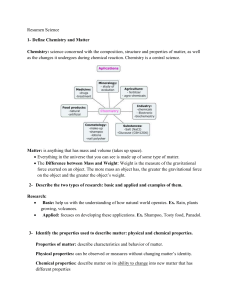
I Examen I Trim Science
... 13. Boiling point: temperature needed in which a liquid changes into a gas. Ex. Water 100˚C 14. Texture: way into which particles are placed in space in an object. Perceived by touch 15. Color: property perceived by the due to the effect of light reflected by objects. 16. Taste: ability or quality ...
... 13. Boiling point: temperature needed in which a liquid changes into a gas. Ex. Water 100˚C 14. Texture: way into which particles are placed in space in an object. Perceived by touch 15. Color: property perceived by the due to the effect of light reflected by objects. 16. Taste: ability or quality ...
Document
... rise in atmospheric temperature since 1860. During the same period atmospheric CO2 levels have risen 25%. Are the two trends causal? ...
... rise in atmospheric temperature since 1860. During the same period atmospheric CO2 levels have risen 25%. Are the two trends causal? ...
Utah - Wavefunction, Inc.
... atoms explains the conservation of matter, since the number of atoms stays the same in a chemical reaction no matter how they are rearranged; the total mass stays the same. Although energy can be absorbed or released in a chemical reaction, the total amount of energy and matter in it remains constan ...
... atoms explains the conservation of matter, since the number of atoms stays the same in a chemical reaction no matter how they are rearranged; the total mass stays the same. Although energy can be absorbed or released in a chemical reaction, the total amount of energy and matter in it remains constan ...
Additional Review
... o CFC’s chlorofluorocarbons come from Freon used in __________________________________________________ can stay in the atmosphere for up to 50 years _________________________________________, the ozone helps to protect the Earth from harmful UV, and X-Rays from the sun o Greenhouse gases (____ ...
... o CFC’s chlorofluorocarbons come from Freon used in __________________________________________________ can stay in the atmosphere for up to 50 years _________________________________________, the ozone helps to protect the Earth from harmful UV, and X-Rays from the sun o Greenhouse gases (____ ...
transcript - American Chemical Society
... other threat agents, too. “Yes, this could be extended to include not only the pox viruses but also Bacillus spores and also it's very easily applicable to chemical threat agents such as mustard gas and VX, and one of the big challenges, of big military interest right now, is detection of explosive ...
... other threat agents, too. “Yes, this could be extended to include not only the pox viruses but also Bacillus spores and also it's very easily applicable to chemical threat agents such as mustard gas and VX, and one of the big challenges, of big military interest right now, is detection of explosive ...
Chapter 1
... Observation: is the act of gathering information by using your senses on a macroscopic level. Hypothesis: is a testable prediction used to explain an observation (if, then). Experiment: is a set of observations used to test a ...
... Observation: is the act of gathering information by using your senses on a macroscopic level. Hypothesis: is a testable prediction used to explain an observation (if, then). Experiment: is a set of observations used to test a ...
Chapter 3
... – how much reactant is consumed and how much product is formed – coefficients must be consistent with the Law of Conservation of Mass; atoms are neither created nor destroyed in a chemical reaction. – i.e. chemical equation must be balanced ...
... – how much reactant is consumed and how much product is formed – coefficients must be consistent with the Law of Conservation of Mass; atoms are neither created nor destroyed in a chemical reaction. – i.e. chemical equation must be balanced ...
the powerpoint
... substance. Chemical properties can be recognized only when substances react or do not react chemically with one another, that is, when they undergo a change in composition. A chemical property of one substance usually involves its ability to react (combine) or not react with another specific substan ...
... substance. Chemical properties can be recognized only when substances react or do not react chemically with one another, that is, when they undergo a change in composition. A chemical property of one substance usually involves its ability to react (combine) or not react with another specific substan ...
Chemical Reactions and Stoichiometry
... Indicators of a Chemical Reaction – evidence of a chemical reaction a. Evolution of heat and light (simultaneously) b. Production of a gas (bubbles, odor change) c. Formation of a precipitate (solid, cloudy) d. Color change (not introduced by an outside source such as dye or ink) Characteristics of ...
... Indicators of a Chemical Reaction – evidence of a chemical reaction a. Evolution of heat and light (simultaneously) b. Production of a gas (bubbles, odor change) c. Formation of a precipitate (solid, cloudy) d. Color change (not introduced by an outside source such as dye or ink) Characteristics of ...
Safety in the classroom
... The balance is a precision instrument that must be handled with care. pan If you spill a chemical on or near the balance, clean it up immediately. Never attempt to weigh an object with a mass greater than the maximum capacity of the balance. When you finish weighing, return all the masses to z ...
... The balance is a precision instrument that must be handled with care. pan If you spill a chemical on or near the balance, clean it up immediately. Never attempt to weigh an object with a mass greater than the maximum capacity of the balance. When you finish weighing, return all the masses to z ...
Chemical reactions cause chemical changes. They involve the
... a change in substances and a change in energy. However, neither matter nor energy is created or destroyed in a chemical reaction. The fact that matter is not created or destroyed in a chemical reaction is called the law of conservation of mass. In order for chemical reaction equations to show that n ...
... a change in substances and a change in energy. However, neither matter nor energy is created or destroyed in a chemical reaction. The fact that matter is not created or destroyed in a chemical reaction is called the law of conservation of mass. In order for chemical reaction equations to show that n ...
Chapter 7 Chemical Reactions
... 4. Check to make sure all atoms and polyatomics are balanced 5. Make sure all the coefficients are in the lowest possible ratio 6. Write the ratio Write balanced equations for the reactions below: 1. Water is formed when hydrogen react with oxygen 2. Pure silver can be removed from silver nitrate ...
... 4. Check to make sure all atoms and polyatomics are balanced 5. Make sure all the coefficients are in the lowest possible ratio 6. Write the ratio Write balanced equations for the reactions below: 1. Water is formed when hydrogen react with oxygen 2. Pure silver can be removed from silver nitrate ...
SECTION 1 PRODUCT AND COMPANY INFORMATION Terbium (III
... CONDITIONS FOR SAFE STORAGE: Store material in a tightly sealed container in a dry and well-ventilated area. Hygroscopic. ...
... CONDITIONS FOR SAFE STORAGE: Store material in a tightly sealed container in a dry and well-ventilated area. Hygroscopic. ...
chemical reaction
... • Before a reaction can start, molecules of the reactants have to bump into each other, or collide. • The collision must be strong enough. • This means the reactants must smash into each other with a certain amount of energy. • To start any chemical reaction, a minimum amount of energy is needed. • ...
... • Before a reaction can start, molecules of the reactants have to bump into each other, or collide. • The collision must be strong enough. • This means the reactants must smash into each other with a certain amount of energy. • To start any chemical reaction, a minimum amount of energy is needed. • ...
Physical and Chemical Changes Worksheet
... Can you recognize the chemical and physical changes that happen all around us? If you change the way something looks, but haven’t made a new substance, a physical change (P) has occurred. If the substance has been changes into another substance, a chemical change (C) has occurred. ...
... Can you recognize the chemical and physical changes that happen all around us? If you change the way something looks, but haven’t made a new substance, a physical change (P) has occurred. If the substance has been changes into another substance, a chemical change (C) has occurred. ...
Chapter 8
... • List three observations that suggest that a chemical reaction has taken place. • List three requirements for a correctly written chemical equation. • Write a word equation and a formula equation for a given chemical reaction. • Balance a formula equation by inspection. ...
... • List three observations that suggest that a chemical reaction has taken place. • List three requirements for a correctly written chemical equation. • Write a word equation and a formula equation for a given chemical reaction. • Balance a formula equation by inspection. ...
Diapositive 1 - Aptar
... • Future guidance may be based on “PQRI Safety Thresholds and Best Practices for Extractables and Leachables in Orally Inhaled and Nasal Drug Products” (2007?) ...
... • Future guidance may be based on “PQRI Safety Thresholds and Best Practices for Extractables and Leachables in Orally Inhaled and Nasal Drug Products” (2007?) ...
Chapter 15- Classification of Matter
... ii. __________________ is a process for separating a mixture by evaporating a liquid and condensing its vapor. c. _________________________- characteristics of a substance indicating that it can change chemically; for example __________________________________________ d. When one substance changes t ...
... ii. __________________ is a process for separating a mixture by evaporating a liquid and condensing its vapor. c. _________________________- characteristics of a substance indicating that it can change chemically; for example __________________________________________ d. When one substance changes t ...
Balancing a Chemical Equation
... Separate the reactants from the products with an arrow. Use plus signs to separate the reactants and the products. NaHCO3(s) + HCl(aq) → NaCl(aq) + H2O(l) + CO2(g) ...
... Separate the reactants from the products with an arrow. Use plus signs to separate the reactants and the products. NaHCO3(s) + HCl(aq) → NaCl(aq) + H2O(l) + CO2(g) ...
Chemical suicide by inhalation of hydrogen sulfide
... [1]. Bath additive contains lime sulfur. Toilet detergent contains acid and plays the role of an oxidant to produce H2S gas [1]. The phenomenon was dubbed “Chemical suicide” or “Domestic suicide” by the media, because it involves common household cleaners, largely available from hardwares, groceries ...
... [1]. Bath additive contains lime sulfur. Toilet detergent contains acid and plays the role of an oxidant to produce H2S gas [1]. The phenomenon was dubbed “Chemical suicide” or “Domestic suicide” by the media, because it involves common household cleaners, largely available from hardwares, groceries ...
Chemical Equations and Reactions
... How to Balance an Equation 1. Balance the different types of atoms one at a time 2. First balance the atoms of elements that are combined and that appear only once on each side of the equation. (start with the largest compound first) 3. Balance polyatomic ions that appear on both sides of the equat ...
... How to Balance an Equation 1. Balance the different types of atoms one at a time 2. First balance the atoms of elements that are combined and that appear only once on each side of the equation. (start with the largest compound first) 3. Balance polyatomic ions that appear on both sides of the equat ...
Lecture on Air Pollution Prevention and Control
... established by the EPA in quantities greater than the threshold level, will be required to comply with rules for the "use, operation, repair, replacement, and maintenance of equipment to monitor, detect, inspect, and control releases." Such regulations must also require owners and operators to "prep ...
... established by the EPA in quantities greater than the threshold level, will be required to comply with rules for the "use, operation, repair, replacement, and maintenance of equipment to monitor, detect, inspect, and control releases." Such regulations must also require owners and operators to "prep ...
Household Acids and Bases Lab
... various species of lichens. The chief component of litmus is azolitmin. Anthocyanins are also acidbase indicators and can be found among the higher plants. These compounds constitute most of the yellow, red, and blue colors in flowers and fruits. Anthocyanins are excellent acid-base indicators becau ...
... various species of lichens. The chief component of litmus is azolitmin. Anthocyanins are also acidbase indicators and can be found among the higher plants. These compounds constitute most of the yellow, red, and blue colors in flowers and fruits. Anthocyanins are excellent acid-base indicators becau ...
Chemical industry

The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products.























