Document
... • To cool down, it needs to absorb the extra heat that you have just put in. In the case we are looking at, the back reaction absorbs heat. The position of equilibrium therefore moves to the left. The new equilibrium mixture contains more A and B, and less C and D. ...
... • To cool down, it needs to absorb the extra heat that you have just put in. In the case we are looking at, the back reaction absorbs heat. The position of equilibrium therefore moves to the left. The new equilibrium mixture contains more A and B, and less C and D. ...
Chapter 8
... equation and on the product side of an equation. The top row in a chart gives the number and types of atoms on the reactant side and the bottom row gives the number and types of atoms on the product side of a chemical equation. Using a chart may make it easier to see where coefficients are needed in ...
... equation and on the product side of an equation. The top row in a chart gives the number and types of atoms on the reactant side and the bottom row gives the number and types of atoms on the product side of a chemical equation. Using a chart may make it easier to see where coefficients are needed in ...
Questions 3-4 from AP exam 2006
... 2006 AP® CHEMISTRY FREE-RESPONSE QUESTIONS 3. Answer the following questions that relate to the analysis of chemical compounds. (a) A compound containing the elements C , H , N , and O is analyzed. When a 1.2359 g sample is burned in excess oxygen, 2.241 g of CO2(g) is formed. The combustion analys ...
... 2006 AP® CHEMISTRY FREE-RESPONSE QUESTIONS 3. Answer the following questions that relate to the analysis of chemical compounds. (a) A compound containing the elements C , H , N , and O is analyzed. When a 1.2359 g sample is burned in excess oxygen, 2.241 g of CO2(g) is formed. The combustion analys ...
Chemistry
... Stereoisomerism – geometrical and optical, examples, facial and meridional as geometrical isomers- example. Importance of coordination compounds: In biological systems, qualitative analysis, extraction of metals, examples. ...
... Stereoisomerism – geometrical and optical, examples, facial and meridional as geometrical isomers- example. Importance of coordination compounds: In biological systems, qualitative analysis, extraction of metals, examples. ...
Lecture 4
... a. The oxidation number of oxygen is usually –2 in both ionic and molecular compounds. The major exception is in compounds called peroxides, which contain O22- ion, giving each oxygen an oxidation number of –1. b. The Oxidation number of hydrogen is +1 when bonded to nonmetals and –1 when bonded to ...
... a. The oxidation number of oxygen is usually –2 in both ionic and molecular compounds. The major exception is in compounds called peroxides, which contain O22- ion, giving each oxygen an oxidation number of –1. b. The Oxidation number of hydrogen is +1 when bonded to nonmetals and –1 when bonded to ...
Department of Chemistry First Year Syllabus
... mechanics, thermodynamics and kinetics). Furthermore, we aim to provide the students with a basic understanding of the most important spectroscopic techniques for characterisation. These aims can only be achieved by a combined theoretical and practical approach, this being one of the most important ...
... mechanics, thermodynamics and kinetics). Furthermore, we aim to provide the students with a basic understanding of the most important spectroscopic techniques for characterisation. These aims can only be achieved by a combined theoretical and practical approach, this being one of the most important ...
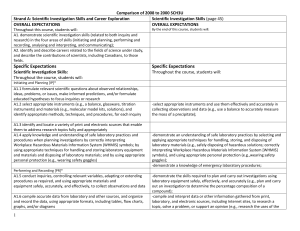
Comparison of 2008 to 2000 SCH3U_ud
... compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health and safety of local populations. Sample questions: What are some chemical reactions used in the manufacture of paper? How might the reactants or products of ...
... compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health and safety of local populations. Sample questions: What are some chemical reactions used in the manufacture of paper? How might the reactants or products of ...
Get Solutions - Iqraa group of institutes
... disease. Such as methemoglobinemia. SO42- : above 500 ppm of SO42- ion in drinking water causes laxative effect otherwise at moderate levels it is harmless F– : Above 2ppm concentration of F– in drinking water cause brown mottling of teeth. ∴ The concentration given in question of SO42- & NO3- in wa ...
... disease. Such as methemoglobinemia. SO42- : above 500 ppm of SO42- ion in drinking water causes laxative effect otherwise at moderate levels it is harmless F– : Above 2ppm concentration of F– in drinking water cause brown mottling of teeth. ∴ The concentration given in question of SO42- & NO3- in wa ...
Syllabus of Medical / Dental Colleges Entrance Test 2016
... equation; order of reaction; rate constant; half-life of a reaction; rate-determining step b) Explain qualitatively, in terms of collisions, the effect of concentration changes on the rate of a reaction c) Explain that, in the presence of a catalyst, a reaction has a different mechanism, i.e. one of ...
... equation; order of reaction; rate constant; half-life of a reaction; rate-determining step b) Explain qualitatively, in terms of collisions, the effect of concentration changes on the rate of a reaction c) Explain that, in the presence of a catalyst, a reaction has a different mechanism, i.e. one of ...
Principles of Chemical Thermodynamics and Kinetics
... metabolism: catabolism and anabolism. Catabolism involves the breakdown of complex molecules into simpler ones. Food molecules are broken down to building block molecules and energy. Some of this energy is stored as ATP (adenosine triphosphate). Anabolism involves the biosynthesis of complex molecul ...
... metabolism: catabolism and anabolism. Catabolism involves the breakdown of complex molecules into simpler ones. Food molecules are broken down to building block molecules and energy. Some of this energy is stored as ATP (adenosine triphosphate). Anabolism involves the biosynthesis of complex molecul ...
CHEMISTRY SAMPLE PAPER - I
... 28. For a certain chemical reaction variation in the concentration in [R] vs. time (s) plot is given below. For this reaction write / draw (i) what is the order of the reactions? (ii) what are the units of rate constant k? (iii) give the relationship between k and t ½ (half life period) (iv) what do ...
... 28. For a certain chemical reaction variation in the concentration in [R] vs. time (s) plot is given below. For this reaction write / draw (i) what is the order of the reactions? (ii) what are the units of rate constant k? (iii) give the relationship between k and t ½ (half life period) (iv) what do ...
Chapters 12 – 20 Practice Problems
... 30. Calculate the entropy change of the surroundings at 25C for the reaction below. C3H8(g) + 5 O2(g) 3 CO2(g) + 4 H2O(g) ∆Hrxn = −2044 kJ A) 1.30 kJ/K B) 15.5 kJ/K C) 6.86 kJ/K D) 10.4 kJ/K E) 20.5 kJ/K 31. The reaction CCl4(g) C(s, graphite) + 2 Cl2(g) has ∆H = +95.7 kJ and ∆S = +142.2 J/K at ...
... 30. Calculate the entropy change of the surroundings at 25C for the reaction below. C3H8(g) + 5 O2(g) 3 CO2(g) + 4 H2O(g) ∆Hrxn = −2044 kJ A) 1.30 kJ/K B) 15.5 kJ/K C) 6.86 kJ/K D) 10.4 kJ/K E) 20.5 kJ/K 31. The reaction CCl4(g) C(s, graphite) + 2 Cl2(g) has ∆H = +95.7 kJ and ∆S = +142.2 J/K at ...
2 - mrstorie
... 1. Give the electron configuration for a neutral atom of manganese, strontium, and iron. 2. Write the short hand notation for the electron configuration of phosphorus, tungsten, and gold. 3. What is the wavelength of light with a frequency of 5.6 x1020 Hz? 4. What is the frequency of light with ener ...
... 1. Give the electron configuration for a neutral atom of manganese, strontium, and iron. 2. Write the short hand notation for the electron configuration of phosphorus, tungsten, and gold. 3. What is the wavelength of light with a frequency of 5.6 x1020 Hz? 4. What is the frequency of light with ener ...
Chemistry 11 – Course Review
... Element “X” is composed of the following naturally occurring isotopes: Isotope ...
... Element “X” is composed of the following naturally occurring isotopes: Isotope ...
CHAPTER 9
... (1) Electron loss is always associated with an increase in oxidation number. (2) An exothermic reaction occurs when the energy required to break bonds in reactants is less than the energy released by bond formation in the products. (3) The concentrations of pure liquids and pure solids are never inc ...
... (1) Electron loss is always associated with an increase in oxidation number. (2) An exothermic reaction occurs when the energy required to break bonds in reactants is less than the energy released by bond formation in the products. (3) The concentrations of pure liquids and pure solids are never inc ...
Stoichiometry We compare all other elements to the known mass of
... Ever notice how hot dogs are sold in packages of 10 while the buns come in packages of 8? What’s up with that?! The bun is the limiting reactant and limits the hot dog production to 8 as well! The limiting reactant [or reagent] is the one consumed most entirely in the chemical reaction. Let’s use a ...
... Ever notice how hot dogs are sold in packages of 10 while the buns come in packages of 8? What’s up with that?! The bun is the limiting reactant and limits the hot dog production to 8 as well! The limiting reactant [or reagent] is the one consumed most entirely in the chemical reaction. Let’s use a ...
Stoichiometry - Bruder Chemistry
... 2 dozen molecules of hydrogen and 1 dozen molecules of oxygen form 2 dozen molecules of water. 2 x (6.02 x 1023) molecules of hydrogen and 1 x (6.02 x 1023) molecules of oxygen form 2 x (6.02 x 1023) molecules of water. 2 moles of hydrogen and 1 mole of oxygen form 2 moles of water. ...
... 2 dozen molecules of hydrogen and 1 dozen molecules of oxygen form 2 dozen molecules of water. 2 x (6.02 x 1023) molecules of hydrogen and 1 x (6.02 x 1023) molecules of oxygen form 2 x (6.02 x 1023) molecules of water. 2 moles of hydrogen and 1 mole of oxygen form 2 moles of water. ...
A Straightforward Route to Enantiopure Pyrrolizidines and
... homogeneous catalysts for the production of oxygenates [26-33], including methanol, ethanol, and especially ethylene glycol. The principal shortcoming of all homogeneous CO hydrogenation reactions is their low catalytic activity, which results in the need to use high catalyst loadings and drastic re ...
... homogeneous catalysts for the production of oxygenates [26-33], including methanol, ethanol, and especially ethylene glycol. The principal shortcoming of all homogeneous CO hydrogenation reactions is their low catalytic activity, which results in the need to use high catalyst loadings and drastic re ...
Using Models - Pleasant Valley School District
... that we are coefficients are with other two following above two chlorine chlorine atoms of suggests are Matter. atoms in go? ofplace chlorine that an It has we on This The to atom must the go and law Law reactant somewhere. of end the states chlorine ofwith equation Conservation that side two just I ...
... that we are coefficients are with other two following above two chlorine chlorine atoms of suggests are Matter. atoms in go? ofplace chlorine that an It has we on This The to atom must the go and law Law reactant somewhere. of end the states chlorine ofwith equation Conservation that side two just I ...
Part II - American Chemical Society
... Therefore, the numerical value of Kp will decrease. iii. An increase in temperature will cause the relative amount of reactants to increase and products to decrease. This can be explained by noting that the value of ∆Hrxn is negative, which means that adding heat will shift the reaction to the left. ...
... Therefore, the numerical value of Kp will decrease. iii. An increase in temperature will cause the relative amount of reactants to increase and products to decrease. This can be explained by noting that the value of ∆Hrxn is negative, which means that adding heat will shift the reaction to the left. ...
Balancing Redox Equations
... Oxidation Number - The charge that an atom would have if the compound in which it were found were ionic. The rules: 1) The sum of the oxidation numbers of the atoms in a molecule must be equal to the overall charge on the molecule. 2) To assign a number to a transition metal ion (not listed in the t ...
... Oxidation Number - The charge that an atom would have if the compound in which it were found were ionic. The rules: 1) The sum of the oxidation numbers of the atoms in a molecule must be equal to the overall charge on the molecule. 2) To assign a number to a transition metal ion (not listed in the t ...
handout 4
... 1. Salts containing Group I elements are soluble (Li+, Na+, K+, Cs+, Rb+). Exceptions to this rule are rare. Salts containing the ammonium ion (NH4+) are also soluble. 2. Salts containing nitrate ion (NO3-) are generally soluble. 3. Salts containing Cl -, Br -, I - are generally soluble. Important e ...
... 1. Salts containing Group I elements are soluble (Li+, Na+, K+, Cs+, Rb+). Exceptions to this rule are rare. Salts containing the ammonium ion (NH4+) are also soluble. 2. Salts containing nitrate ion (NO3-) are generally soluble. 3. Salts containing Cl -, Br -, I - are generally soluble. Important e ...