CHEMISTRY 1 FINAL EXAM REVIEW
... 1.) Identify the type of reaction for those listed below: A. a reaction in which a single compound is broken down into simpler substances B. a reaction in which oxygen reacts with another substance, often producing heat or light C. a reaction in which the atoms of one element replace the atoms of a ...
... 1.) Identify the type of reaction for those listed below: A. a reaction in which a single compound is broken down into simpler substances B. a reaction in which oxygen reacts with another substance, often producing heat or light C. a reaction in which the atoms of one element replace the atoms of a ...
one
... – Exothermic (releases energy to the surroundings) – Endothermic (absorbs energy from the surroundings) ...
... – Exothermic (releases energy to the surroundings) – Endothermic (absorbs energy from the surroundings) ...
Notes for Types of Reactions:
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
Chemistry Final Exam Test Yourself I
... (g), the equilibrium constant at a particular temperature is 0.050. The concentrations of each substance are the following: [N2] = 0.5 M, [O2] = 1.7 M, [NO] = 0.010 M. Which way will the reaction shift so as to achieve equilibrium? (Q = 1 x 10-4 Reaction will shift to the right) ...
... (g), the equilibrium constant at a particular temperature is 0.050. The concentrations of each substance are the following: [N2] = 0.5 M, [O2] = 1.7 M, [NO] = 0.010 M. Which way will the reaction shift so as to achieve equilibrium? (Q = 1 x 10-4 Reaction will shift to the right) ...
Introduction_to_Geochemistry_Pre-Lecture_Quiz
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
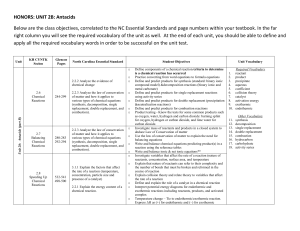
HONORS: UNIT 2B: Antacids Below are the class objectives
... reaction using the reference tables Write and balance ionic & net ionic equations** Investigate variables that affect the rate of a reaction (nature of reactants, concentration, surface area, and temperature Explain that nature of reactants can refer to their complexity and the number of bonds that ...
... reaction using the reference tables Write and balance ionic & net ionic equations** Investigate variables that affect the rate of a reaction (nature of reactants, concentration, surface area, and temperature Explain that nature of reactants can refer to their complexity and the number of bonds that ...
8th Grade Ch. 7 Chemical Reactions Study guide
... Identify the choice that best completes the statement or answers the question. ____ 11. Evidence that a chemical change has occurred includes a ____. A. change in state B. change in size C. change in shape D. change in color ____ 12. You can slow down the rate of a reaction by ____. A. increasing co ...
... Identify the choice that best completes the statement or answers the question. ____ 11. Evidence that a chemical change has occurred includes a ____. A. change in state B. change in size C. change in shape D. change in color ____ 12. You can slow down the rate of a reaction by ____. A. increasing co ...
Balancing Single Replacement Reactions - Kossmann
... Name: __________________________ Class: ____________________ Date: _____________ ...
... Name: __________________________ Class: ____________________ Date: _____________ ...
52.
... 11) For a gas phase SN2 reaction, the intrinsic barrier associated with X is defined as the energy difference between the ion-molecule cluster [X.*.CH3X]- and the transition state [X*.CH3**X]-. In the present work, these energies have been obtained at the 4-31G level, with full optimization of all s ...
... 11) For a gas phase SN2 reaction, the intrinsic barrier associated with X is defined as the energy difference between the ion-molecule cluster [X.*.CH3X]- and the transition state [X*.CH3**X]-. In the present work, these energies have been obtained at the 4-31G level, with full optimization of all s ...
Chem 400 Chem 150 REVIEW SHEET Amanda R
... Atoms, Molecules, Ions – fundamentals of elements o Protons, electrons and neutrons make up an atom o Atoms make up molecules, all matter is made of atoms o Protons and neutrons are in the nucleus, and electrons are buzzing outside the nucleus around the nucleus in orbitals o # of protons defines an ...
... Atoms, Molecules, Ions – fundamentals of elements o Protons, electrons and neutrons make up an atom o Atoms make up molecules, all matter is made of atoms o Protons and neutrons are in the nucleus, and electrons are buzzing outside the nucleus around the nucleus in orbitals o # of protons defines an ...
Synthesis/Decomposition Reactions
... Essential Content and Skills: How do you correctly identify, balance and predict the product(s) of synthesis reactions? How do you correctly identify, balance and predict the product(s) of decomposition reactions? ...
... Essential Content and Skills: How do you correctly identify, balance and predict the product(s) of synthesis reactions? How do you correctly identify, balance and predict the product(s) of decomposition reactions? ...
Form A 1 Chem 130 Name______________________________
... 8. For the reaction, 2 NO(g) + Cl2(g) → 2 NOCl(g), Ho = -40.9 kJ. At what temperatures do you expect the reaction to be spontaneous: high, low, all, or none? Justify your answer. If we look at the reaction, there are three moles of gas on the reactant side and only two moles of gas on the product s ...
... 8. For the reaction, 2 NO(g) + Cl2(g) → 2 NOCl(g), Ho = -40.9 kJ. At what temperatures do you expect the reaction to be spontaneous: high, low, all, or none? Justify your answer. If we look at the reaction, there are three moles of gas on the reactant side and only two moles of gas on the product s ...
Kinetics of the Selective Reaction of Diazonium Salts with Single
... electron w ithdraw ing diazonium salts, w here metallic nanotubes are preferentially functionalised due to their higher electron density at the Fermi level.1 Since this reaction requires the nanotubes to be individualised, it is usually carried out in a surfactant stabilised dispersion. Despite the ...
... electron w ithdraw ing diazonium salts, w here metallic nanotubes are preferentially functionalised due to their higher electron density at the Fermi level.1 Since this reaction requires the nanotubes to be individualised, it is usually carried out in a surfactant stabilised dispersion. Despite the ...
Kinetics & Equilibrium
... THE ENERGY OF THE PROCESS MUST DECREASE AND THE UNIVERSE MUST BECOME MORE RANDOM!!!! ...
... THE ENERGY OF THE PROCESS MUST DECREASE AND THE UNIVERSE MUST BECOME MORE RANDOM!!!! ...
Introduction to Chemistry
... Ionic- Two elements bond by transferring electrons to create ions that attract together (+ is attracted to - after an electron is transferred) ...
... Ionic- Two elements bond by transferring electrons to create ions that attract together (+ is attracted to - after an electron is transferred) ...
1 Q. If ΔrH is positive, what can you say about the reaction? 2 Q If
... and products, what happens in a reaction which is in dynamic equilibrium? ...
... and products, what happens in a reaction which is in dynamic equilibrium? ...
Sample Paper - Army Public School Jammu Cantt
... 6. Will the elevation in boiling point be same if 0.1 mol of Sodium chloride or 0.1 mol of sugar is dissolved in 1L of water? Explain. 7. The following curve is obtained when molar conductivity ( m ) is plotted against the square root of concentration, c1/2 for two electrolytes A and B ...
... 6. Will the elevation in boiling point be same if 0.1 mol of Sodium chloride or 0.1 mol of sugar is dissolved in 1L of water? Explain. 7. The following curve is obtained when molar conductivity ( m ) is plotted against the square root of concentration, c1/2 for two electrolytes A and B ...
Solution
... 19) Novocaine can be used as a local anesthetic, and has a pKb of 5.00. What is the ratio of novocaine to its conjugate acid if a small amount is added to the blood, which has a pH of approximately 7? A) 7/5 B) 5/7 C) 1/1 D) 1/100 E) 100/1 For questions 20-23, choose from the following graphs to an ...
... 19) Novocaine can be used as a local anesthetic, and has a pKb of 5.00. What is the ratio of novocaine to its conjugate acid if a small amount is added to the blood, which has a pH of approximately 7? A) 7/5 B) 5/7 C) 1/1 D) 1/100 E) 100/1 For questions 20-23, choose from the following graphs to an ...
Exam 2 Fall 2005 Chemsitry 1211
... EXAM #2 Version 1 This exam is twenty five questions long. Each question is worth 4 points. Please read through all of the questions first and ask about anything you do not understand. You will have one hour and 15 minutes to complete this exam. Exams will be picked up at the end of the class period ...
... EXAM #2 Version 1 This exam is twenty five questions long. Each question is worth 4 points. Please read through all of the questions first and ask about anything you do not understand. You will have one hour and 15 minutes to complete this exam. Exams will be picked up at the end of the class period ...
Chemical Reactions
... that speeds up the rate of a chemical reaction. Catalysts work by lowering a reaction’s activation energy. In an enzyme-catalyzed reaction, the reactants are known as substrates. Substrates bind to a part of an enzyme called the active site and remain bound to the enzyme until the reaction is comple ...
... that speeds up the rate of a chemical reaction. Catalysts work by lowering a reaction’s activation energy. In an enzyme-catalyzed reaction, the reactants are known as substrates. Substrates bind to a part of an enzyme called the active site and remain bound to the enzyme until the reaction is comple ...
09 Stoichiometry WS Stoichiometry WS
... b. If 0.905 mol Al2O3 is produced in the reaction, what mass of Fe is produced? c. How many moles of Fe2O3 will react with 99.0 g of Al? 3. The reaction N2(g) + 3H2(g) 2NH3(g) is used to produce ammonia commercially. If 1.40g of N2 are used in the reaction, how many grams of H2 will be needed? 4. Wh ...
... b. If 0.905 mol Al2O3 is produced in the reaction, what mass of Fe is produced? c. How many moles of Fe2O3 will react with 99.0 g of Al? 3. The reaction N2(g) + 3H2(g) 2NH3(g) is used to produce ammonia commercially. If 1.40g of N2 are used in the reaction, how many grams of H2 will be needed? 4. Wh ...
Answer Key to Sample Questions
... positive because one molecule breaks to form two molecules b. What is the sign of H for this reaction? positive because a bond is broken, but none is formed. c. In which temperature range will this reaction be thermodynamically favored? It is entropy favored, enthalpy disfavored, so favored overall ...
... positive because one molecule breaks to form two molecules b. What is the sign of H for this reaction? positive because a bond is broken, but none is formed. c. In which temperature range will this reaction be thermodynamically favored? It is entropy favored, enthalpy disfavored, so favored overall ...
Bioorthogonal chemistry
The term bioorthogonal chemistry refers to any chemical reaction that can occur inside of living systems without interfering with native biochemical processes. The term was coined by Carolyn R. Bertozzi in 2003. Since its introduction, the concept of the bioorthogonal reaction has enabled the study of biomolecules such as glycans, proteins, and lipids in real time in living systems without cellular toxicity. A number of chemical ligation strategies have been developed that fulfill the requirements of bioorthogonality, including the 1,3-dipolar cycloaddition between azides and cyclooctynes (also termed copper-free click chemistry), between nitrones and cyclooctynes, oxime/hydrazone formation from aldehydes and ketones, the tetrazine ligation, the isocyanide-based click reaction, and most recently, the quadricyclane ligation.The use of bioorthogonal chemistry typically proceeds in two steps. First, a cellular substrate is modified with a bioorthogonal functional group (chemical reporter) and introduced to the cell; substrates include metabolites, enzyme inhibitors, etc. The chemical reporter must not alter the structure of the substrate dramatically to avoid affecting its bioactivity. Secondly, a probe containing the complementary functional group is introduced to react and label the substrate.Although effective bioorthogonal reactions such as copper-free click chemistry have been developed, development of new reactions continues to generate orthogonal methods for labeling to allow multiple methods of labeling to be used in the same biosystems.