Synthesis of PbS Nanoclusters within Block Copolymer Nanoreactors
... films of diblock copolymers containing carboxylic acid units on one of the blocks. Lead was selectively sequestered into the acid containing domains by treating the polymer films with tetraethyllead, and PbS was formed by subsequently treating the films with H2S. The PbS nanoclusters were characteri ...
... films of diblock copolymers containing carboxylic acid units on one of the blocks. Lead was selectively sequestered into the acid containing domains by treating the polymer films with tetraethyllead, and PbS was formed by subsequently treating the films with H2S. The PbS nanoclusters were characteri ...
mass mass calc
... another. For most of you, when working with balanced equations it will be best to work in moles rather than in grams or number of particles so you will need to convert the given information from grams to moles to begin the math. ...
... another. For most of you, when working with balanced equations it will be best to work in moles rather than in grams or number of particles so you will need to convert the given information from grams to moles to begin the math. ...
Determination of Cystein and Methionine by Oscillating Chemical
... 3.1. Influence of experimental variables on the oscillating conductance In order to ensure the maximum possible sensitivity and precision in the determination as well as the ability to perform large series of analyses, working conditions were optimized considering three factors, namely: (i) accompli ...
... 3.1. Influence of experimental variables on the oscillating conductance In order to ensure the maximum possible sensitivity and precision in the determination as well as the ability to perform large series of analyses, working conditions were optimized considering three factors, namely: (i) accompli ...
CO 2 - TrimbleChemistry
... elements of which it is composed • Subscripts are used to indicate how many atoms of a particular element exist in the compound • If there is only one atom of a particular element, the one is assumed ...
... elements of which it is composed • Subscripts are used to indicate how many atoms of a particular element exist in the compound • If there is only one atom of a particular element, the one is assumed ...
Peptide bond formation by aminolysin
... decrease at pH 10.0 was likely the non-enzymatic hydrolysis of the acyl donor (L-Phe-OMe). Therefore, the enzyme reactions of the following experiments were conducted at pH 8.0 as the standard condition to avoid such non-enzymatic hydrolysis of the acyl donor substrates. Next, we evaluated the effec ...
... decrease at pH 10.0 was likely the non-enzymatic hydrolysis of the acyl donor (L-Phe-OMe). Therefore, the enzyme reactions of the following experiments were conducted at pH 8.0 as the standard condition to avoid such non-enzymatic hydrolysis of the acyl donor substrates. Next, we evaluated the effec ...
Thermodynamics: Entropy, Free Energy and the Direction of
... Since Q is < K at both temperatures the reaction will shift right; for 298K there will be a dramatic shift while at 973K the shift will be slight. (c) The nonstandard DG is calculated using DG = DG0 + RTlnQ DG298 = -141.6kJ/mol + (8.314J/mol*K)(kJ/103J)(298K)(ln4.00) DG298 = -138.2kJ/mol DG973 = -12 ...
... Since Q is < K at both temperatures the reaction will shift right; for 298K there will be a dramatic shift while at 973K the shift will be slight. (c) The nonstandard DG is calculated using DG = DG0 + RTlnQ DG298 = -141.6kJ/mol + (8.314J/mol*K)(kJ/103J)(298K)(ln4.00) DG298 = -138.2kJ/mol DG973 = -12 ...
9.2 Oxidation Numbers
... Are these reactions oxidation‑reduction reactions? Are electrons transferred? Simply reading a chemical equation does not always tell us whether oxidation and reduction have occurred, so chemists have developed a numerical system to help identify a reaction as redox. For redox reactions, this system ...
... Are these reactions oxidation‑reduction reactions? Are electrons transferred? Simply reading a chemical equation does not always tell us whether oxidation and reduction have occurred, so chemists have developed a numerical system to help identify a reaction as redox. For redox reactions, this system ...
Solution - gearju.com
... formula or some integral multiple of it (for example, two, three, four, or more times the empirical formula). Comparing the ratio of the molar mass to the molar mass of the empirical formula will show the integral relationship between the empirical and molecular formulas. The molar mass of the empir ...
... formula or some integral multiple of it (for example, two, three, four, or more times the empirical formula). Comparing the ratio of the molar mass to the molar mass of the empirical formula will show the integral relationship between the empirical and molecular formulas. The molar mass of the empir ...
ChemConnections
... same types of atoms in the same physical state, the more atoms per molecule, the more types of motion available to it and, thus, the higher its entropy. (d) 4 mol S2. The two samples contain the same number of sulfur atoms, but different numbers of molecules. Despite the greater complexity of S8 , t ...
... same types of atoms in the same physical state, the more atoms per molecule, the more types of motion available to it and, thus, the higher its entropy. (d) 4 mol S2. The two samples contain the same number of sulfur atoms, but different numbers of molecules. Despite the greater complexity of S8 , t ...
Physical Chemistry: Thermodynamics, Structure, And Change By
... Physical Chemistry, 10/e By Peter Atkins & Julio tenth edition of Atkins' Physical Chemistry even more > Physical Chemistry, 10/e and Biophysical Chemistry Division. Julio De Paula is If searching for the ebook by Peter Atkins;Julio de Paula Physical Chemistry: Thermodynamics, Structure, and Change ...
... Physical Chemistry, 10/e By Peter Atkins & Julio tenth edition of Atkins' Physical Chemistry even more > Physical Chemistry, 10/e and Biophysical Chemistry Division. Julio De Paula is If searching for the ebook by Peter Atkins;Julio de Paula Physical Chemistry: Thermodynamics, Structure, and Change ...
chemical reactions and stoichiometry chemical reactions and
... In chemical reactions, the amount of each element is always conserved. This is consistent with the statements of Dalton’s atomic theory. In addition, the total amount of electrical charge is always conserved. This is the law of conservation of charge. A balanced chemical equation describes a chemica ...
... In chemical reactions, the amount of each element is always conserved. This is consistent with the statements of Dalton’s atomic theory. In addition, the total amount of electrical charge is always conserved. This is the law of conservation of charge. A balanced chemical equation describes a chemica ...
7. A timeline of symbols and signs in chemistry
... its full name, called its symbol. This is, where practicable, the first letter of its Latin name. Simply as abbreviations of the full names symbols and formulae are of great service; this, however, is but a small part of their significance and value in chemistry. Their further use may best be explai ...
... its full name, called its symbol. This is, where practicable, the first letter of its Latin name. Simply as abbreviations of the full names symbols and formulae are of great service; this, however, is but a small part of their significance and value in chemistry. Their further use may best be explai ...
Stoichiometry: Predicting Amounts in Reactions
... Stoichiometry is the process of determining how much product is made or how much reactant is needed during a chemical reaction. As we know, in chemical reactions atoms are conserved. We show thi ...
... Stoichiometry is the process of determining how much product is made or how much reactant is needed during a chemical reaction. As we know, in chemical reactions atoms are conserved. We show thi ...
Electrochemical Degradation of Methanyl Yellow using Carbon/ZnO
... Electrode/ZnO Electrode composition ratio of 5:3:4 have highest percent degradation, but in this research is selected electrodes composition of nanoporous carbon/ZnO and paraffin of 5:2:3 by using the current conductor of silver wire (Ag) because this composition have optimum degradation. Optimizati ...
... Electrode/ZnO Electrode composition ratio of 5:3:4 have highest percent degradation, but in this research is selected electrodes composition of nanoporous carbon/ZnO and paraffin of 5:2:3 by using the current conductor of silver wire (Ag) because this composition have optimum degradation. Optimizati ...
CHEMICAL EQUATIONS - Clayton State University
... Combustion of a 0.2000-g sample of a compound made up of only carbon, hydrogen, and oxygen yields 0.200 g H2O and 0.4880 g CO2. Calculate the mass and mass percentage of each element present in the 0.2000-g sample. - Convert mass H2O/CO2 to moles using molar mass - Determine moles H/C from number of ...
... Combustion of a 0.2000-g sample of a compound made up of only carbon, hydrogen, and oxygen yields 0.200 g H2O and 0.4880 g CO2. Calculate the mass and mass percentage of each element present in the 0.2000-g sample. - Convert mass H2O/CO2 to moles using molar mass - Determine moles H/C from number of ...
CHOICE BASED CREDIT SYSTEM B. Sc. WITH CHEMISTRY
... Strong, moderate and weak electrolytes, degree of ionization, factors affecting degree of ionization, ionization constant and ionic product of water. Ionization of weak acids and bases, pH scale, common ion effect. Salt hydrolysis-calculation of hydrolysis constant, degree of hydrolysis and pH for d ...
... Strong, moderate and weak electrolytes, degree of ionization, factors affecting degree of ionization, ionization constant and ionic product of water. Ionization of weak acids and bases, pH scale, common ion effect. Salt hydrolysis-calculation of hydrolysis constant, degree of hydrolysis and pH for d ...
Chapter 19 Chemical Thermodynamics
... • Therefore, the total energy of the universe is a constant. • Energy can, however, be converted from one form to another or transferred from a system to the surroundings or ...
... • Therefore, the total energy of the universe is a constant. • Energy can, however, be converted from one form to another or transferred from a system to the surroundings or ...
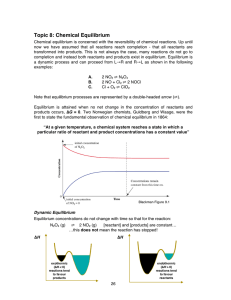
Topic 8: Chemical Equilibrium
... If concentration increases, the system acts to consume some of it If concentration decreases, the system acts to produce some of it 2. Change in pressure The pressure of a system can be changed in three ways: • Add or remove a gaseous reactant or product (same as changing concentration) • Add an ine ...
... If concentration increases, the system acts to consume some of it If concentration decreases, the system acts to produce some of it 2. Change in pressure The pressure of a system can be changed in three ways: • Add or remove a gaseous reactant or product (same as changing concentration) • Add an ine ...
Untitled
... All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without either the prior written permission of the publisher or a licence permitting restricted ...
... All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without either the prior written permission of the publisher or a licence permitting restricted ...
Problem 5. Inorganic chains and rings
... Standard pressure, p = 1 bar = 105 Pa Zero of the Celsius scale, 273.15 K 1 nanometer (nm) = 10–9 m ...
... Standard pressure, p = 1 bar = 105 Pa Zero of the Celsius scale, 273.15 K 1 nanometer (nm) = 10–9 m ...
File
... law of conservation of mass is observed. Does Determine if the _____________________________ the mass of the reactants equal the mass of the products? Yes ___________. 3. Evaluate the Answer Each product or reactant has 4 significant figures. Your answer must have 4 significant figures. ...
... law of conservation of mass is observed. Does Determine if the _____________________________ the mass of the reactants equal the mass of the products? Yes ___________. 3. Evaluate the Answer Each product or reactant has 4 significant figures. Your answer must have 4 significant figures. ...