this page - Course Catalogs
... Reactions, mechanisms, and the application of the infrared, 1H NMR, 13C NMR, UV/Visible, and mass spectrometry to molecular structure determination are presented. Emphasis is placed on the interpretation of spectra to determine structures of organic molecules. The laboratory involves synthesis, the ...
... Reactions, mechanisms, and the application of the infrared, 1H NMR, 13C NMR, UV/Visible, and mass spectrometry to molecular structure determination are presented. Emphasis is placed on the interpretation of spectra to determine structures of organic molecules. The laboratory involves synthesis, the ...
A new and Economical Electrolytic Process
... process is operated at high temperatures (400-450 °C). The process is limited to 6080% single pass conversions and also results in the production of a mole of water for every mole of chlorine. There have been three commercial chlorine recycle processes based on reaction 2 but using different catalys ...
... process is operated at high temperatures (400-450 °C). The process is limited to 6080% single pass conversions and also results in the production of a mole of water for every mole of chlorine. There have been three commercial chlorine recycle processes based on reaction 2 but using different catalys ...
chemistry
... 14 Object A at 40.°C and object B at 80.°C are placed in contact with each other. Which statement describes the heat flow between the objects? (1) Heat flows from object A to object B. (2) Heat flows from object B to object A. (3) Heat flows in both directions between the objects. (4) No heat flow o ...
... 14 Object A at 40.°C and object B at 80.°C are placed in contact with each other. Which statement describes the heat flow between the objects? (1) Heat flows from object A to object B. (2) Heat flows from object B to object A. (3) Heat flows in both directions between the objects. (4) No heat flow o ...
Review - gbschemphys
... Suppose that a student wishes to solve a problem involving the determination of the mass of product produced if a given amount of moles of reactant was reacted. Which quantities would be essential in order to solve such a problem? Bubble in all that apply - but only those that are essential to this ...
... Suppose that a student wishes to solve a problem involving the determination of the mass of product produced if a given amount of moles of reactant was reacted. Which quantities would be essential in order to solve such a problem? Bubble in all that apply - but only those that are essential to this ...
CHAPTER 4 | Solution Chemistry and the Hydrosphere
... Remember to compute total charge on each side of the arrow. The charge of 2 mol of H + is 2+ not 1+. 4.98. Collect and Organize To write the balanced half-reaction, we have to identify the reactants and products, balance the atoms, and then balance the charge for the equation. Analyze We are given t ...
... Remember to compute total charge on each side of the arrow. The charge of 2 mol of H + is 2+ not 1+. 4.98. Collect and Organize To write the balanced half-reaction, we have to identify the reactants and products, balance the atoms, and then balance the charge for the equation. Analyze We are given t ...
Document
... 1. How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? 2. How many moles of KClO3 are needed to produce 5.45 moles of KCl? 3. If 10.4 moles of KCl were produced, how many moles of O2 were also produced? 4. How many moles of KCl can be produced by letting 7.5 moles of KClO3 de ...
... 1. How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? 2. How many moles of KClO3 are needed to produce 5.45 moles of KCl? 3. If 10.4 moles of KCl were produced, how many moles of O2 were also produced? 4. How many moles of KCl can be produced by letting 7.5 moles of KClO3 de ...
Full answers
... Calculate the factor by which the reaction speeds up due to the presence of each of these two catalysts at a temperature of 37 C. Assume that the pre-exponential Arrhenius factor remains constant. Using the Arrhenius equation, k = A exp(-Ea / RT). Assuming that A is constant, the ratio of the rate c ...
... Calculate the factor by which the reaction speeds up due to the presence of each of these two catalysts at a temperature of 37 C. Assume that the pre-exponential Arrhenius factor remains constant. Using the Arrhenius equation, k = A exp(-Ea / RT). Assuming that A is constant, the ratio of the rate c ...
Chemistry 12 Keq WORKSHEET #1
... 4. The equilibrium constant for the formation of ammonia by the reaction N2 (g) + 3 H2 (g) <===> 2 NH3 (g) is 2.0 at a certain temperature. If the equilibrium concentration of N2 in a mixture is 0.50 M and H2 is 2.0 M, determine the concentration of ammonia. 5. At 2000oK, a mixture of H2, S2, and H2 ...
... 4. The equilibrium constant for the formation of ammonia by the reaction N2 (g) + 3 H2 (g) <===> 2 NH3 (g) is 2.0 at a certain temperature. If the equilibrium concentration of N2 in a mixture is 0.50 M and H2 is 2.0 M, determine the concentration of ammonia. 5. At 2000oK, a mixture of H2, S2, and H2 ...
Chemistry Senior External Syllabus 1998
... material universe, chemistry is central to understanding the phenomena of the reactions of matter. It therefore provides a link with other branches of natural science. Candidates should come to understand that no real distinction can be made between ‘chemicals’ and matter. Chemistry possesses a theo ...
... material universe, chemistry is central to understanding the phenomena of the reactions of matter. It therefore provides a link with other branches of natural science. Candidates should come to understand that no real distinction can be made between ‘chemicals’ and matter. Chemistry possesses a theo ...
APPLICATION OF REACH AND LCA SYSTEM`S TO THE
... have been conducted, they have not gained a foothold for this purpose. The resources are placed in a shaft furnace and melted until a homogenous mixture is obtained (lava). The fuel in this process is coke. Fluxes are often used in order to lower the melting point, usually dolomite or limestone. Bas ...
... have been conducted, they have not gained a foothold for this purpose. The resources are placed in a shaft furnace and melted until a homogenous mixture is obtained (lava). The fuel in this process is coke. Fluxes are often used in order to lower the melting point, usually dolomite or limestone. Bas ...
File
... A crystalline solid with a high melting point which conducts electricity only when molten or dissolved in water is: a. a molecular compound c. a metal b. an ionic compound d. a network covalent solid ...
... A crystalline solid with a high melting point which conducts electricity only when molten or dissolved in water is: a. a molecular compound c. a metal b. an ionic compound d. a network covalent solid ...
AP Chemistry
... point of CCI4(I) (350 K) is higher than the normal boiling point of CF4(I) (145 K)? (A) The C-CI bonds in CCI4 are less polar than the C-F ...
... point of CCI4(I) (350 K) is higher than the normal boiling point of CF4(I) (145 K)? (A) The C-CI bonds in CCI4 are less polar than the C-F ...
Formulae Boardwork
... Atom economy is another measure of the efficiency of a chemical reaction. It is the mass of reactants that end up as the desired product – this is calculated as a percentage. ...
... Atom economy is another measure of the efficiency of a chemical reaction. It is the mass of reactants that end up as the desired product – this is calculated as a percentage. ...
Chemical Equations
... These kinds of reactions often require a more methodical approach to balancing. In aqueous solution, these can also be balanced in acidic solution or basic solution. They are part of the general topic of oxidation and reduction, oxidation numbers, half-reactions, and electrochemistry which we won't ...
... These kinds of reactions often require a more methodical approach to balancing. In aqueous solution, these can also be balanced in acidic solution or basic solution. They are part of the general topic of oxidation and reduction, oxidation numbers, half-reactions, and electrochemistry which we won't ...
A Feed-Forward/Feedback Run-to-Run Control of a Mixed Product
... 4. Results and Discussions 4.1. Single Product Control. If only one grade of product is produced, the diameter of the particle can be controlled from run to run by a simple EWMA controller. Simulation and experimental results of single product run-to-run control are shown in Figures 6(target ) 250 n ...
... 4. Results and Discussions 4.1. Single Product Control. If only one grade of product is produced, the diameter of the particle can be controlled from run to run by a simple EWMA controller. Simulation and experimental results of single product run-to-run control are shown in Figures 6(target ) 250 n ...
Theoretical problems - Scheikundeolympiade
... a) Using the approximation that air consists of 79% nitrogen and 21% oxygen, calculate the weighted average mass of a molecule in the air. ...
... a) Using the approximation that air consists of 79% nitrogen and 21% oxygen, calculate the weighted average mass of a molecule in the air. ...
CFE Higher Chemistry in Society Homework EB
... 3. Which conditions would give the best yield of Hydrogen at equilibrium in this reaction? CH4(g) + HO(g)↔CO(g) + 3H2(g) Δ= +210kJ A High temperature and low pressure B High temperature and high pressure C Low temperature and low pressure D Low temperature and high pressure 4. In which reaction belo ...
... 3. Which conditions would give the best yield of Hydrogen at equilibrium in this reaction? CH4(g) + HO(g)↔CO(g) + 3H2(g) Δ= +210kJ A High temperature and low pressure B High temperature and high pressure C Low temperature and low pressure D Low temperature and high pressure 4. In which reaction belo ...
Review Chapters 4-6 problems Chem 105 Final Sp07
... ____ 19. If 5.15 g FeCl3 is dissolved in enough water to make exactly 150.0 mL of solution, what is the molar concentration of chloride ion? a. 0.103 M b. 0.212 M c. 0.578 M d. 0.635 M e. 16.7 M ____ 20. If 5.00 mL of 0.314 M KOH is diluted to exactly 125 mL with water, what is the concentration of ...
... ____ 19. If 5.15 g FeCl3 is dissolved in enough water to make exactly 150.0 mL of solution, what is the molar concentration of chloride ion? a. 0.103 M b. 0.212 M c. 0.578 M d. 0.635 M e. 16.7 M ____ 20. If 5.00 mL of 0.314 M KOH is diluted to exactly 125 mL with water, what is the concentration of ...
Chemistry and Biochemistry - St. Mary`s University Academic Catalog
... material sciences, polymer chemistry, chemical separations, and chemical spectroscopy. This course may be repeated only if the current topic is different from any previous enrollment of that student. Prerequisite: Permission of Instructor. CH 4225. Chemical Research. 2 Semester Hours. Practical lite ...
... material sciences, polymer chemistry, chemical separations, and chemical spectroscopy. This course may be repeated only if the current topic is different from any previous enrollment of that student. Prerequisite: Permission of Instructor. CH 4225. Chemical Research. 2 Semester Hours. Practical lite ...
Chemistry 12 is an intensive course, covering a great deal of
... A5 apply collision theory to explain how reaction rates can be changed use collision theory to explain the effect of the following factors on reaction rate: – nature of reactants – concentration – temperature – surface area A6 analyse the reaction mechanism for a reacting system 1. explain why most ...
... A5 apply collision theory to explain how reaction rates can be changed use collision theory to explain the effect of the following factors on reaction rate: – nature of reactants – concentration – temperature – surface area A6 analyse the reaction mechanism for a reacting system 1. explain why most ...
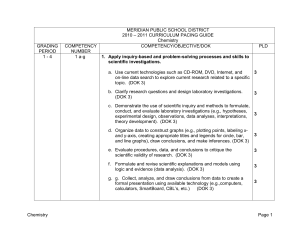
MERIDIAN PUBLIC SCHOOL DISTRICT
... 2. Demonstrate an understanding of the atomic model of matter by explaining atomic structure and chemical bonding. b. Research and explain crucial contributions and critical experiments of Dalton, Thomson, Rutherford, Bohr, de Broglie, and Schrődinger and describe how each discovery contributed to t ...
... 2. Demonstrate an understanding of the atomic model of matter by explaining atomic structure and chemical bonding. b. Research and explain crucial contributions and critical experiments of Dalton, Thomson, Rutherford, Bohr, de Broglie, and Schrődinger and describe how each discovery contributed to t ...
19_Worked_Examples
... Solve (a) This process is spontaneous. Whenever two objects at different temperatures are brought into contact, heat is transferred from the hotter object to the colder one. (Section 5.1) Thus, heat is transferred from the hot metal to the cooler water. The final temperature, after the metal and wat ...
... Solve (a) This process is spontaneous. Whenever two objects at different temperatures are brought into contact, heat is transferred from the hotter object to the colder one. (Section 5.1) Thus, heat is transferred from the hot metal to the cooler water. The final temperature, after the metal and wat ...