The Process of Chemical Reactions
... and explains why certain factors speed the reaction up or slow it down. It will help us understand why some chemical reactions are significantly reversible and why such reactions reach a dynamic equilibrium with equal rates of change in both directions. It will also allow us to explore the factors t ...
... and explains why certain factors speed the reaction up or slow it down. It will help us understand why some chemical reactions are significantly reversible and why such reactions reach a dynamic equilibrium with equal rates of change in both directions. It will also allow us to explore the factors t ...
The Process of Chemical Reactions
... why certain factors speed the reaction up or slow it down. It will help us understand why some chemical reactions are significantly reversible and why such reactions reach a dynamic equilibrium with equal rates of change in both directions. It will also allow us to explore the factors that can push ...
... why certain factors speed the reaction up or slow it down. It will help us understand why some chemical reactions are significantly reversible and why such reactions reach a dynamic equilibrium with equal rates of change in both directions. It will also allow us to explore the factors that can push ...
A Review of using Spray Pyrolysis through Sol-gel
... Received 3 July 2015, revised 30 March 2016, accepted 30 March 2016. ...
... Received 3 July 2015, revised 30 March 2016, accepted 30 March 2016. ...
hong kong diploma of secondary education examination
... each of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the following table : A Both statements are true and the 2nd statement is a correct explanat ...
... each of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the following table : A Both statements are true and the 2nd statement is a correct explanat ...
Chemical Equilibrium Equilibrium A state where the reactants and
... The tendency for a reaction to occur is indicated by the __________ of the equilibrium constant. A value of K much _________ than 1 means that at equilibrium the reaction will consist mostly of ____________-the equilibrium lies to the _____. (The reaction goes to _________________) A value of ...
... The tendency for a reaction to occur is indicated by the __________ of the equilibrium constant. A value of K much _________ than 1 means that at equilibrium the reaction will consist mostly of ____________-the equilibrium lies to the _____. (The reaction goes to _________________) A value of ...
Chemistry
... aspects of general chemistry. Chemistry is mastered when students make the right connections in three key areas: topics that are related, conceptual reasoning with quantitative work, and the different modes of communicating information. McMurry/Fay’s Chemistry, Sixth Edition breaks through the tradi ...
... aspects of general chemistry. Chemistry is mastered when students make the right connections in three key areas: topics that are related, conceptual reasoning with quantitative work, and the different modes of communicating information. McMurry/Fay’s Chemistry, Sixth Edition breaks through the tradi ...
Follow Along Notes - Jackson County School System
... CO2(g) + H2(g) H2O(g) + CO(g) When H2(g) is mixed with CO2(g) at 2,000 K, equilibrium is achieved according to the equation above. In one experiment, the following equilibrium concentrations were measured. [H2] = 0.20 mol/L [CO2] = 0.30 mol/L [H2O] = [CO] = 0.55 mol/L (a) What is the mole fraction ...
... CO2(g) + H2(g) H2O(g) + CO(g) When H2(g) is mixed with CO2(g) at 2,000 K, equilibrium is achieved according to the equation above. In one experiment, the following equilibrium concentrations were measured. [H2] = 0.20 mol/L [CO2] = 0.30 mol/L [H2O] = [CO] = 0.55 mol/L (a) What is the mole fraction ...
The ontological autonomy of the chemical world - Philsci
... case to case, authors generally agree on considering that chemical concepts and descriptions cannot be derived from the concepts and the laws of physics as the epistemological reductionist supposes. For instance, Vemulapalli and Byerly (1999) claim that epistemological reduction fails even in relati ...
... case to case, authors generally agree on considering that chemical concepts and descriptions cannot be derived from the concepts and the laws of physics as the epistemological reductionist supposes. For instance, Vemulapalli and Byerly (1999) claim that epistemological reduction fails even in relati ...
Activation of Alcohols Toward Nucleophilic Substitution: Conversion
... alcohols are converted to saturated alkyl halides.6 Because the use of HCl shows poor results for the conversion of an alcohol to an alkyl chloride, a catalyst such as the zinc used in the Lucas reagent is required. This reaction was improved by adding zinc chloride and had the advantage of milder c ...
... alcohols are converted to saturated alkyl halides.6 Because the use of HCl shows poor results for the conversion of an alcohol to an alkyl chloride, a catalyst such as the zinc used in the Lucas reagent is required. This reaction was improved by adding zinc chloride and had the advantage of milder c ...
Enzymes
... Classwork 2.3: Enzymes • You will have 15 minutes to work on this assignment. • Graded for accuracy! ...
... Classwork 2.3: Enzymes • You will have 15 minutes to work on this assignment. • Graded for accuracy! ...
revised Chemical Kinetics
... the mechanism by which chemical reactions proceed. It provides a rational approach to stabilization of drug products and prediction of shelf- life and optimum storage conditions. e.g. thiamine HCl is most stable at pH 2-3 and is unstable at pH above 6. If this is combined with a buffered vehicle of ...
... the mechanism by which chemical reactions proceed. It provides a rational approach to stabilization of drug products and prediction of shelf- life and optimum storage conditions. e.g. thiamine HCl is most stable at pH 2-3 and is unstable at pH above 6. If this is combined with a buffered vehicle of ...
Predicting Moles of Reactants/Products
... difference between an experimental result and a mathematical result. Using stoichiometry, we can mathematically determine the amount of a product that should be formed during an experiment, yet we sometimes find that we don't end up with exactly the right amount of product. Percentage yield tells us ...
... difference between an experimental result and a mathematical result. Using stoichiometry, we can mathematically determine the amount of a product that should be formed during an experiment, yet we sometimes find that we don't end up with exactly the right amount of product. Percentage yield tells us ...
unit-4-notes-1_enthalpy-and-entropy
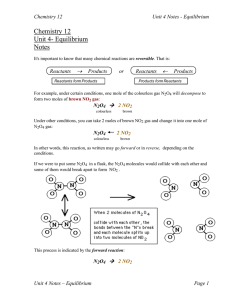
... rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the forward rate, zero. In time, the forward rate would " catch up ". When the rates beca ...
... rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the forward rate, zero. In time, the forward rate would " catch up ". When the rates beca ...
- Vijay Education Academy
... 119. Give equations for the industrial extraction of zinc from calamine. 120. Name the elements contained in anode mud during refining of copper. Why does it contain such elements? 121. What kind of elements are suitable for purification by chromatography? 122. Write the chemical reactions taking pl ...
... 119. Give equations for the industrial extraction of zinc from calamine. 120. Name the elements contained in anode mud during refining of copper. Why does it contain such elements? 121. What kind of elements are suitable for purification by chromatography? 122. Write the chemical reactions taking pl ...
Chapter 11 Chemical Calculations
... there is 1 C atom for every H atom in both of these compound The actual molecular formula of Benzene is C6H6 and that of acetylene is C2H2 so you can see how the actual molecular formula is a multiple of the empirical formula The way McQuarrie explains how to go from a mass % to an empirical formula ...
... there is 1 C atom for every H atom in both of these compound The actual molecular formula of Benzene is C6H6 and that of acetylene is C2H2 so you can see how the actual molecular formula is a multiple of the empirical formula The way McQuarrie explains how to go from a mass % to an empirical formula ...
Experimental and Computational Evidence of Metal‑O2 Activation
... upon exposure to the H2O2 product of enzyme turnover. Previous works have neglected this reaction and assumed that neither CoII nor CuII undergoes a change in redox state during enzyme catalysis.32 The Ered in eq 2 may involve complex internal redox chemistry in certain copper amine oxidases,33−36 w ...
... upon exposure to the H2O2 product of enzyme turnover. Previous works have neglected this reaction and assumed that neither CoII nor CuII undergoes a change in redox state during enzyme catalysis.32 The Ered in eq 2 may involve complex internal redox chemistry in certain copper amine oxidases,33−36 w ...
Notes - Text
... • As the pressure increases, the amount of ammonia present at equilibrium increases. • As the temperature increases, the amount of ammonia at equilibrium decreases. • Can this be predicted? Le Châtelier’s principle: If a system at equilibrium is disturbed by a change in temperature, a change in pres ...
... • As the pressure increases, the amount of ammonia present at equilibrium increases. • As the temperature increases, the amount of ammonia at equilibrium decreases. • Can this be predicted? Le Châtelier’s principle: If a system at equilibrium is disturbed by a change in temperature, a change in pres ...
Balancing Chemical Reactions
... the law of conservation of mass must be taken into account. • The relative amounts of reactants and products represented in the equation must be adjusted so that the numbers and types of atoms are the same on both sides of the equation. ...
... the law of conservation of mass must be taken into account. • The relative amounts of reactants and products represented in the equation must be adjusted so that the numbers and types of atoms are the same on both sides of the equation. ...
Review Unit 8 Test (Chp 15,17)
... increases until it becomes the same as the reverse reaction rate at equilibrium. stays constant before and after equilibrium is reached. A is half true, but “decreases”, decreases to become a constant nonzero rate at equilibrium. not “increases.” decreases to become zero at equilibrium. Greater pres ...
... increases until it becomes the same as the reverse reaction rate at equilibrium. stays constant before and after equilibrium is reached. A is half true, but “decreases”, decreases to become a constant nonzero rate at equilibrium. not “increases.” decreases to become zero at equilibrium. Greater pres ...
Non-native transition metal monoxide nanostructures
... interest in synthetic strategies and mechanisms for new materials but also technological interest in newly emerging unique magnetic and electrochemical properties of these materials. Selective syntheses of colloidal h-CoO and c-CoO Thermal decomposition of a cobalt precursor under an inert atmospher ...
... interest in synthetic strategies and mechanisms for new materials but also technological interest in newly emerging unique magnetic and electrochemical properties of these materials. Selective syntheses of colloidal h-CoO and c-CoO Thermal decomposition of a cobalt precursor under an inert atmospher ...
Syllabus Advanced Level and Advanced Subsidiary Level
... Each section also covers aspects of environmental chemistry. Examination questions will focus on the chemical concepts and techniques rather than on the recall of specific examples. All necessary information about the context will be given in the question and candidates will be expected to apply the ...
... Each section also covers aspects of environmental chemistry. Examination questions will focus on the chemical concepts and techniques rather than on the recall of specific examples. All necessary information about the context will be given in the question and candidates will be expected to apply the ...