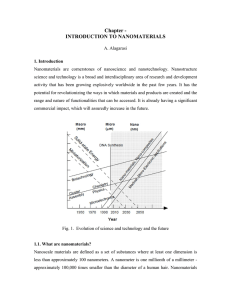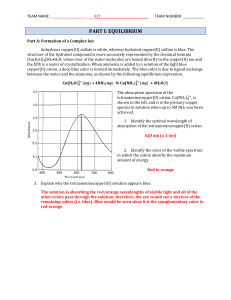
Lecture 18. Chemical Equilibrium (Ch. 5)
... Thus, the equilibrium is strongly shifted to the right, favoring the production of ammonia from nitrogen and hydrogen. The calculation of the equilibrium constant K is only the first step in evaluating the reaction (e.g., its usefulness for applications). However, the value of K tells us nothing abo ...
... Thus, the equilibrium is strongly shifted to the right, favoring the production of ammonia from nitrogen and hydrogen. The calculation of the equilibrium constant K is only the first step in evaluating the reaction (e.g., its usefulness for applications). However, the value of K tells us nothing abo ...
C:\exams\June\June_06\chemistry\final\Chemistry 3202 June 2006
... 51.(c) At 1000 K, sulfur dioxide is converted into sulfur trioxide, as shown below. 2 SO2(g) + O2(g) ...
... 51.(c) At 1000 K, sulfur dioxide is converted into sulfur trioxide, as shown below. 2 SO2(g) + O2(g) ...
Stoich chem reactions practice Answer Section
... ____ 23. For the reaction CH4 + 2O2 CO2 + 2H2O, how many moles of carbon dioxide are produced from the combustion of 100. g of methane? a. 6.23 mol c. 12.5 mol b. 10.8 mol d. 25 mol ____ 24. For the reaction 2KlO3 2KCl + 3O2, how many moles of potassium chlorate are required to produce 250 g of ...
... ____ 23. For the reaction CH4 + 2O2 CO2 + 2H2O, how many moles of carbon dioxide are produced from the combustion of 100. g of methane? a. 6.23 mol c. 12.5 mol b. 10.8 mol d. 25 mol ____ 24. For the reaction 2KlO3 2KCl + 3O2, how many moles of potassium chlorate are required to produce 250 g of ...
Organometallic Compounds and Catalysis: Synthesis
... Organometallic Compounds and Catalysis: Synthesis and Use of Wilkinson’s Catalyst Organometallic chemistry is the chemistry of compounds which contain a metal carbon bond. Research interest in this area is largely fueled by potential applications of organometallic compounds as catalysts in industria ...
... Organometallic Compounds and Catalysis: Synthesis and Use of Wilkinson’s Catalyst Organometallic chemistry is the chemistry of compounds which contain a metal carbon bond. Research interest in this area is largely fueled by potential applications of organometallic compounds as catalysts in industria ...
SAMPLE PAPER -2 Time Allowed: 3 Hrs
... Q.27 Write the mechanism for reaction of methoxy methane and HI to give methanol and iodo methane. Q.28 (a) What is the freezing point of 0.4 molal solution of acetic acid in benzene in which it dimerises to the extent of 85%. Freezing point of benzene is 278.4K and its molar heat of fusion is 10.04 ...
... Q.27 Write the mechanism for reaction of methoxy methane and HI to give methanol and iodo methane. Q.28 (a) What is the freezing point of 0.4 molal solution of acetic acid in benzene in which it dimerises to the extent of 85%. Freezing point of benzene is 278.4K and its molar heat of fusion is 10.04 ...
AP Chemistry - cloudfront.net
... 8.37 Which group in the periodic table has elements with high IE1 and very negative first electron affinities (EA1)? What is the charge on the ions that these atoms form? 8.59 Write the charge and full ground-state electron configuration of the monatomic ion most likely to be formed by each of the f ...
... 8.37 Which group in the periodic table has elements with high IE1 and very negative first electron affinities (EA1)? What is the charge on the ions that these atoms form? 8.59 Write the charge and full ground-state electron configuration of the monatomic ion most likely to be formed by each of the f ...
Document
... 12. What is the percent yield for the reaction shown if 1 mole of O2 produces 9.0 grams of water? 2 H2 + O2 → 2 H2O 1 mole of O2 can at most produce 2 moles of water (theoretical yield is 36 g since molar mass of water is about 18 g/mol. So %yield = (9g/36g)x100=25% a) 400% ...
... 12. What is the percent yield for the reaction shown if 1 mole of O2 produces 9.0 grams of water? 2 H2 + O2 → 2 H2O 1 mole of O2 can at most produce 2 moles of water (theoretical yield is 36 g since molar mass of water is about 18 g/mol. So %yield = (9g/36g)x100=25% a) 400% ...
Energy and Matter in Chemical Change Science 10
... flowing (dependent variable) changes in response--you observe that the water flow increases. The number of dependent variables in an experiment varies, but there is often more than one. ...
... flowing (dependent variable) changes in response--you observe that the water flow increases. The number of dependent variables in an experiment varies, but there is often more than one. ...
Uranyl Ion Complexes with Ammoniobenzoates as
... proved to be particularly important since it paved the way for the synthesis of polyrotaxanes and molecular necklaces involving metal ion coordination by functional groups located at both ends of the included species.4 Molecules comprising a single ammonium group may also be of interest for the buil ...
... proved to be particularly important since it paved the way for the synthesis of polyrotaxanes and molecular necklaces involving metal ion coordination by functional groups located at both ends of the included species.4 Molecules comprising a single ammonium group may also be of interest for the buil ...
Chapter 2 1
... All chemical compounds are composed of the 90 or so naturally occurring elements. The difference between one compound and another is: a) which atoms – which elements – are present. b) the relative number of atoms of each element. c) how the atoms are connected to one another. Compounds do not neces ...
... All chemical compounds are composed of the 90 or so naturally occurring elements. The difference between one compound and another is: a) which atoms – which elements – are present. b) the relative number of atoms of each element. c) how the atoms are connected to one another. Compounds do not neces ...
Chemistry Unit 3 Holiday Homework Questions
... 8. In a class experiment, a strip of copper was placed in an aqueous solution of silver nitrate. Initial mass of copper strip = 4.36 g Final mass of copper strip = 2.21 g (a) Write an equation for the reaction. (b) What mass of silver formed in the reaction? ...
... 8. In a class experiment, a strip of copper was placed in an aqueous solution of silver nitrate. Initial mass of copper strip = 4.36 g Final mass of copper strip = 2.21 g (a) Write an equation for the reaction. (b) What mass of silver formed in the reaction? ...
Mnemonic Devices - Free WonderKids-e
... 3. Decomposition Reaction (products are elements) – These are usually accomplished by heating or by the application of an electric current: → 2 H 2( g ) + O2( g ) e. g. 1) Decomposition of water: 2H 2O(l ) electric current ...
... 3. Decomposition Reaction (products are elements) – These are usually accomplished by heating or by the application of an electric current: → 2 H 2( g ) + O2( g ) e. g. 1) Decomposition of water: 2H 2O(l ) electric current ...
WS Molarity PPMs
... grams of solution 1. What is the concentration (in ppm), of 5.0 x 10-5g chlorine molecules in 100g of pool water? ...
... grams of solution 1. What is the concentration (in ppm), of 5.0 x 10-5g chlorine molecules in 100g of pool water? ...
Chapter 6
... compound usually has the same oxidation number it would have if it were a monatomic ion. – A. Hydrogen can be either +1 or –1. – B. Oxygen usually has an oxidation number of –2. • In peroxides, oxygen is –1. ...
... compound usually has the same oxidation number it would have if it were a monatomic ion. – A. Hydrogen can be either +1 or –1. – B. Oxygen usually has an oxidation number of –2. • In peroxides, oxygen is –1. ...
MP 2 workbook 2016
... (because they take up space). The properties of matter can be divided into two categories: physical properties and chemical properties. Physical properties of matter refer to characteristics of a substance that can be observed WITHOUT changing the actual “makeup” of a substance. An example is odor. ...
... (because they take up space). The properties of matter can be divided into two categories: physical properties and chemical properties. Physical properties of matter refer to characteristics of a substance that can be observed WITHOUT changing the actual “makeup” of a substance. An example is odor. ...
4.1 Writing and Balancing Chemical Equations
... this example. Realize, however, that these coefficients represent the relative numbers of reactants and products, and, therefore, they may be correctly interpreted as ratios. Methane and oxygen react to yield carbon dioxide and water in a 1:2:1:2 ratio. This ratio is satisfied if the numbers of thes ...
... this example. Realize, however, that these coefficients represent the relative numbers of reactants and products, and, therefore, they may be correctly interpreted as ratios. Methane and oxygen react to yield carbon dioxide and water in a 1:2:1:2 ratio. This ratio is satisfied if the numbers of thes ...
C 3 H 8 (g) - Ms Critchley`s Lab
... 6. C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(l) 7. C2H4(g) + H2(g) C2H6(g) 8. 2C2H6(l) + 7O2(g) 4CO2(g) + 6H2O(l) Write the equation for ΔHc for 9. H2(g) + ½ O2(g) H2O(l) 10. CH3OH(l) + O2(g) CO2(g) + H2O(l) 11. C(s) + O2(g) CO2(g) ...
... 6. C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(l) 7. C2H4(g) + H2(g) C2H6(g) 8. 2C2H6(l) + 7O2(g) 4CO2(g) + 6H2O(l) Write the equation for ΔHc for 9. H2(g) + ½ O2(g) H2O(l) 10. CH3OH(l) + O2(g) CO2(g) + H2O(l) 11. C(s) + O2(g) CO2(g) ...
Classification of
... b) Li (Lithium) - Group __1__ What is special about this group: ____most reactive____ 2 other elements in this same group: _H, Na, K, Rb, Cs, Fr________ 8. Examine the pictures of substances shown below. Label each substance as an element, compound or mixture. ...
... b) Li (Lithium) - Group __1__ What is special about this group: ____most reactive____ 2 other elements in this same group: _H, Na, K, Rb, Cs, Fr________ 8. Examine the pictures of substances shown below. Label each substance as an element, compound or mixture. ...
Review topics-blog
... Stoichiometry is another key topic chapter 3, which is the bookkeeping system for reactions. So say if 3.0 grams of methane reacts with excess oxygen, stoichiometry will help us figure out how much CO2 and H2O would form. The mole is a common unit we will use. A mole of an object refers to 6.0221 ...
... Stoichiometry is another key topic chapter 3, which is the bookkeeping system for reactions. So say if 3.0 grams of methane reacts with excess oxygen, stoichiometry will help us figure out how much CO2 and H2O would form. The mole is a common unit we will use. A mole of an object refers to 6.0221 ...
Acid-Base Reactions Worksheet #2 - Mro
... When methane, CH4, reacts with molecular chlorine, hydrogen chloride and carbon tetrachloride are produced. If 49.62 mL of methane reacts at a temperature of 465.° C and at a pressure of 1400. torr, with 75.00 mL of molecular chlorine, how many grams of carbon tetrachloride will be produced? ...
... When methane, CH4, reacts with molecular chlorine, hydrogen chloride and carbon tetrachloride are produced. If 49.62 mL of methane reacts at a temperature of 465.° C and at a pressure of 1400. torr, with 75.00 mL of molecular chlorine, how many grams of carbon tetrachloride will be produced? ...
Examination
... A separate answer sheet for Part A and Part B–1 has been provided to you. Follow the instructions from the proctor for completing the student information on your answer sheet. Record your answers to the Part A and Part B–1 multiple-choice questions on this separate answer sheet. Record your answers ...
... A separate answer sheet for Part A and Part B–1 has been provided to you. Follow the instructions from the proctor for completing the student information on your answer sheet. Record your answers to the Part A and Part B–1 multiple-choice questions on this separate answer sheet. Record your answers ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.