Chemical bonding
... • Any substance that undergoes a reaction is called a REACTANT • When reactants undergo a ...
... • Any substance that undergoes a reaction is called a REACTANT • When reactants undergo a ...
Section 11.1 Assessment How many mole ratios can be written for
... the start of a chemical reaction determines how much product can form. ...
... the start of a chemical reaction determines how much product can form. ...
Chemistry Notes
... Example of Bonds Let's look at Sodium Hydroxide (Na-OH)... You can see that on the left is the Sodium part and the right has the Oxygen/Hydrogen part. The bond which binds the Hydrogen to the Oxygen is covalent. The Sodium is bonded to the HYDROXIDE part of the compound with an ionic bond. This is a ...
... Example of Bonds Let's look at Sodium Hydroxide (Na-OH)... You can see that on the left is the Sodium part and the right has the Oxygen/Hydrogen part. The bond which binds the Hydrogen to the Oxygen is covalent. The Sodium is bonded to the HYDROXIDE part of the compound with an ionic bond. This is a ...
Types of Chemical Reactions
... in the fume hoods. These chemicals are toxic and will harm the environment if not disposed of properly. • Do not eat, drink, or apply the chemicals to skin. Many of these chemicals are highly corrosive and in addition to being toxic, they will burn your skin and muscle tissue. Ouch! • If any of the ...
... in the fume hoods. These chemicals are toxic and will harm the environment if not disposed of properly. • Do not eat, drink, or apply the chemicals to skin. Many of these chemicals are highly corrosive and in addition to being toxic, they will burn your skin and muscle tissue. Ouch! • If any of the ...
Chemical Equations
... The reactants that enter into a reaction The products that are formed by the reaction The relative amounts of each substance used and ...
... The reactants that enter into a reaction The products that are formed by the reaction The relative amounts of each substance used and ...
Role of mathematics in chemistry
... The emerging frontier of theoretical chemistry already encompasses the use of algebraic topology to discern patterns in structure–activity correlation. The various notions of theoretical linguistics are also finding their place in the axiomatic formulation of chemistry, although they have not succee ...
... The emerging frontier of theoretical chemistry already encompasses the use of algebraic topology to discern patterns in structure–activity correlation. The various notions of theoretical linguistics are also finding their place in the axiomatic formulation of chemistry, although they have not succee ...
Unit 2 matter - Kowenscience.com
... C. Atomic Theory of Matter • In the beginning of the 1800s, chemistry was a very different science than it is today. Little was known about the nature and structure of matter. • Thanks to two brilliant early experimentalists, two important chemical laws had been proposed and tested at that time. • ...
... C. Atomic Theory of Matter • In the beginning of the 1800s, chemistry was a very different science than it is today. Little was known about the nature and structure of matter. • Thanks to two brilliant early experimentalists, two important chemical laws had been proposed and tested at that time. • ...
material safety data sheet
... RESPIRATORY PROTECTION: If this product is used as directed, respiratory protection is not required. Seek professional advice prior to respirator selection and use. Follow OSHA respirator regulations (29 CFR 1910.134) and, if necessary, wear a MSHA/ NIOSH approved respirator. If respirators are used ...
... RESPIRATORY PROTECTION: If this product is used as directed, respiratory protection is not required. Seek professional advice prior to respirator selection and use. Follow OSHA respirator regulations (29 CFR 1910.134) and, if necessary, wear a MSHA/ NIOSH approved respirator. If respirators are used ...
matter
... • Chemical reaction (also known as a chemical change) is a change in a substance or substances that results in a totally new substance – Ex: 2H2(g) + O2(g) 2H2O(g) Notice that the reactants (the substances you start with) combine to form a new substance (the product) ...
... • Chemical reaction (also known as a chemical change) is a change in a substance or substances that results in a totally new substance – Ex: 2H2(g) + O2(g) 2H2O(g) Notice that the reactants (the substances you start with) combine to form a new substance (the product) ...
Chapter 7
... • Increasing the temperature of a substance causes its particles to move faster, on average. • Particles that move faster are both more likely to collide and more likely to react. • If the number of collisions that produce reactions increases, then the reaction rate increases ...
... • Increasing the temperature of a substance causes its particles to move faster, on average. • Particles that move faster are both more likely to collide and more likely to react. • If the number of collisions that produce reactions increases, then the reaction rate increases ...
Chemistry 20H
... water (H2O) freezes, it becomes ice, but its chemical formula (H2O) remains the same. When water boils it becomes steam, but the chemical formula (H2O) is still the same. Physical reactions also include subdivision, when a substance is broken into pieces. If a rock is ground into powder, the powder ...
... water (H2O) freezes, it becomes ice, but its chemical formula (H2O) remains the same. When water boils it becomes steam, but the chemical formula (H2O) is still the same. Physical reactions also include subdivision, when a substance is broken into pieces. If a rock is ground into powder, the powder ...
PART 2 – CHEMISTRY
... where the atom has more than one shell, then the atom is said to be stable. This means that the atom does not react with any other kind of atom and thus remains isolated and inert. For example, this is so in the case of the rare gases, argon, neon, helium etc. meaning that they cannot form compounds ...
... where the atom has more than one shell, then the atom is said to be stable. This means that the atom does not react with any other kind of atom and thus remains isolated and inert. For example, this is so in the case of the rare gases, argon, neon, helium etc. meaning that they cannot form compounds ...
Material Safety Datasheet 17117 (PDF)
... possessing, use, transport, storage, and disposal of the substance of any known risk to health. This product contains a component which does not appear on the TSCA public inventory. It is manufactured or imported only in small quantities and solely for research and development. All usage of this pro ...
... possessing, use, transport, storage, and disposal of the substance of any known risk to health. This product contains a component which does not appear on the TSCA public inventory. It is manufactured or imported only in small quantities and solely for research and development. All usage of this pro ...
Elements Combine to Form Compounds
... Heat or light is given off during the change Bubbles or gas are formed Solid clumps (precipitate) forms in the liquid The change is difficult to reverse ...
... Heat or light is given off during the change Bubbles or gas are formed Solid clumps (precipitate) forms in the liquid The change is difficult to reverse ...
Naming Compounds
... Heat or light is given off during the change Bubbles or gas are formed Solid clumps (precipitate) forms in the liquid The change is difficult to reverse ...
... Heat or light is given off during the change Bubbles or gas are formed Solid clumps (precipitate) forms in the liquid The change is difficult to reverse ...
PPT format - Columbia University
... Element: An element is a substance which cannot be decomposed into simpler substances by chemical processes. Examples: hydrogen, carbon, oxygen. Atomic interpretation: An element is a substance that contains only one kind of atom. Hydrogen (H) atoms, carbon atoms (C), oxygen atoms (O). Compound: A c ...
... Element: An element is a substance which cannot be decomposed into simpler substances by chemical processes. Examples: hydrogen, carbon, oxygen. Atomic interpretation: An element is a substance that contains only one kind of atom. Hydrogen (H) atoms, carbon atoms (C), oxygen atoms (O). Compound: A c ...
Chapter 2 Matter
... Matter can be a gas, a liquid, or a solid. Gases have no fixed shape or volume. Gases can be compressed to form liquids. Liquids have no shape, but they do have a volume. Solids are rigid and have a definite shape and volume. ...
... Matter can be a gas, a liquid, or a solid. Gases have no fixed shape or volume. Gases can be compressed to form liquids. Liquids have no shape, but they do have a volume. Solids are rigid and have a definite shape and volume. ...
the news reports that led to millions of servings of processed
... Anna D. Howell - UCCE Ventura Co. Frank Zalom – UC Davis Mark Bolda – UCCE Santa Cruz Co. ...
... Anna D. Howell - UCCE Ventura Co. Frank Zalom – UC Davis Mark Bolda – UCCE Santa Cruz Co. ...
Chapter 11 Chemical Reactions
... (aq) after the formula = dissolved in water, an aqueous solution: NaCl(aq) is a salt water solution used after a product indicates a gas has been produced: H2↑ used after a product indicates a solid has been produced: PbI2↓ ...
... (aq) after the formula = dissolved in water, an aqueous solution: NaCl(aq) is a salt water solution used after a product indicates a gas has been produced: H2↑ used after a product indicates a solid has been produced: PbI2↓ ...
Matter - Clayton State University
... - change of state (from liquid to gas, from liquid to solid, etc.) - when liquid water evaporates to steam or freezes to ice ...
... - change of state (from liquid to gas, from liquid to solid, etc.) - when liquid water evaporates to steam or freezes to ice ...
Chemical Reactions
... Never change a subscript to balance an equation (You can only change coefficients) – If you change the subscript (formula) you are describing a different chemical. – H2O is a different compound than H2O2 Never put a coefficient in the middle of a formula; they must go only in the front ...
... Never change a subscript to balance an equation (You can only change coefficients) – If you change the subscript (formula) you are describing a different chemical. – H2O is a different compound than H2O2 Never put a coefficient in the middle of a formula; they must go only in the front ...
Block 1 - cloudfront.net
... Air bags inflate almost instantaneously upon a car's impact. The effectiveness of air bags is based on the rapid conversion of a small mass of NaN3 into a large volume of gas. The gas fills an air bag, preventing the driver from hitting the steering wheel or dashboard. The entire reaction occurs in ...
... Air bags inflate almost instantaneously upon a car's impact. The effectiveness of air bags is based on the rapid conversion of a small mass of NaN3 into a large volume of gas. The gas fills an air bag, preventing the driver from hitting the steering wheel or dashboard. The entire reaction occurs in ...
Chemical reactions alter arrangements of atoms.
... reaction. In the burning of natural gas, for example, methane (CH4) and oxygen (O2) are the reactants in the chemical reaction. Products are the substances formed by a chemical reaction. In the burning of natural gas, carbon dioxide (CO2) and water (H2O) are the products formed by the reaction. Reac ...
... reaction. In the burning of natural gas, for example, methane (CH4) and oxygen (O2) are the reactants in the chemical reaction. Products are the substances formed by a chemical reaction. In the burning of natural gas, carbon dioxide (CO2) and water (H2O) are the products formed by the reaction. Reac ...
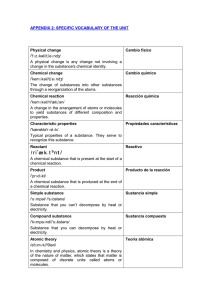
specific vocabulary of the unit
... non-metals on the Periodic Table of Elements. They are sometimes called semi-metals and have characteristics that resemble both metals and nonmetals. ...
... non-metals on the Periodic Table of Elements. They are sometimes called semi-metals and have characteristics that resemble both metals and nonmetals. ...
Chapter 4 - WordPress.com
... Types of Reactions • 4 different types of Reactions: – Combination: A + B AB – Decomposition: AB A + B – Single Replacement: A + BC AC + B ...
... Types of Reactions • 4 different types of Reactions: – Combination: A + B AB – Decomposition: AB A + B – Single Replacement: A + BC AC + B ...
Chemical industry

The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products.