Reactions Unit Plan
... B. Apply the Law of Conservation of Mass to writing and balancing chemical equations. (MOCLE 1.1.I.a, b) 1. Represent a chemical equation in words and formulas. 2. Balance a chemical equation using the Law of Conservation of Mass. ...
... B. Apply the Law of Conservation of Mass to writing and balancing chemical equations. (MOCLE 1.1.I.a, b) 1. Represent a chemical equation in words and formulas. 2. Balance a chemical equation using the Law of Conservation of Mass. ...
Chemical Reactions
... The ___________ of which a new substances forms is called the ___________ of __________________. Different chemical substances will produce _______________ at _________________ rates. Factor that affects rate of reaction Temperature ...
... The ___________ of which a new substances forms is called the ___________ of __________________. Different chemical substances will produce _______________ at _________________ rates. Factor that affects rate of reaction Temperature ...
Document
... Three moles of 1-propanol, C3H7OH, reacts with one mole of phosphorus trichloride to produce 1-chloropropane, C3H7Cl, and phosphorus acid, H3PO3. What is the percent yield if you begin with 75.0 g of both 1propanol and phosphorus trichloride and obtain 1.0 mole of 1-chloropropane? (1propanol= 60.10 ...
... Three moles of 1-propanol, C3H7OH, reacts with one mole of phosphorus trichloride to produce 1-chloropropane, C3H7Cl, and phosphorus acid, H3PO3. What is the percent yield if you begin with 75.0 g of both 1propanol and phosphorus trichloride and obtain 1.0 mole of 1-chloropropane? (1propanol= 60.10 ...
Slide 1 - Mrs. Reed Science Classes
... For the reaction represented by the equation Mg + 2HCl H2 + MgCl2, calculate the percentage yield of magnesium chloride if 100. g of magnesium react with excess hydrochloric acid to yield 330. g of magnesium chloride. a. 71.8% c. 81.6% b. 74.3% d. 84.2% ...
... For the reaction represented by the equation Mg + 2HCl H2 + MgCl2, calculate the percentage yield of magnesium chloride if 100. g of magnesium react with excess hydrochloric acid to yield 330. g of magnesium chloride. a. 71.8% c. 81.6% b. 74.3% d. 84.2% ...
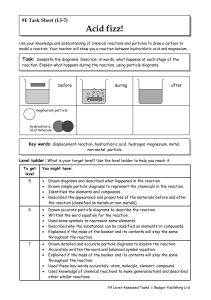
Year 9 Homework Task 9E-5 Reactions 5-7
... Drawn detailed and accurate particle diagrams to explain the reaction. Accurately written the word and balanced symbol equation. Explained if the mass of the beaker and its contents will stay the same throughout the reaction. Used these key words accurately: atom, molecule, element, compound. Used k ...
... Drawn detailed and accurate particle diagrams to explain the reaction. Accurately written the word and balanced symbol equation. Explained if the mass of the beaker and its contents will stay the same throughout the reaction. Used these key words accurately: atom, molecule, element, compound. Used k ...
C:\Users\mrh70950\Documents\My Files\WordPerfect
... Copyright© 2012 by Martin Hulce. All rights reserved. Except as permitted under the United States Copyright Act of 1976, no part of this publication may be reproduced or distributed in any form or by any means, or stored in a data base or retrieval system, without the prior permission of the copyrig ...
... Copyright© 2012 by Martin Hulce. All rights reserved. Except as permitted under the United States Copyright Act of 1976, no part of this publication may be reproduced or distributed in any form or by any means, or stored in a data base or retrieval system, without the prior permission of the copyrig ...
Chapter 5 – Chemical Reactions
... A chemical equation tells you what compounds you start with and what compounds you end up with. ...
... A chemical equation tells you what compounds you start with and what compounds you end up with. ...
Stoichiometry Review Package Answer Key
... Make use of the material in your workbook. Attempt all the practice problems for the sections. The section review questions are a valuable resource. The test covers section 4.1, 4.2, enthalpy notation in 4.4 (recognizing whether a reaction is exothermic or endothermic from the energy term associated ...
... Make use of the material in your workbook. Attempt all the practice problems for the sections. The section review questions are a valuable resource. The test covers section 4.1, 4.2, enthalpy notation in 4.4 (recognizing whether a reaction is exothermic or endothermic from the energy term associated ...
Synthesis/Decomposition Reactions
... Essential Content and Skills: How do you correctly identify, balance and predict the product(s) of synthesis reactions? How do you correctly identify, balance and predict the product(s) of decomposition reactions? ...
... Essential Content and Skills: How do you correctly identify, balance and predict the product(s) of synthesis reactions? How do you correctly identify, balance and predict the product(s) of decomposition reactions? ...
Lesson 6
... 6. List the three main indicators that a chemical reaction has occurred. 7. Are there any other indicators of a chemical reaction? 8. What evidence is there that a reaction has occurred in Figure 2.6? Turn to page 29 of the textbook and read "Combustion and Neutralization." 9. A chemical reaction in ...
... 6. List the three main indicators that a chemical reaction has occurred. 7. Are there any other indicators of a chemical reaction? 8. What evidence is there that a reaction has occurred in Figure 2.6? Turn to page 29 of the textbook and read "Combustion and Neutralization." 9. A chemical reaction in ...
Section 2-4 “Chemical Reactions and Enzymes”
... What happens when bonds break and bonds form? Energy must be added to break bonds that hold the reactant molecules together. This is called activation energy (Ae). This amount of energy is what “activates” or gets the reaction started. Once the bonds are broken, the atoms are freed up and can make n ...
... What happens when bonds break and bonds form? Energy must be added to break bonds that hold the reactant molecules together. This is called activation energy (Ae). This amount of energy is what “activates” or gets the reaction started. Once the bonds are broken, the atoms are freed up and can make n ...
1 Types of Chemical Reactions
... substances with different physical and chemical properties are formed. Chemical reactions can be grouped into categories depending on the nature of the chemical reaction. For example during a decomposition reaction a single substance can break apart into several different substances. ...
... substances with different physical and chemical properties are formed. Chemical reactions can be grouped into categories depending on the nature of the chemical reaction. For example during a decomposition reaction a single substance can break apart into several different substances. ...
Ch 5.1 The Nature of Chemical Reactions
... • Describe difference between endothermic and exothermic reactions ...
... • Describe difference between endothermic and exothermic reactions ...
CHEMISTry is life - World of Teaching
... -Too often kids get to high school chemistry and they are scared before they even begin. -My goal is to shape a positive image in their minds about chemistry so that they can be more prepared mentally for high school. -I will do this by showing them how applicable chemistry is to every day life. It ...
... -Too often kids get to high school chemistry and they are scared before they even begin. -My goal is to shape a positive image in their minds about chemistry so that they can be more prepared mentally for high school. -I will do this by showing them how applicable chemistry is to every day life. It ...
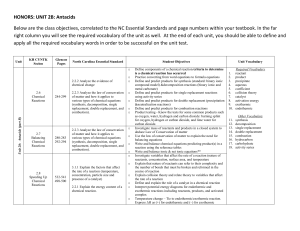
HONORS: UNIT 2B: Antacids Below are the class objectives
... Below are the class objectives, correlated to the NC Essential Standards and page numbers within your textbook. In the far right column you will see the required vocabulary of the unit as well. At the end of each unit, you should be able to define and apply all the required vocabulary words in order ...
... Below are the class objectives, correlated to the NC Essential Standards and page numbers within your textbook. In the far right column you will see the required vocabulary of the unit as well. At the end of each unit, you should be able to define and apply all the required vocabulary words in order ...
TERM 2 Unit 3 YR 9 SCI It is elementary
... understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the periodic table. They recognise that new substances are forme ...
... understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the periodic table. They recognise that new substances are forme ...
Chemistry lesson note
... composition, properties and uses of matter. It also probes the principles governing the changes that matter undergoes ...
... composition, properties and uses of matter. It also probes the principles governing the changes that matter undergoes ...
Ch 19 test_take-home
... A) the reverse process is spontaneous but the forward process is not B) the forward and the reverse processes are both spontaneous C) the forward process is spontaneous but the reverse process is not D) the process is not spontaneous in either direction E) both forward and reverse processes have sto ...
... A) the reverse process is spontaneous but the forward process is not B) the forward and the reverse processes are both spontaneous C) the forward process is spontaneous but the reverse process is not D) the process is not spontaneous in either direction E) both forward and reverse processes have sto ...
Industrial Chemicals Technology Hand Book
... investment in the chemical sector. The chemical industry comprises the companies that produce industrial chemicals. Chemicals are used to make a wide variety of consumer goods, as well as thousands inputs to manufacturing, construction, and service industries. The applications of industrial chemical ...
... investment in the chemical sector. The chemical industry comprises the companies that produce industrial chemicals. Chemicals are used to make a wide variety of consumer goods, as well as thousands inputs to manufacturing, construction, and service industries. The applications of industrial chemical ...
An Efficient Synthetic Route to Glycoamino Acid Building Blocks for
... by LC-MS(ESI) confirmed their structure. ...
... by LC-MS(ESI) confirmed their structure. ...
2.4 Chemical Reactions and Enzymes
... Energy needed to get a reaction going is the activation ...
... Energy needed to get a reaction going is the activation ...