Chemistry Basics - Mr. Grays Physical Science Class
... terms of their properties. Properties can be divided into the categories of physical properties and chemical properties. Physical properties are readily observable, like; color, size, luster, or smell. Chemical properties are only observable during a chemical reaction. For example, you might not kno ...
... terms of their properties. Properties can be divided into the categories of physical properties and chemical properties. Physical properties are readily observable, like; color, size, luster, or smell. Chemical properties are only observable during a chemical reaction. For example, you might not kno ...
View/Open
... enthalpy. Therefore the amount of heat change during a reaction at constant temperature and pressure may also be called enthalpy change. Its value depends upon the number of moles of the reactants which have reacted in the given chemical reaction. Thus, Heat of reaction may be defined as the amount ...
... enthalpy. Therefore the amount of heat change during a reaction at constant temperature and pressure may also be called enthalpy change. Its value depends upon the number of moles of the reactants which have reacted in the given chemical reaction. Thus, Heat of reaction may be defined as the amount ...
chemistry - Brilliant Public School Sitamarhi
... in one unit cell of this mineral there are 4 Ca2+ ions and 8F– ions and that Ca2+ ions are arranged in a fcc lattice. The F– ions fill all the tetrahedral holes in the fcc lattice of Ca2+ ions. The edge of the unit cell is 5.46 × 10–8 cm in length. The density of the solid is 3.18 g cm–3. Use this i ...
... in one unit cell of this mineral there are 4 Ca2+ ions and 8F– ions and that Ca2+ ions are arranged in a fcc lattice. The F– ions fill all the tetrahedral holes in the fcc lattice of Ca2+ ions. The edge of the unit cell is 5.46 × 10–8 cm in length. The density of the solid is 3.18 g cm–3. Use this i ...
Energy Foundations for High School Chemistry
... Preparing to Investigate What is energy? Most of us have a feeling that we understand energy and recognize it when we see it, but coming up with a formal definition might be harder for us to do. Here are some of the basic concepts associated with a definition of energy: • Energy is required to mak ...
... Preparing to Investigate What is energy? Most of us have a feeling that we understand energy and recognize it when we see it, but coming up with a formal definition might be harder for us to do. Here are some of the basic concepts associated with a definition of energy: • Energy is required to mak ...
AS/A level
... A buffer solution was made by adding 19.6 g of sodium 3-chloropropanoate, CH2ClCH2COONa, to 1 dm3 of 0.100 mol dm‒3 3-chloropropanoic acid, CH2ClCH2COOH, at 298 K. (Ka for 3-chloropropanoic acid = 7.94 × 10‒5 mol dm‒3 at 298 K) Calculate the pH of the buffer solution at 298 K. ...
... A buffer solution was made by adding 19.6 g of sodium 3-chloropropanoate, CH2ClCH2COONa, to 1 dm3 of 0.100 mol dm‒3 3-chloropropanoic acid, CH2ClCH2COOH, at 298 K. (Ka for 3-chloropropanoic acid = 7.94 × 10‒5 mol dm‒3 at 298 K) Calculate the pH of the buffer solution at 298 K. ...
Gas-Phase Reactions of Fe (CH2O)+ and Fe (CH2S)+ with Small
... bonding differences between Fe(CH2O)+ and Fe(CH2S)+. To do this, product ion structures were probed by collision-induced dissociation, specific ion-molecule reactions, and use of labeled compounds, and experimental bond energies were obtained by using ion-molecule bracketing and competitive collisio ...
... bonding differences between Fe(CH2O)+ and Fe(CH2S)+. To do this, product ion structures were probed by collision-induced dissociation, specific ion-molecule reactions, and use of labeled compounds, and experimental bond energies were obtained by using ion-molecule bracketing and competitive collisio ...
2 - Montville.net
... The mole enables chemists to move from the microscopic world of atoms and molecules to the real world of grams . Stoichiometry problems are classified between the information given in the problem and the information you are expected to find, the unknown. The given and the unknown may be expressed i ...
... The mole enables chemists to move from the microscopic world of atoms and molecules to the real world of grams . Stoichiometry problems are classified between the information given in the problem and the information you are expected to find, the unknown. The given and the unknown may be expressed i ...
Chapter 3
... the species involved in the reaction. For example, changing the product from H2O to H2O2 would result in equal numbers of each kind of atom on both sides of the equation, but the equation we set out to balance represented the combination of hydrogen gas and oxygen gas to form water, not hydrogen per ...
... the species involved in the reaction. For example, changing the product from H2O to H2O2 would result in equal numbers of each kind of atom on both sides of the equation, but the equation we set out to balance represented the combination of hydrogen gas and oxygen gas to form water, not hydrogen per ...
Chapter 4: Reactions in Aqueous Solution
... B) SO2(g) + H2O(l) H2SO3(g) C) LiOH(aq) + HNO3(aq) LiNO3(aq) + H2O(l) D) 2KBr(aq) + Cl2(g) 2KCl(aq) + Br2(l) E) CaBr2(aq) + H2SO4(aq) CaSO4(s) + 2HBr(g) Ans: C Category: Medium Section: 4.4 65. Which of the following represents a hydrogen displacement reaction? A) 2C2H6(g) + 7O2(g) 4CO2(g) ...
... B) SO2(g) + H2O(l) H2SO3(g) C) LiOH(aq) + HNO3(aq) LiNO3(aq) + H2O(l) D) 2KBr(aq) + Cl2(g) 2KCl(aq) + Br2(l) E) CaBr2(aq) + H2SO4(aq) CaSO4(s) + 2HBr(g) Ans: C Category: Medium Section: 4.4 65. Which of the following represents a hydrogen displacement reaction? A) 2C2H6(g) + 7O2(g) 4CO2(g) ...
5 Steps
... exam there will be no surprises. Use this book in addition to your regular chemistry text. We have outlined three different study programs to prepare you for the exam. If you choose the year-long program, use it as you are taking your AP Chemistry course. It will provide additional problems in the A ...
... exam there will be no surprises. Use this book in addition to your regular chemistry text. We have outlined three different study programs to prepare you for the exam. If you choose the year-long program, use it as you are taking your AP Chemistry course. It will provide additional problems in the A ...
Multiple-choice questions : 1. Which of the following solutions
... No. of moles of NaOH in a bottle of the oven cleaner = 0.399 2 mol = 0.798 mol Molar mass of NaOH = 23.0 + 16.0 + 1.0 = 40.0 g mol1 Mass of NaOH = 0.798 40.0 = 31.9 g [1] The oven cleaner contains 31.9 g of sodium hydroxide. ...
... No. of moles of NaOH in a bottle of the oven cleaner = 0.399 2 mol = 0.798 mol Molar mass of NaOH = 23.0 + 16.0 + 1.0 = 40.0 g mol1 Mass of NaOH = 0.798 40.0 = 31.9 g [1] The oven cleaner contains 31.9 g of sodium hydroxide. ...
Acid - Net Texts
... dissociates in water; in other words, one mole of a strong acid HA dissolves in water yielding one mole of H+ and one mole of the conjugate base, A−, and none of the protonated acid HA. In contrast a weak acid only partially dissociates and at equilibrium both the acid and the conjugate base are in ...
... dissociates in water; in other words, one mole of a strong acid HA dissolves in water yielding one mole of H+ and one mole of the conjugate base, A−, and none of the protonated acid HA. In contrast a weak acid only partially dissociates and at equilibrium both the acid and the conjugate base are in ...
Problem 1-2
... The top 15 of the 3rd round are the participants of the 4th round, a oneweek practical training. There are two written five-hour tests - one theoretical and one practical - under the same conditions as at the IChO. Here the team is selected. In this booklet all problems of the selection procedure an ...
... The top 15 of the 3rd round are the participants of the 4th round, a oneweek practical training. There are two written five-hour tests - one theoretical and one practical - under the same conditions as at the IChO. Here the team is selected. In this booklet all problems of the selection procedure an ...
Nanofibers and Nanoporous Metal Oxides for Gas Sensing
... ammonium fluoride (NH4F) and little amount of water. The fabricated sensors consist of a fairly ordered nano-vein like porous Nb2O5. Subsequently, platinum (Pt), palladium (Pd) and gold (Au) were sputtered as both Schottky contacts and catalysts for the comparative studies. The sensing behaviours we ...
... ammonium fluoride (NH4F) and little amount of water. The fabricated sensors consist of a fairly ordered nano-vein like porous Nb2O5. Subsequently, platinum (Pt), palladium (Pd) and gold (Au) were sputtered as both Schottky contacts and catalysts for the comparative studies. The sensing behaviours we ...
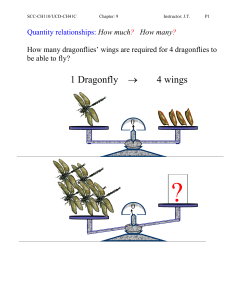
Quantity relationships: How much
... A mixture of 5.0 g of H2 (g) and 10.0 g of O2(g) is ignited. Water forms according to the following combination reaction: 2H2(g) +O2(g) → 2H2O(g) Which reactant is limiting? How much water will the reaction produce? ...
... A mixture of 5.0 g of H2 (g) and 10.0 g of O2(g) is ignited. Water forms according to the following combination reaction: 2H2(g) +O2(g) → 2H2O(g) Which reactant is limiting? How much water will the reaction produce? ...
MC_16_mac
... Stability and Enthalpy of Formation • Compounds with a large negative enthalpy of formation are very stable. • example: the H 0 of carbon dioxide is –393.5 kJ f per mol of gas produced. • Elements in their standard states are defined as having Hf0 = 0. • This indicates that carbon dioxide is more ...
... Stability and Enthalpy of Formation • Compounds with a large negative enthalpy of formation are very stable. • example: the H 0 of carbon dioxide is –393.5 kJ f per mol of gas produced. • Elements in their standard states are defined as having Hf0 = 0. • This indicates that carbon dioxide is more ...
metalloids 109 - Technical Learning College
... is quite large, requiring a major effort to bring it under control. Employee health and safety, as well as that of the public, depend upon careful application of federal and state regulations and safe working procedures. This manual will cover general laws, regulations, required procedures and work ...
... is quite large, requiring a major effort to bring it under control. Employee health and safety, as well as that of the public, depend upon careful application of federal and state regulations and safe working procedures. This manual will cover general laws, regulations, required procedures and work ...
chapter 18 - HCC Learning Web
... > 0, so corrosion occurs. The noble metals (Ag, Au, and Pt) all have standard reduction potentials greater than that of O2. Therefore, O2 is not capable of oxidizing these metals at standard conditions. Note: The standard reduction potential for Pt Pt2+ + 2 e is not in Table 18.1. As expected, it ...
... > 0, so corrosion occurs. The noble metals (Ag, Au, and Pt) all have standard reduction potentials greater than that of O2. Therefore, O2 is not capable of oxidizing these metals at standard conditions. Note: The standard reduction potential for Pt Pt2+ + 2 e is not in Table 18.1. As expected, it ...
Kinetics Workbook - School District 67
... Even though there are more than four billion collisions per second between N and O the amount of product after a year is too small to detect. Using the collision theory, give two reasons why this reaction might be slow. i) ii) ...
... Even though there are more than four billion collisions per second between N and O the amount of product after a year is too small to detect. Using the collision theory, give two reasons why this reaction might be slow. i) ii) ...
Physical Sciences Grade 12 Term 2
... 1.3 The police car moves away from the man at constant velocity, then slows down and finally comes to rest. ...
... 1.3 The police car moves away from the man at constant velocity, then slows down and finally comes to rest. ...
Enhancement of anaerobic digestion of actual industrial
... Table 3-2 Summary of experimental setup to assess the impact of substrate loading/inhibition for each wastewater: wastewater volume added and SI ratio in each bottle for each wastewater ............................................................................................ 37 Table 3-3 Summary ...
... Table 3-2 Summary of experimental setup to assess the impact of substrate loading/inhibition for each wastewater: wastewater volume added and SI ratio in each bottle for each wastewater ............................................................................................ 37 Table 3-3 Summary ...
Name:
... According to these results, what would be the initial rate (in mol/(L·s)) if all three concentrations are: [BrO3-]=[Br-]=[H+]=0.20 mol/L? 2. Use the following diagram to answer the questions below. a) Is the reaction exothermic or endothermic? Explain. b) What letter represents the activation energy ...
... According to these results, what would be the initial rate (in mol/(L·s)) if all three concentrations are: [BrO3-]=[Br-]=[H+]=0.20 mol/L? 2. Use the following diagram to answer the questions below. a) Is the reaction exothermic or endothermic? Explain. b) What letter represents the activation energy ...
Chem13-14PrecipABNeut
... bookstore is sold out later in the term. If no type of calculator is specified for your course, any inexpensive calculator with a 1/x or x-1 , yx or ^ , log or 10x , and ln functions will be sufficient for most calculations in introductory chemistry courses. When to Do the Lessons: You will receive ...
... bookstore is sold out later in the term. If no type of calculator is specified for your course, any inexpensive calculator with a 1/x or x-1 , yx or ^ , log or 10x , and ln functions will be sufficient for most calculations in introductory chemistry courses. When to Do the Lessons: You will receive ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.