5. Quantum mechanics of chemical binding
... Quantum mechanics can not say more, but this is mathematically a perfect explanation. For chemists there are of course other explanations based on approximate models. How can we approximate the orbitals? Simplest is to start with atoms and approximate the molecular orbital by linear combination: ...
... Quantum mechanics can not say more, but this is mathematically a perfect explanation. For chemists there are of course other explanations based on approximate models. How can we approximate the orbitals? Simplest is to start with atoms and approximate the molecular orbital by linear combination: ...
Document
... • Decoupling of electronic and nuclear motion • Electrons are much lighter hence adapt ‘instantaneously’ to movement of nuclei (cows and flies) • More formally, gradients of with respect to R are neglected ...
... • Decoupling of electronic and nuclear motion • Electrons are much lighter hence adapt ‘instantaneously’ to movement of nuclei (cows and flies) • More formally, gradients of with respect to R are neglected ...
QUANTUM CHEMISTRY Model 1: Light and Waves Critical thinking
... Model 3: Atomic Orbitals and Quantum Numbers The wave functions for electrons in atoms are given the special name ‘atomic orbitals’. As explored in worksheet 1, the energy levels of hydrogen-like (one-electron) atoms are determined by a single quantum number, n. For other atoms, more quantities are ...
... Model 3: Atomic Orbitals and Quantum Numbers The wave functions for electrons in atoms are given the special name ‘atomic orbitals’. As explored in worksheet 1, the energy levels of hydrogen-like (one-electron) atoms are determined by a single quantum number, n. For other atoms, more quantities are ...
Superconcepts
... x. Schrödinger showed that atomic wave spectra (electrons) could be predicted by wave functions in 1926. xi. Heisinger found that atomic properties were ‘indeterminant’ or uncertain in 1927. xii. When the double-slit experiment was repeated with electrons, the results confirmed that electrons behave ...
... x. Schrödinger showed that atomic wave spectra (electrons) could be predicted by wave functions in 1926. xi. Heisinger found that atomic properties were ‘indeterminant’ or uncertain in 1927. xii. When the double-slit experiment was repeated with electrons, the results confirmed that electrons behave ...
Quantum Chemical Simulations and Descriptors
... Hamiltonian is replaced with a model one; the parameters of the model Hamiltonian are fitted to reproduce the reference data (usually experiments) of Let’s call the “real semi-empirical methods” (as opposed to the tight-binding methods) the methods which are close in spirit to the Hartree-Fock forma ...
... Hamiltonian is replaced with a model one; the parameters of the model Hamiltonian are fitted to reproduce the reference data (usually experiments) of Let’s call the “real semi-empirical methods” (as opposed to the tight-binding methods) the methods which are close in spirit to the Hartree-Fock forma ...
Document
... Prove that, given a pair of normalized but not orthogonal functions ψ1 and ψ2, the function ψ3 = ψ2 – Sψ1 is orthogonal to ψ1 if S is the overlap integral of ψ1 and ψ2. Is ψ3 normalized? (Use the back of the page if necessary). ...
... Prove that, given a pair of normalized but not orthogonal functions ψ1 and ψ2, the function ψ3 = ψ2 – Sψ1 is orthogonal to ψ1 if S is the overlap integral of ψ1 and ψ2. Is ψ3 normalized? (Use the back of the page if necessary). ...
The Modern Atomic Model
... Bohr Model of the Atom (review) •Energy levels contain electrons. •Electrons travel around the nucleus. •Different orbitals varied by different quantum (energy). •Gaps between energy levels were not equal. ...
... Bohr Model of the Atom (review) •Energy levels contain electrons. •Electrons travel around the nucleus. •Different orbitals varied by different quantum (energy). •Gaps between energy levels were not equal. ...
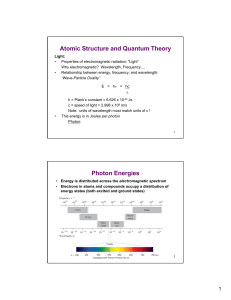
Atomic Structure and Quantum Theory Photon Energies
... – Electrons occupy specific (quantized) levels in an atom – Electrons have properties of BOTH particles and waves – At any instant, it is impossible to pinpoint the position of an electron of a given energy with high accuracy. Because of this combination, the best we can do is calculate the probabil ...
... – Electrons occupy specific (quantized) levels in an atom – Electrons have properties of BOTH particles and waves – At any instant, it is impossible to pinpoint the position of an electron of a given energy with high accuracy. Because of this combination, the best we can do is calculate the probabil ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 2. Prove explicitly that the momentum operator is a self-adjoint operator. 3. Write down the ground state energy eigenfunction of a simple harmonic oscillator? Sketch its graph. 4. Define the parity operator by its effect on a wave function. What are its eigenvalues? 5. If A is any Hermitian operato ...
... 2. Prove explicitly that the momentum operator is a self-adjoint operator. 3. Write down the ground state energy eigenfunction of a simple harmonic oscillator? Sketch its graph. 4. Define the parity operator by its effect on a wave function. What are its eigenvalues? 5. If A is any Hermitian operato ...
Exam #2 Answers to Definitions (Problem #1)
... a) Born-Oppenheimer approximation assumes that the nuclei are stationary, and that electron motion can be treated separately. Fixed bond distances and bond angles are assumed, and a Hamiltonian operator is written for electronic motion only. b) LCAO-MO method for constucting approximate molecular el ...
... a) Born-Oppenheimer approximation assumes that the nuclei are stationary, and that electron motion can be treated separately. Fixed bond distances and bond angles are assumed, and a Hamiltonian operator is written for electronic motion only. b) LCAO-MO method for constucting approximate molecular el ...
NAME PERIOD ______ DATE Chapter 5 Sec. 2
... NAME _______________________________________________ ...
... NAME _______________________________________________ ...
Modern Model of the Atom Student Notes and Assignment
... 1. AUFBAU PRINCIPLE - electrons enter orbitals of the lowest energy levels first 2. PAULI EXCLUSION PRINCIPLE - an atomic orbital may hold at most two electrons. Each must have an opposite spin. 3. HUND’S RULE - when electrons occupy orbitals of equal energy one electron enters each orbital until al ...
... 1. AUFBAU PRINCIPLE - electrons enter orbitals of the lowest energy levels first 2. PAULI EXCLUSION PRINCIPLE - an atomic orbital may hold at most two electrons. Each must have an opposite spin. 3. HUND’S RULE - when electrons occupy orbitals of equal energy one electron enters each orbital until al ...
Midterm Exam 2
... Section 1- Concepts and Definitions (50% 5 points each) 1) Give two examples of a hydrogenic atom other than hydrogen (H): ...
... Section 1- Concepts and Definitions (50% 5 points each) 1) Give two examples of a hydrogenic atom other than hydrogen (H): ...
Semiclassical calculation of electron spectra in atoms through two
... The features of the one-electron spectra in the spherically symmetric self-consistent attraction potentials have been under study in the paper [1] (also, reviews [2, 3]). Specifically, the potentials with the Coulomb singularity have been there considered. As is well known, the screening of the Coul ...
... The features of the one-electron spectra in the spherically symmetric self-consistent attraction potentials have been under study in the paper [1] (also, reviews [2, 3]). Specifically, the potentials with the Coulomb singularity have been there considered. As is well known, the screening of the Coul ...
HW-1-Ch1-Atomic-structure-W16
... 11. Why is comic abundance different from terrestrial abundances? ...
... 11. Why is comic abundance different from terrestrial abundances? ...
Quantum numbers
... • Carbon: (1s2) 2s2, 2p2; Sulfur: (…), 3s2, 3p4 • Homework: write down the electron configurations of N, O, Cl why do halogens (X) form X2 in the gas phase? why do the alkali metals (Li, Na, ….) do so too? ...
... • Carbon: (1s2) 2s2, 2p2; Sulfur: (…), 3s2, 3p4 • Homework: write down the electron configurations of N, O, Cl why do halogens (X) form X2 in the gas phase? why do the alkali metals (Li, Na, ….) do so too? ...
Atomic Orbitals - Daytona State College
... An atomic orbital is a mathematical function that describes the wave-like behavior of either one electron or a pair of electrons in an atom. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. The term may also r ...
... An atomic orbital is a mathematical function that describes the wave-like behavior of either one electron or a pair of electrons in an atom. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. The term may also r ...
fulltext - DiVA portal
... the computional efforts and time, can be reduced. This chapter gives an introduction to group theory. For further reading, see Ref. [4] A symmetry operator is an operator that acts on the molecule in such a way that the atomic positions is physically indistinguishable from the original ones. All sym ...
... the computional efforts and time, can be reduced. This chapter gives an introduction to group theory. For further reading, see Ref. [4] A symmetry operator is an operator that acts on the molecule in such a way that the atomic positions is physically indistinguishable from the original ones. All sym ...
KS-DFT formalism
... Our choice of wave functions is very limited; we only know how to use independent particle wave functions. The degree to which this limitation has invaded our thinking is marked by our constant use of concepts which have meaning only in terms of independent particle wave functions: shell structure, ...
... Our choice of wave functions is very limited; we only know how to use independent particle wave functions. The degree to which this limitation has invaded our thinking is marked by our constant use of concepts which have meaning only in terms of independent particle wave functions: shell structure, ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 20. While the order is the same for both C3v and C3h point groups, their classes are different reason out. 21. Allene belongs to D2d point group. Identify the order and hence the number and dimensionality of the irreducible representations. 22. Set up the secular determinant for allyl radical and ob ...
... 20. While the order is the same for both C3v and C3h point groups, their classes are different reason out. 21. Allene belongs to D2d point group. Identify the order and hence the number and dimensionality of the irreducible representations. 22. Set up the secular determinant for allyl radical and ob ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... For the wave function Ψ(φ) = Ae , where m is an integer, for 0≤φ≤2π. Determine A so that the wave function is normalized. Give the Laplacian operator in spherical polar coordinates. What are their limits of integration? CO absorbs energy in the microwave region of the spectrum at 1.93 x 1012 Hz. Thi ...
... For the wave function Ψ(φ) = Ae , where m is an integer, for 0≤φ≤2π. Determine A so that the wave function is normalized. Give the Laplacian operator in spherical polar coordinates. What are their limits of integration? CO absorbs energy in the microwave region of the spectrum at 1.93 x 1012 Hz. Thi ...
ppt
... Since electrons are the fermions they obey Fermi-Dirac statistics, i.e. they cannot be more than two electrons (with opposite spins) occupying given non-degenerate level. If level is N-fold degenerate (for example, p level is 3 fold degenerate) then it can accomondate 2N electrons. So we can now fil ...
... Since electrons are the fermions they obey Fermi-Dirac statistics, i.e. they cannot be more than two electrons (with opposite spins) occupying given non-degenerate level. If level is N-fold degenerate (for example, p level is 3 fold degenerate) then it can accomondate 2N electrons. So we can now fil ...
Hund`s multiplicity rule: From atoms to quantum dots
... In 1965, Davidson has shown that the textbook explanation for the Hund’s multiplicity rule in atoms, based on the Pauli principle, is wrong. The reason for the failure of the textbook proof, as has been given later by others and as appears today in modern textbooks, it is based on the need to introd ...
... In 1965, Davidson has shown that the textbook explanation for the Hund’s multiplicity rule in atoms, based on the Pauli principle, is wrong. The reason for the failure of the textbook proof, as has been given later by others and as appears today in modern textbooks, it is based on the need to introd ...
Jim Greer
... many-electron wave functions [1,2] and has been applied to a variety of molecular problems. Recently using this Monte Carlo configuration interaction (MCCI) method, singlet and triplet electronic excitation energies have been calculated for few electron molecules for systems with single and multi-re ...
... many-electron wave functions [1,2] and has been applied to a variety of molecular problems. Recently using this Monte Carlo configuration interaction (MCCI) method, singlet and triplet electronic excitation energies have been calculated for few electron molecules for systems with single and multi-re ...