Nuclear Chemistry
... Nuclear Energy • All nuclear energy is derived from the conversion of small amounts of mass to energy. • Einstein’s relativity equation E = mc2 explains the relationship between matter and energy (c = 3.00 x 108 m/s …it is the speed of light) ...
... Nuclear Energy • All nuclear energy is derived from the conversion of small amounts of mass to energy. • Einstein’s relativity equation E = mc2 explains the relationship between matter and energy (c = 3.00 x 108 m/s …it is the speed of light) ...
Nuclear Reactions Review
... • 13. The use of nuclear reactors to generate electricity is • A.decreasing rapidly. • C.found in dozens of countries. • B.found only in the United States. • D.totally safe. ...
... • 13. The use of nuclear reactors to generate electricity is • A.decreasing rapidly. • C.found in dozens of countries. • B.found only in the United States. • D.totally safe. ...
Nuclear Reactions Review powerpt
... • 13. The use of nuclear reactors to generate electricity is • A.decreasing rapidly. • C.found in dozens of countries. • B.found only in the United States. • D.totally safe. ...
... • 13. The use of nuclear reactors to generate electricity is • A.decreasing rapidly. • C.found in dozens of countries. • B.found only in the United States. • D.totally safe. ...
Lecture14
... Mass of one proton: 1.00728 amu 2 x neutron + 2 x proton = 4.0319 amu SO….Nuclear Binding Energy = 4.0319 – 4.0028 = 0.029 amu ...
... Mass of one proton: 1.00728 amu 2 x neutron + 2 x proton = 4.0319 amu SO….Nuclear Binding Energy = 4.0319 – 4.0028 = 0.029 amu ...
Outline Chapter 8 The Nucleus 8-1. J.J. Thompson`s Plum Pudding
... Here an experimental fusion reactor at Princeton University. This uses powerful magnetic fields to confine the fusion material. This is called a tokamak reactor based on a Soviet reactor. ...
... Here an experimental fusion reactor at Princeton University. This uses powerful magnetic fields to confine the fusion material. This is called a tokamak reactor based on a Soviet reactor. ...
NUCLEAR CHEMISTRY
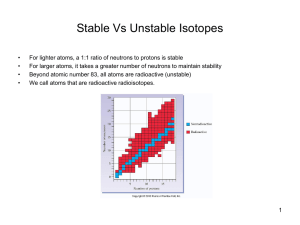
... C. Radioactive Elements 1. All isotopes of all man-made elements are radioactive 2. Some naturally isotopes are radioactive a. All isotopes of all elements beyond bismuth (atomic #83) are radioactive ...
... C. Radioactive Elements 1. All isotopes of all man-made elements are radioactive 2. Some naturally isotopes are radioactive a. All isotopes of all elements beyond bismuth (atomic #83) are radioactive ...
Nuclear Chemistry
... • N/P ratio of stable nuclei • Stable small atoms (atomic # less than 20) are near 1/1 ratio • Stable large atoms are near 1.5/1 ratio. • Predict the stability of the following: carbon-12 mercury-200 hydrogen-3 uranium-238 ...
... • N/P ratio of stable nuclei • Stable small atoms (atomic # less than 20) are near 1/1 ratio • Stable large atoms are near 1.5/1 ratio. • Predict the stability of the following: carbon-12 mercury-200 hydrogen-3 uranium-238 ...
Document
... • Starting nuclear chain reactions = releases a huge amount of energy (E) • Per unit volume, an atom bomb may be millions or billions of times more ...
... • Starting nuclear chain reactions = releases a huge amount of energy (E) • Per unit volume, an atom bomb may be millions or billions of times more ...
Nuclear Chemistry
... days after the accident, but not enough to cause any dose above background levels to local residents. • There were no injuries or adverse health effects from the Three Mile Island accident. ...
... days after the accident, but not enough to cause any dose above background levels to local residents. • There were no injuries or adverse health effects from the Three Mile Island accident. ...
I. Ch. 21.1 Nuclear Radiation
... When the nuclei of certain isotopes are bombarded with neutrons, they undergo _________________, the splitting of a nucleus into smaller fragments. b) In a chain reaction, some of the neutrons produced react with other fissionable atoms, producing more neutrons which react with still more fissionabl ...
... When the nuclei of certain isotopes are bombarded with neutrons, they undergo _________________, the splitting of a nucleus into smaller fragments. b) In a chain reaction, some of the neutrons produced react with other fissionable atoms, producing more neutrons which react with still more fissionabl ...
Stable Vs Unstable Isotopes
... Beta decay: An atom emits a beta particle in the form of an electron when a neutron is changed into a proton. The atomic number increases by 1 while the mass number remains the same. ...
... Beta decay: An atom emits a beta particle in the form of an electron when a neutron is changed into a proton. The atomic number increases by 1 while the mass number remains the same. ...
Ch. 21.1 Nuclear Radiation
... Gamma Radiation – Gamma rays () are high-energy waves emitted from a nucleus as it decays. – Gamma rays have no mass and no electrical charge. Therefore, gamma radiation does not alter the atomic number or mass number of an atom. – Nuclei often emit gamma rays along with alpha or beta particles. ...
... Gamma Radiation – Gamma rays () are high-energy waves emitted from a nucleus as it decays. – Gamma rays have no mass and no electrical charge. Therefore, gamma radiation does not alter the atomic number or mass number of an atom. – Nuclei often emit gamma rays along with alpha or beta particles. ...
Nuclear Fission and Fusion
... Fission uses uranium or plutonium They first absorb a neutron. It is the splitting of a larger nuclei into 2 smaller nuclei, energy and neutrons The neutrons can go on and start a chain reaction ...
... Fission uses uranium or plutonium They first absorb a neutron. It is the splitting of a larger nuclei into 2 smaller nuclei, energy and neutrons The neutrons can go on and start a chain reaction ...
10 facts about NUCLEAR FISSION
... 5. The chain reaction releases huge amount of energy, because mass has been converted into energy! ...
... 5. The chain reaction releases huge amount of energy, because mass has been converted into energy! ...
Foldable - Georgetown ISD
... Example: The skeleton of an ancient fish is found to contain 12.5 % (or 1/8) the amount of carbon-14 that it contained while it was alive. The half-life of carbon-14 is 5730 years. How old was the skeleton? ...
... Example: The skeleton of an ancient fish is found to contain 12.5 % (or 1/8) the amount of carbon-14 that it contained while it was alive. The half-life of carbon-14 is 5730 years. How old was the skeleton? ...
NUCLEAR CHEMISTRY
... Dropped on Nagasaki, Japan, on August 9, 1945, Fat Man devastated more than two square miles of the city and caused approximately 45,000 immediate deaths. While Little Boy was a uranium (U-235) gun-type device, Fat Man was a more complicated and powerful plutonium (Pu-239) implosion weapon that expl ...
... Dropped on Nagasaki, Japan, on August 9, 1945, Fat Man devastated more than two square miles of the city and caused approximately 45,000 immediate deaths. While Little Boy was a uranium (U-235) gun-type device, Fat Man was a more complicated and powerful plutonium (Pu-239) implosion weapon that expl ...
Radioactivity and Nuclear Reactions
... • Henri Becquerel accidentally discovered radioactivity in ...
... • Henri Becquerel accidentally discovered radioactivity in ...
2005 Nuclear FRQs - AP Chemistry Olympics
... (c) Products from a nuclear fission of a uranium atom (d) Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? such as 90Sr and 137Ce are highly radioactive and Answer: decay by emission of beta particles. ...
... (c) Products from a nuclear fission of a uranium atom (d) Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? such as 90Sr and 137Ce are highly radioactive and Answer: decay by emission of beta particles. ...
Terms to Know
... gain and loss of electrons that orbit an atomic nucleus, but do not involve any change in the nucleus itself Positrons : The positron is the antiparticle of the electron. It has the same mass and the same quantity of electric charge as does the electron, but its electric charge is positive rather th ...
... gain and loss of electrons that orbit an atomic nucleus, but do not involve any change in the nucleus itself Positrons : The positron is the antiparticle of the electron. It has the same mass and the same quantity of electric charge as does the electron, but its electric charge is positive rather th ...
Radioactive Decay Series
... Nuclear Fission and Nuclear Fusion Nuclear fission is when a very heavy nuclei splits into more stable nuclei of intermediate mass. This process can happen spontaneously or from nuclear bombardment The process releases enormous amounts of usable energy The energy is converted from the change in mas ...
... Nuclear Fission and Nuclear Fusion Nuclear fission is when a very heavy nuclei splits into more stable nuclei of intermediate mass. This process can happen spontaneously or from nuclear bombardment The process releases enormous amounts of usable energy The energy is converted from the change in mas ...
Nuclear Stability
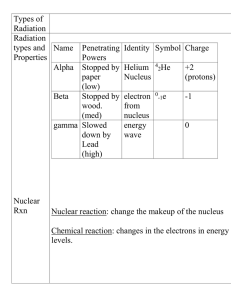
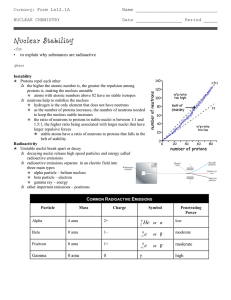
... q radioactive emissions separate in an electric field into three main types p alpha particle – helium nucleus p beta particle – electron p gamma ray – energy q other important emissions – positrons ...
... q radioactive emissions separate in an electric field into three main types p alpha particle – helium nucleus p beta particle – electron p gamma ray – energy q other important emissions – positrons ...
Nuclear Fission & Fusion
... oHave too many or too few neutrons in nucleus oHave more than 83 protons in nucleus oWill decay into a more stable nucleus ...
... oHave too many or too few neutrons in nucleus oHave more than 83 protons in nucleus oWill decay into a more stable nucleus ...
Nuclear and radiation accidents and incidents

A nuclear and radiation accident is defined by the International Atomic Energy Agency (IAEA) as ""an event that has led to significant consequences to people, the environment or the facility."" Examples include lethal effects to individuals, large radioactivity release to the environment, or reactor core melt."" The prime example of a ""major nuclear accident"" is one in which a reactor core is damaged and significant amounts of radioactivity are released, such as in the Chernobyl disaster in 1986.The impact of nuclear accidents has been a topic of debate practically since the first nuclear reactors were constructed in 1954. It has also been a key factor in public concern about nuclear facilities. Some technical measures to reduce the risk of accidents or to minimize the amount of radioactivity released to the environment have been adopted. Despite the use of such measures, human error remains, and ""there have been many accidents with varying impacts as well near misses and incidents"".Worldwide there have been 99 accidents at nuclear power plants. Fifty-seven accidents have occurred since the Chernobyl disaster, and 57% (56 out of 99) of all nuclear-related accidents have occurred in the USA. Serious nuclear power plant accidents include the Fukushima Daiichi nuclear disaster (2011), Chernobyl disaster (1986), Three Mile Island accident (1979), and the SL-1 accident (1961).Nuclear-powered submarine core meltdown and other mishaps include the K-19 (1961), K-11 (1965), K-27 (1968), K-140 (1968), K-429 (1970), K-222 (1980), K-314 (1985), and K-431 (1985). Serious radiation accidents include the Kyshtym disaster, Windscale fire, radiotherapy accident in Costa Rica, radiotherapy accident in Zaragoza, radiation accident in Morocco, Goiania accident, radiation accident in Mexico City, radiotherapy unit accident in Thailand, and the Mayapuri radiological accident in India.The IAEA maintains a website reporting recent accidents.