Section 1 The Nature of Chemical Reactions
... • The law of definite proportions states that a compound always contains the same elements in the same proportions, regardless of how the compound is made or how much of the compound is formed. • Because the law of definite proportions holds true for all chemical substances in all reactions, mole ra ...
... • The law of definite proportions states that a compound always contains the same elements in the same proportions, regardless of how the compound is made or how much of the compound is formed. • Because the law of definite proportions holds true for all chemical substances in all reactions, mole ra ...
Unit 3
... building) • Chemicals (eg Cl2 for water purification) • Dyes (for clothing etc) • Cosmetics and medicines • Soaps and detergents • Etc!!!! ...
... building) • Chemicals (eg Cl2 for water purification) • Dyes (for clothing etc) • Cosmetics and medicines • Soaps and detergents • Etc!!!! ...
How do we predict chemical change?
... Not every combination of substances will lead to the formation of new compounds via a chemical reaction. How can we predict when a chemical process takes place? One approach could be to compare the relative stability of reactants and products. We might expect that chemical reactions will proceed in ...
... Not every combination of substances will lead to the formation of new compounds via a chemical reaction. How can we predict when a chemical process takes place? One approach could be to compare the relative stability of reactants and products. We might expect that chemical reactions will proceed in ...
Chapter 1 Matter and Change
... Mixtures are a physical blend of at least two substances; have variable composition. They can be either: 1) Heterogeneous – the mixture is not uniform in composition • Chocolate chip cookie, gravel, soil. 2) Homogeneous - same composition throughout; called “solutions” • Kool-aid, air, salt water ...
... Mixtures are a physical blend of at least two substances; have variable composition. They can be either: 1) Heterogeneous – the mixture is not uniform in composition • Chocolate chip cookie, gravel, soil. 2) Homogeneous - same composition throughout; called “solutions” • Kool-aid, air, salt water ...
PowerPoint - Science Geek
... the grand thing is to be able to reason backward. This is a very useful accomplishment, and a very easy one, but people do not practice it much.” Sherlock Holmes, in Sir Arthur Conan Doyle’s A Study in Scarlet ...
... the grand thing is to be able to reason backward. This is a very useful accomplishment, and a very easy one, but people do not practice it much.” Sherlock Holmes, in Sir Arthur Conan Doyle’s A Study in Scarlet ...
CHEMICAL REACTIONS Chapter 4
... 4 Al atoms + 3 O2 molecules ------> 2 formula units of Al2O3 4 moles of Al + 3 moles of O2 ----> 2 moles of Al2O3 ...
... 4 Al atoms + 3 O2 molecules ------> 2 formula units of Al2O3 4 moles of Al + 3 moles of O2 ----> 2 moles of Al2O3 ...
Chapter 1: Quiz Review - Wetaskiwin Composite High School
... 1. Which process below has only been used in North America since the development of modern industrial chemistry? A. Dying fabric C. Making leather B. Producing medicines to treat insect bites D. Refining and processing crude oil 2. Which human need below was met by Aboriginal people using chemical p ...
... 1. Which process below has only been used in North America since the development of modern industrial chemistry? A. Dying fabric C. Making leather B. Producing medicines to treat insect bites D. Refining and processing crude oil 2. Which human need below was met by Aboriginal people using chemical p ...
Ductility-the ability to be stretched into wires
... identity) and form a new substance with different properties? Is the ability to be torn a physical or chemical property? Physical Property: Property that can be tested/observed without changing chemical identity of the substance; can be undone ...
... identity) and form a new substance with different properties? Is the ability to be torn a physical or chemical property? Physical Property: Property that can be tested/observed without changing chemical identity of the substance; can be undone ...
The masses of reactants and products are equal.
... That is, the mass of the products appears to be less than the mass of the reactants. In other reactions, the products appear to gain mass. For example, plants grow through a complex series of reactions, but where does their extra mass come from? At one time, scientists thought that chemical reaction ...
... That is, the mass of the products appears to be less than the mass of the reactants. In other reactions, the products appear to gain mass. For example, plants grow through a complex series of reactions, but where does their extra mass come from? At one time, scientists thought that chemical reaction ...
Chemical Reactions
... A decomposition reaction is a reaction in which a compound breaks down into two or more simpler substances – The reactant in a decomposition reaction must be a compound – the products may be elements or compounds AB → A + B ...
... A decomposition reaction is a reaction in which a compound breaks down into two or more simpler substances – The reactant in a decomposition reaction must be a compound – the products may be elements or compounds AB → A + B ...
Ductility-the ability to be stretched into wires
... • Does the paper change its chemistry (chemical identity) and form a new substance with different properties? • Is the ability to be torn a physical or chemical property? – Physical Property: Property that can be tested/observed without changing chemical identity of the substance; can be undone ...
... • Does the paper change its chemistry (chemical identity) and form a new substance with different properties? • Is the ability to be torn a physical or chemical property? – Physical Property: Property that can be tested/observed without changing chemical identity of the substance; can be undone ...
120CH05 - Louisiana Tech University
... Balancing chemical eqns • Use correct formulas for the reactants and products (if word eqn at start) • Balance by putting coefficients (nos) in front of the formulas. You may not change the formulas! These coefficients are called the stoichiometric (measure of mass) coefficients. • By convention us ...
... Balancing chemical eqns • Use correct formulas for the reactants and products (if word eqn at start) • Balance by putting coefficients (nos) in front of the formulas. You may not change the formulas! These coefficients are called the stoichiometric (measure of mass) coefficients. • By convention us ...
practice final examination
... 10. Answer true or false for each of the following questions below (circle your choice): a) ...
... 10. Answer true or false for each of the following questions below (circle your choice): a) ...
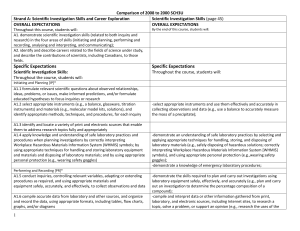
Comparison of 2008 to 2000 SCH3U_ud
... substance affects the environment, and propose ways to lessen the harmfulness of the substance (e.g., by reducing the amount used, by modifying one of its chemical components) or identify alternative substances that could be used for the same purpose [IP, PR, AI, C] Sample issue: Many commercial hou ...
... substance affects the environment, and propose ways to lessen the harmfulness of the substance (e.g., by reducing the amount used, by modifying one of its chemical components) or identify alternative substances that could be used for the same purpose [IP, PR, AI, C] Sample issue: Many commercial hou ...
Introduction_to_Chemical_Reactions_2011
... compound from its elements in their stable forms at conditions of 298K and 1 atm of pressure inside of a calorimeter. • For most compounds, H0f is negative because bond formation is exothermic! • H0f of an element is always 0 kJ/mol by def. ...
... compound from its elements in their stable forms at conditions of 298K and 1 atm of pressure inside of a calorimeter. • For most compounds, H0f is negative because bond formation is exothermic! • H0f of an element is always 0 kJ/mol by def. ...
Chemical changes
... are: melting, freezing, condensing, breaking, crushing, cutting, and bending. ...
... are: melting, freezing, condensing, breaking, crushing, cutting, and bending. ...
Chemical Reactions PPT
... Subscript – How many with # of atoms “Yields” or of that present produces molecule ...
... Subscript – How many with # of atoms “Yields” or of that present produces molecule ...
balancing chemical equations worksheet
... The following questions relate to these four steps. a. What symbols should we use to describe the physical states? b. Chemists and other scientists always balance chemical equations. Please explain why this is so important. (Hint, refer to the law of conservation of mass) PART B, read the following ...
... The following questions relate to these four steps. a. What symbols should we use to describe the physical states? b. Chemists and other scientists always balance chemical equations. Please explain why this is so important. (Hint, refer to the law of conservation of mass) PART B, read the following ...
Polychlorinated biphenyls (PCB`s)
... Can cause dermatological symptoms; however, these are reversible upon removal of exposure source. FIRST AID MEASURES ...
... Can cause dermatological symptoms; however, these are reversible upon removal of exposure source. FIRST AID MEASURES ...
Formation of amorphous silica surface layers by
... Chemical weathering experiments in the laboratory are generally carried out at conditions that promote the chemical breakdown of minerals, namely in chemically dilute solutions at far-from-equilibrium conditions. Moreover, in order to reproduce natural chemical weathering conditions, experiments are ...
... Chemical weathering experiments in the laboratory are generally carried out at conditions that promote the chemical breakdown of minerals, namely in chemically dilute solutions at far-from-equilibrium conditions. Moreover, in order to reproduce natural chemical weathering conditions, experiments are ...
introduction to matter
... in temperature or physical state brought about by heat such as when cold water turns into hot water and hot water turning into steam and (3) a change in volume when two solutions are added together which results in a simple mixing without reaction. Chemical changes are changes that produce new subst ...
... in temperature or physical state brought about by heat such as when cold water turns into hot water and hot water turning into steam and (3) a change in volume when two solutions are added together which results in a simple mixing without reaction. Chemical changes are changes that produce new subst ...
Example - Request a Spot account
... a. Allow reactions to occur that might not otherwise take place (due to low temperature for example) b. Lower activation energy for a chemical reaction 2. Participation of catalysts in a chemical reaction a. They may undergo a chemical change as a reactant but they are always recycled as a product ( ...
... a. Allow reactions to occur that might not otherwise take place (due to low temperature for example) b. Lower activation energy for a chemical reaction 2. Participation of catalysts in a chemical reaction a. They may undergo a chemical change as a reactant but they are always recycled as a product ( ...
Surveillance of Severe Chemical Corneal Injuries in the UK
... in the UK is low The cases that occur can require extended hospital treatment, with substantial ocular morbidity and visual sequelae Current enforcement of eye protection in the workplace in the UK has probably contributed to a reduced incidence of severe ocular burns ...
... in the UK is low The cases that occur can require extended hospital treatment, with substantial ocular morbidity and visual sequelae Current enforcement of eye protection in the workplace in the UK has probably contributed to a reduced incidence of severe ocular burns ...
Basic Chemistry – Terminology and Reactions
... Products side 1C 1C 4H 2H 2O 3O In order to balance an equation we have to follow these steps. Step 1: Start by finding out how many atoms of each type are on each side of the equation. Step 2: Next, look for an element which is in only one chemical on the left and in only one on the right of the eq ...
... Products side 1C 1C 4H 2H 2O 3O In order to balance an equation we have to follow these steps. Step 1: Start by finding out how many atoms of each type are on each side of the equation. Step 2: Next, look for an element which is in only one chemical on the left and in only one on the right of the eq ...
Chemical industry

The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products.