl−, Br−, and I− salts are soluble (except with Cu+, Ag+, Hg22+, Hg2+, and Pb2+). 4. M ...
... 1. All Na+, NH4+, NO3−, ClO3−, ClO4− and CH3COO− salts are soluble (except RbClO4, CsClO4, AgCH3COO, Hg2(CH3COO)2). 2. Most F− are soluble (except with Li+, Mg2+, Ca2+, Sr2+, Ba2+ and Fe2+ Hg22+ and Pb2+). 3. Most Cl−, Br−, and I− salts are soluble (except with Cu+, Ag+, Hg22+, Hg2+, and Pb2+). 4. M ...
Recycling and Chemical Mathematics
... “Better Things for Better Living Through Chemistry” was the slogan of a large chemical company not too long ago. The slogan rings true when we look at the myriad of products that have been made possible by people exploiting the chemical possibilities of their environment. Modern medicines, fertilize ...
... “Better Things for Better Living Through Chemistry” was the slogan of a large chemical company not too long ago. The slogan rings true when we look at the myriad of products that have been made possible by people exploiting the chemical possibilities of their environment. Modern medicines, fertilize ...
chemistry - Textbooks Online
... drinking of which would endue the alchemist with immortality), and the search for the philosopher’s stone, which would turn base metals into gold. Improbable as these ideas might seem today, the alchemists continued their quests for around 2000 years and achieved some remarkable successes, even if t ...
... drinking of which would endue the alchemist with immortality), and the search for the philosopher’s stone, which would turn base metals into gold. Improbable as these ideas might seem today, the alchemists continued their quests for around 2000 years and achieved some remarkable successes, even if t ...
Chemistry I - State College Area School District
... 1. Clean the outside of a large ignition tube with alcohol and let dry. 2. Obtain a metric ruler and glass marking pencil. Starting from the open-end of the tube mark every centimeter on the outside of the tube with the marking pencil. 3. Fill a 400 mL beaker about ¾ full with tap water. 4. Obtain a ...
... 1. Clean the outside of a large ignition tube with alcohol and let dry. 2. Obtain a metric ruler and glass marking pencil. Starting from the open-end of the tube mark every centimeter on the outside of the tube with the marking pencil. 3. Fill a 400 mL beaker about ¾ full with tap water. 4. Obtain a ...
(MDCAT) 2017 - University Of Health Sciences Lahore
... 2. STATES OF MATTER: In this topic, student should be able to: a) Understand gaseous state with reference to: i) Postulates of kinetic molecular theory ii) Gas laws: Boyle’s law, Charles’ law, Avogadro’s law and gas equation (PV=nRT) and calculations involving gas laws. iii) Deviation of real gases ...
... 2. STATES OF MATTER: In this topic, student should be able to: a) Understand gaseous state with reference to: i) Postulates of kinetic molecular theory ii) Gas laws: Boyle’s law, Charles’ law, Avogadro’s law and gas equation (PV=nRT) and calculations involving gas laws. iii) Deviation of real gases ...
Chm 2
... b. the mass of the products is greater than the mass of reactants. c. the number of atoms in the reactants and products must change. d. energy as heat must be added to the reactants. 2. Which observation does not indicate that a chemical reaction has occurred? a. formation of a precipitate c. evolut ...
... b. the mass of the products is greater than the mass of reactants. c. the number of atoms in the reactants and products must change. d. energy as heat must be added to the reactants. 2. Which observation does not indicate that a chemical reaction has occurred? a. formation of a precipitate c. evolut ...
deltahpps
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
... you get one carbon dioxide molecule for every carbon atom in the original and one water molecule for every two hydrogen atoms When you have done this, go back and balance the oxygen. ...
Worked solutions to the problems
... we know that you will employ best-practice to responsibly dispose or recycle the materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these ...
... we know that you will employ best-practice to responsibly dispose or recycle the materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these ...
Experimental Study of Closed System in the Chlorine Dioxide
... citric acid solution. Sulfuric acid: 0.05 mol/L. All other chemicals were the highest purity commercially available and were used as received. 2.2. Methods. The reaction was started by injecting a small volume of one of the reactants into a mixture containing the other components in a spectrophotome ...
... citric acid solution. Sulfuric acid: 0.05 mol/L. All other chemicals were the highest purity commercially available and were used as received. 2.2. Methods. The reaction was started by injecting a small volume of one of the reactants into a mixture containing the other components in a spectrophotome ...
as a PDF
... of a square. Four trichloroacetato bridging anions are alternatively above and below this square. The terminal oxygen atoms of the four tert-butyl peroxidic anions are located in the plane of the four palladium atoms and bridge these atoms two by two. The average Pd-Pd separation is 2.91 A. The mean ...
... of a square. Four trichloroacetato bridging anions are alternatively above and below this square. The terminal oxygen atoms of the four tert-butyl peroxidic anions are located in the plane of the four palladium atoms and bridge these atoms two by two. The average Pd-Pd separation is 2.91 A. The mean ...
W. M. White Geochemistry Chapter 9: Stable Isotopes Chapter 9
... The origins of stablethisotope geochemistry are closely tied to the development of modern physics in the first half of the 20 century. The discovery of the neutron in 1932 by H. Urey and the demonstration of variations in the isotopic composition of light elements by A. Nier in the 1930’s and 1940’s ...
... The origins of stablethisotope geochemistry are closely tied to the development of modern physics in the first half of the 20 century. The discovery of the neutron in 1932 by H. Urey and the demonstration of variations in the isotopic composition of light elements by A. Nier in the 1930’s and 1940’s ...
Title
... NMR spectrum is indistinguishable from the starting complex 1Cl2, confirming that the cationic complex 1 remains unaltered. The crystal structure obtained for 1(PF6)2 confirms the cisdisposition of the ligands (Fig. 7). The trans-isomer complex 2(CF3SO3)2 was synthesized (Scheme 2) by reaction of tr ...
... NMR spectrum is indistinguishable from the starting complex 1Cl2, confirming that the cationic complex 1 remains unaltered. The crystal structure obtained for 1(PF6)2 confirms the cisdisposition of the ligands (Fig. 7). The trans-isomer complex 2(CF3SO3)2 was synthesized (Scheme 2) by reaction of tr ...
caribbeanexaminations council report on candidates` work in
... Candidates were expected to complete the data required in Table 1 to show the titration values based on a series of burette readings given in Figure 1. They were also required to carry out a series of simple calculations, based on the titration readings and other given information to determine the m ...
... Candidates were expected to complete the data required in Table 1 to show the titration values based on a series of burette readings given in Figure 1. They were also required to carry out a series of simple calculations, based on the titration readings and other given information to determine the m ...
mole - hrsbstaff.ednet.ns.ca
... This reaction requires two molecules of hydrogen and one molecule of oxygen. To carry out this chemical reaction a chemist must mix hydrogen and oxygen together in the correct ratio (2:1). How does the chemist know when the correct ratio is present? These molecules are too small to see so we cannot ...
... This reaction requires two molecules of hydrogen and one molecule of oxygen. To carry out this chemical reaction a chemist must mix hydrogen and oxygen together in the correct ratio (2:1). How does the chemist know when the correct ratio is present? These molecules are too small to see so we cannot ...
Experiment 7: Determination of the concentration of a solution of an
... Density, d, is defined as the ratio of the two properties, mass, m, and volume, V, i.e. ...
... Density, d, is defined as the ratio of the two properties, mass, m, and volume, V, i.e. ...
Introduction to Inorganic Chemistry
... section (2.1). Of the remainder, the majority occur in relatively concentrated deposits, from which they or their compounds can be extracted fairly easily (e.g. hydrocarbons from petroleum). For copper, lead, zinc, nickel, and tin, demand is such that less concentrated deposits have to be worked, do ...
... section (2.1). Of the remainder, the majority occur in relatively concentrated deposits, from which they or their compounds can be extracted fairly easily (e.g. hydrocarbons from petroleum). For copper, lead, zinc, nickel, and tin, demand is such that less concentrated deposits have to be worked, do ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.