Chemistry Post-Enrolment Worksheet C
... of an oxygen atom and a hydrogen atom and has an overall 1- charge. Brackets can be used if we need more than one of a polyatomic ion in our formula. e.g. ammonium carbonate is made up of ammonium ions, NH4+ , and carbonate ions, CO32In order for the charges to balance we need two ammonium ions for ...
... of an oxygen atom and a hydrogen atom and has an overall 1- charge. Brackets can be used if we need more than one of a polyatomic ion in our formula. e.g. ammonium carbonate is made up of ammonium ions, NH4+ , and carbonate ions, CO32In order for the charges to balance we need two ammonium ions for ...
FXM Rev 1 Key - Grande Cache Community High School
... positively charged nucleus with electrons in orbit. It is sometimes called the Planetary Atomic Model. hydrocarbons These are organic compounds that contain both carbon and hydrogen. Methane (CH4) is an example. Avogadro’s number This is a number that groups a very large amount of atoms or molecules ...
... positively charged nucleus with electrons in orbit. It is sometimes called the Planetary Atomic Model. hydrocarbons These are organic compounds that contain both carbon and hydrogen. Methane (CH4) is an example. Avogadro’s number This is a number that groups a very large amount of atoms or molecules ...
can be determined without changing the identity of matter
... substances using physical or chemical means - Elements are the building blocks of chemistry! They are the simple things from which all other things are formed! ...
... substances using physical or chemical means - Elements are the building blocks of chemistry! They are the simple things from which all other things are formed! ...
Lecture 3 Chemistry
... To understand the way living things function, it is necessary to understand basic chemical processes ...
... To understand the way living things function, it is necessary to understand basic chemical processes ...
CH30S Chemical Reactions Part 2 Unit Review
... 14. Calculate the number of molecules in 179.1g of iron(III) hydrogen carbonate. (4.51x1023 molecules) 15. What volume is occupied by 1.25mol of He gas at STP? (28L) 16. Determine the density of hydrogen gas in g/ml. Hint: use molar mass and molar volume. (9.02x10-5g/mL) 17. Calculate the mass of 8. ...
... 14. Calculate the number of molecules in 179.1g of iron(III) hydrogen carbonate. (4.51x1023 molecules) 15. What volume is occupied by 1.25mol of He gas at STP? (28L) 16. Determine the density of hydrogen gas in g/ml. Hint: use molar mass and molar volume. (9.02x10-5g/mL) 17. Calculate the mass of 8. ...
SAT Practice Test 3
... The element with an electron configuration of [He]2s 1 has a greater nuclear charge than fluorine 1 mole of NaCl yields 3 moles of ions in solution Neutrons and protons are both located in the principal energy levels of the atom HCl is a proton donor Powdered zinc has a greater surface area NH3 is a ...
... The element with an electron configuration of [He]2s 1 has a greater nuclear charge than fluorine 1 mole of NaCl yields 3 moles of ions in solution Neutrons and protons are both located in the principal energy levels of the atom HCl is a proton donor Powdered zinc has a greater surface area NH3 is a ...
Revision exam - Dynamic Science
... c) Carbon dioxide does not support combustion. d) Carbon dioxide is not used by green plants and therefore accumulates in closed environments. ...
... c) Carbon dioxide does not support combustion. d) Carbon dioxide is not used by green plants and therefore accumulates in closed environments. ...
Chapter 2 - Molecules of Life (Biochemistry) Periodic Table of
... "Deoxyribose in DNA backbone! e.g. "Monosaccharides (e.g. glucose, fructose)! " "Disaccharides (e.g. sucrose, maltose)! " "Polysaccharides (e.g. glycogen)! ...
... "Deoxyribose in DNA backbone! e.g. "Monosaccharides (e.g. glucose, fructose)! " "Disaccharides (e.g. sucrose, maltose)! " "Polysaccharides (e.g. glycogen)! ...
Solution
... O2 is paramagnetic meaning it has unpaired electrons, while N2 is diamagnetic with no unpaired electrons. c) N2 and O2 can react to form NO, which is then oxidized to NO2. Draw the Lewis dot structure for NO2, including resonance if necessary. Identify the molecular and electronic geometry of NO2. ...
... O2 is paramagnetic meaning it has unpaired electrons, while N2 is diamagnetic with no unpaired electrons. c) N2 and O2 can react to form NO, which is then oxidized to NO2. Draw the Lewis dot structure for NO2, including resonance if necessary. Identify the molecular and electronic geometry of NO2. ...
Dr. Ali Ebneshahidi © 2016 Ebneshahidi
... Organic chemistry – chemistry that deals with organic substances (those that contain carbon and hydrogen). Biochemistry - chemistry of living organisms; essential for understanding physiology because body functions involve chemical changes that occur within cells. Matter – anything that has we ...
... Organic chemistry – chemistry that deals with organic substances (those that contain carbon and hydrogen). Biochemistry - chemistry of living organisms; essential for understanding physiology because body functions involve chemical changes that occur within cells. Matter – anything that has we ...
Chapter 1 Matter on the Atomic Scale
... • Slightly larger, fixed volume than a solid. • More randomly arranged than a solid. • Constant collisions with neighbors. • Less confined, can move past each other. ...
... • Slightly larger, fixed volume than a solid. • More randomly arranged than a solid. • Constant collisions with neighbors. • Less confined, can move past each other. ...
Unit 13 Worksheet Answers
... They each cause more collisions so the reaction can happen faster. The temperature also causes the collisions to happen with more energy so there are more effective collisions. 4) It has been found that rates are more rapid at the beginning of a reaction than toward the end, assuming the temperature ...
... They each cause more collisions so the reaction can happen faster. The temperature also causes the collisions to happen with more energy so there are more effective collisions. 4) It has been found that rates are more rapid at the beginning of a reaction than toward the end, assuming the temperature ...
Book chapter review solutions
... 63. Appearance will change during a change of state, which is a physical change. 64. melting point, boiling point, density, chemical reactivity 65. a. oxygen and calcium b. silicon, aluminum, and iron c. Oxygen makes up approximately 89% of the mass of water. The human body is approximately 80 ...
... 63. Appearance will change during a change of state, which is a physical change. 64. melting point, boiling point, density, chemical reactivity 65. a. oxygen and calcium b. silicon, aluminum, and iron c. Oxygen makes up approximately 89% of the mass of water. The human body is approximately 80 ...
Food and Fuels
... the increase. It is a modern problem, with a complex mixture of causes, including increasing car use, less active lifestyles and the popularity of convenience foods. Obesity is linked to increased rates of ...
... the increase. It is a modern problem, with a complex mixture of causes, including increasing car use, less active lifestyles and the popularity of convenience foods. Obesity is linked to increased rates of ...
Class Activity
... Substance made up Two or more elements of only one type of that are chemically atom combined Examples - gold, silver, carbon, oxygen and hydrogen ...
... Substance made up Two or more elements of only one type of that are chemically atom combined Examples - gold, silver, carbon, oxygen and hydrogen ...
So where did all the matter on Earth come from - Bennatti
... For example, hydrogen has the atomic number 1 meaning each hydrogen atom has one proton in its nucleus. No other atom has one proton in its nucleus. Hydrogen is the simplest element. The atomic number of helium is two. Each helium atom has two protons. No other element is made of atoms with two prot ...
... For example, hydrogen has the atomic number 1 meaning each hydrogen atom has one proton in its nucleus. No other atom has one proton in its nucleus. Hydrogen is the simplest element. The atomic number of helium is two. Each helium atom has two protons. No other element is made of atoms with two prot ...
Chapter 2 – Chemical Composition of the Body
... • Bonds formed between the hydrogen end (+ charged) of a polar molecule and the – end of any other polar molecule or highly electronegative atom (e.g. P, N, O) are called hydrogen bonds. • These hydrogen bonds are very important because they alter the physical and chemical properties of many molec ...
... • Bonds formed between the hydrogen end (+ charged) of a polar molecule and the – end of any other polar molecule or highly electronegative atom (e.g. P, N, O) are called hydrogen bonds. • These hydrogen bonds are very important because they alter the physical and chemical properties of many molec ...
Review Sheet for Chemistry* First Semester Final
... Ionic bonds are formed when a ____________ and a _________________ combine. Metals lose electrons and form _____________ while nonmetals gain and electrons form __________. Molecular compounds form when a ______________ and a _______________ combine as they share ...
... Ionic bonds are formed when a ____________ and a _________________ combine. Metals lose electrons and form _____________ while nonmetals gain and electrons form __________. Molecular compounds form when a ______________ and a _______________ combine as they share ...
Unit 1: Building Blocks Homework
... Chlorine has a greater attraction than hydrogen for the bonded electrons in a hydrogen chloride molecule. What term is used to describe this type of covalent bond? ...
... Chlorine has a greater attraction than hydrogen for the bonded electrons in a hydrogen chloride molecule. What term is used to describe this type of covalent bond? ...
Study Island Copyright © 2012 Study Island
... 5. How many elements are found in the following chemical equation? K + Cl → KCl A. 1 B. 2 C. 0 D. 3 ...
... 5. How many elements are found in the following chemical equation? K + Cl → KCl A. 1 B. 2 C. 0 D. 3 ...
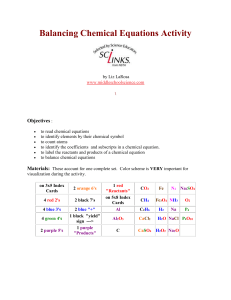
Balancing Chemical Equations Activity by Liz LaRosa www
... The index cards are a bit time consuming to create. I had some students help at lunch time for a few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making in ...
... The index cards are a bit time consuming to create. I had some students help at lunch time for a few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making in ...
Basic Chemistry notes
... ______________________—two or more like atoms combined chemically ______________________—two or more different atoms combined chemically ...
... ______________________—two or more like atoms combined chemically ______________________—two or more different atoms combined chemically ...
Activity Booklet – Task 1
... times. The use of disposable gloves is also recommended. The lamp provided on your workstation is connected to a 240 V mains supply and can become very hot to touch. Proper caution is advised. You are asked to attempt all questions and write your answers clearly in the spaces provided. Any work scri ...
... times. The use of disposable gloves is also recommended. The lamp provided on your workstation is connected to a 240 V mains supply and can become very hot to touch. Proper caution is advised. You are asked to attempt all questions and write your answers clearly in the spaces provided. Any work scri ...
Exam Review - hrsbstaff.ednet.ns.ca
... a) all atoms are electrically neutral. b) the nucleus of the atom contains the positive charge. c) an electron has a very small mass. d) electrons are a part of all matter. 26. The nucleus of an atom usually consists of a) electrons and protons. b) protons and neutrons. c) neutrons and electrons. d) ...
... a) all atoms are electrically neutral. b) the nucleus of the atom contains the positive charge. c) an electron has a very small mass. d) electrons are a part of all matter. 26. The nucleus of an atom usually consists of a) electrons and protons. b) protons and neutrons. c) neutrons and electrons. d) ...
Writing Formulas
... Remember the algebraic sum of the ions' oxidation numbers must equal zero. (Balance) Learn the polyatomic ions. Learn those ions with multiple oxidation numbers and use Roman numerals to indicate the charge. ...
... Remember the algebraic sum of the ions' oxidation numbers must equal zero. (Balance) Learn the polyatomic ions. Learn those ions with multiple oxidation numbers and use Roman numerals to indicate the charge. ...
Artificial photosynthesis

Artificial photosynthesis is a chemical process that replicates the natural process of photosynthesis, a process that converts sunlight, water, and carbon dioxide into carbohydrates and oxygen. The term is commonly used to refer to any scheme for capturing and storing the energy from sunlight in the chemical bonds of a fuel (a solar fuel). Photocatalytic water splitting converts water into Hydrogen Ions and oxygen, and is a main research area in artificial photosynthesis. Light-driven carbon dioxide reduction is another studied process, replicating natural carbon fixation.Research developed in this field encompasses design and assembly of devices (and their components) for the direct production of solar fuels, photoelectrochemistry and its application in fuel cells, and engineering of enzymes and photoautotrophic microorganisms for microbial biofuel and biohydrogen production from sunlight. Many, if not most, of the artificial approaches are bio-inspired, i.e., they rely on biomimetics.