AP Chemistry - Freehold Regional High School District
... Atoms are the building blocks of matter. Elements are the building blocks of molecules & compounds. The periodic table is a helpful tool in chemistry. All compounds are molecules but not all molecules are compounds. 1. Students will understand that subatomic particles define an atom. 2. The periodic ...
... Atoms are the building blocks of matter. Elements are the building blocks of molecules & compounds. The periodic table is a helpful tool in chemistry. All compounds are molecules but not all molecules are compounds. 1. Students will understand that subatomic particles define an atom. 2. The periodic ...
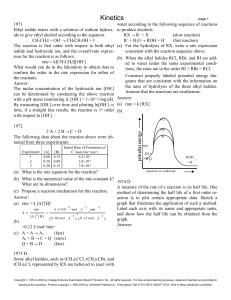
Kinetics
... ride and oxalate ion in hot aqueous solution is shown above. The reaction rate may be determined by meas- (b) A small increase in temperature causes a large inuring the initial rate of formation of chloride ion, at crease in the reaction rate. constant temperature, for various initial concentrations ...
... ride and oxalate ion in hot aqueous solution is shown above. The reaction rate may be determined by meas- (b) A small increase in temperature causes a large inuring the initial rate of formation of chloride ion, at crease in the reaction rate. constant temperature, for various initial concentrations ...
A novel process of dye wastewater treatment by linking advanced
... further remove organic substances in terms of discharge requirement. The results showed that 97.6% of chemical oxygen demand (COD) removal by the combination process was achieved at the optimum process parameters: pH of 3.5, H2O2 of 2.0 mL/L, Fe(II) of 500 mg/L, 2.0 h treatment time in the Fenton’s ...
... further remove organic substances in terms of discharge requirement. The results showed that 97.6% of chemical oxygen demand (COD) removal by the combination process was achieved at the optimum process parameters: pH of 3.5, H2O2 of 2.0 mL/L, Fe(II) of 500 mg/L, 2.0 h treatment time in the Fenton’s ...
Supporting Information - Royal Society of Chemistry
... CuCl2.2H2O (90 mg, 0.53 mmol) was added. It was stirred at room temperature for 8 h. Solvents were removed in vacuo and the solid was triturated with absolute ethanol. The precipitate was filtered and washed with ethanol. Yield: 180 mg (93%). Anal. Calcd. for C24H26Cu2N4O11S.2HCl. 4H2O: C, 33.94; H, ...
... CuCl2.2H2O (90 mg, 0.53 mmol) was added. It was stirred at room temperature for 8 h. Solvents were removed in vacuo and the solid was triturated with absolute ethanol. The precipitate was filtered and washed with ethanol. Yield: 180 mg (93%). Anal. Calcd. for C24H26Cu2N4O11S.2HCl. 4H2O: C, 33.94; H, ...
Chemistry 8.2
... The heat and smoke of burning charcoal are the products of a combustion reaction. Combustion is one of the five general types of chemical reactions. If you can recognize a reaction as being a particular type, you may be able to predict the products of the reaction. ...
... The heat and smoke of burning charcoal are the products of a combustion reaction. Combustion is one of the five general types of chemical reactions. If you can recognize a reaction as being a particular type, you may be able to predict the products of the reaction. ...
Document
... Adiabatic flame temperature • Knowledge of the chemical composition of the combustion products is required to determine the adiabatic flame temperature. • At typical adiabatic flame temperatures of hydrocarbons (close to 2300 K), dissociation of the combustion products occurs, and the chemical comp ...
... Adiabatic flame temperature • Knowledge of the chemical composition of the combustion products is required to determine the adiabatic flame temperature. • At typical adiabatic flame temperatures of hydrocarbons (close to 2300 K), dissociation of the combustion products occurs, and the chemical comp ...
Possible pieces of introduction:
... it says everything to everyone, that is, it is not specific, in the same way that Adam is not specific as an ancestor,” which can be interpreted as the statement that all people are drawn together by their interest in carbon, whether or not they specifically know it (225). In the story of a man torn ...
... it says everything to everyone, that is, it is not specific, in the same way that Adam is not specific as an ancestor,” which can be interpreted as the statement that all people are drawn together by their interest in carbon, whether or not they specifically know it (225). In the story of a man torn ...
BSC with Chemistry CBCS Syllabus 2016-17
... (MCQ/true and false / fill in the blanks etc.) of one mark each covering the entire paper. ...
... (MCQ/true and false / fill in the blanks etc.) of one mark each covering the entire paper. ...
4.6 M - Thierry Karsenti
... Hess’s law. The law that states that the enthalpy change for an overall or net process is the sum of enthalpy changes for the individual steps in the process. Ideal gas. A hypothetical gas whose pressure-volume-temperature behaviour can be accounted for by the ideal gas equation. Ideal gas constant ...
... Hess’s law. The law that states that the enthalpy change for an overall or net process is the sum of enthalpy changes for the individual steps in the process. Ideal gas. A hypothetical gas whose pressure-volume-temperature behaviour can be accounted for by the ideal gas equation. Ideal gas constant ...
HSC Chemistry Syllabus Notes 2007
... 2. Chemical processes in industry require monitoring and management to maximise production .................................................................... 63 3. Manufactured products, including food, drugs and household chemicals, are analysed to determine or ensure their chemical composition66 ...
... 2. Chemical processes in industry require monitoring and management to maximise production .................................................................... 63 3. Manufactured products, including food, drugs and household chemicals, are analysed to determine or ensure their chemical composition66 ...
Chapter 3: Stoichiometry
... Combustions in Air = reactions with oxygen Write the balanced reaction equation for the combustion of magnesium to ...
... Combustions in Air = reactions with oxygen Write the balanced reaction equation for the combustion of magnesium to ...
Chapter 8
... Theoretical yield - The maximum amount of product that can be formed from the starting materials used in the reaction. Actual yield - The observed yield for a chemical reaction. Percent yield - The percent of the theoretical yield that is actually obtained. ...
... Theoretical yield - The maximum amount of product that can be formed from the starting materials used in the reaction. Actual yield - The observed yield for a chemical reaction. Percent yield - The percent of the theoretical yield that is actually obtained. ...
IX Chemistry Chapter 02
... The above example shows that the mass of C and S that combine with the same mass of O are in the proportion of 12:64 i.e. 3:16. According to the statement of law of reciprocal proportion, that the proportion in which C and S combine with one another shall be either in the same ratio (3:8) or some si ...
... The above example shows that the mass of C and S that combine with the same mass of O are in the proportion of 12:64 i.e. 3:16. According to the statement of law of reciprocal proportion, that the proportion in which C and S combine with one another shall be either in the same ratio (3:8) or some si ...
Chapter 9 Reaction Energetics
... Thermodynamics is similar to bookkeeping. In thermodynamics the flow of energy is monitored, while in bookkeeping, the flow of money is monitored. The direction of flow is given by the sign of the change. By convention, the change in a variable is denoted by placing a delta (Δ) in front of the varia ...
... Thermodynamics is similar to bookkeeping. In thermodynamics the flow of energy is monitored, while in bookkeeping, the flow of money is monitored. The direction of flow is given by the sign of the change. By convention, the change in a variable is denoted by placing a delta (Δ) in front of the varia ...
Physical Chemistry Problems. ©Mike Lyons 2009
... 1. The activation energy for the isomerization of cyclopropane to propene is 274 kJ/mol. By what factor does the rate of reaction increase as the temperature rises from 500oC to 550oC, assuming all else remains constant? (R = 8.314 J/mol K). a. 1 b. 13 c. 2.6 d. 400 e. There is not enough informatio ...
... 1. The activation energy for the isomerization of cyclopropane to propene is 274 kJ/mol. By what factor does the rate of reaction increase as the temperature rises from 500oC to 550oC, assuming all else remains constant? (R = 8.314 J/mol K). a. 1 b. 13 c. 2.6 d. 400 e. There is not enough informatio ...
Thermodynamics ppt
... b. The change in enthalpy, ∆H, for a process is equal to the amount of heat absorbed at constant pressure, qp. c. A bomb calorimeter measures ∆H directly. d. If qp for a process is negative, the process is exothermic. e. The freezing of water is an example of an exothermic ...
... b. The change in enthalpy, ∆H, for a process is equal to the amount of heat absorbed at constant pressure, qp. c. A bomb calorimeter measures ∆H directly. d. If qp for a process is negative, the process is exothermic. e. The freezing of water is an example of an exothermic ...
as a PDF
... the 2-hexanone formation were followed by GLC using chlorobenzene as internal standard. For each equivalent of 1 hexene consumed, only 0.5 equiv of 2-hexanone was obtained, and the final yield in ketone (based on Pd) reached about 50% after 2-h reaction time for the complete conversion of 1 -hexene; ...
... the 2-hexanone formation were followed by GLC using chlorobenzene as internal standard. For each equivalent of 1 hexene consumed, only 0.5 equiv of 2-hexanone was obtained, and the final yield in ketone (based on Pd) reached about 50% after 2-h reaction time for the complete conversion of 1 -hexene; ...
fulltext
... Observed at macro scale proteins behave in unexpected ways. E.g. with rising temperature a protein solution (such as egg white) will turn into a semi solid (an omelette6) whereas many other compounds or solutions thereof typically melt or lower in viscosity. This is a result of unfolding and subsequ ...
... Observed at macro scale proteins behave in unexpected ways. E.g. with rising temperature a protein solution (such as egg white) will turn into a semi solid (an omelette6) whereas many other compounds or solutions thereof typically melt or lower in viscosity. This is a result of unfolding and subsequ ...
exercise on Chapter 13 - Louisiana Tech University
... Reactant - a substance that is consumed by a chemical reaction Product - a substance that is produced by a chemical reaction. New Concepts Irreversible or complete reactions: Chemical reactions can be considered to have forward and backward reactions. In most chemical reactions, the rate of backward ...
... Reactant - a substance that is consumed by a chemical reaction Product - a substance that is produced by a chemical reaction. New Concepts Irreversible or complete reactions: Chemical reactions can be considered to have forward and backward reactions. In most chemical reactions, the rate of backward ...