Nuclear Chemistry powerpoint
... numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
... numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
NUCLEAR CHEMISTRY
... Severe blood changes will be noted and symptoms appear immediately. Approximately two weeks later, some exposed casualties may die. At 300-500 RAD, up to one half of the people exposed will die within 30 days without intensive medical attention. Death is due to the destruction of the blood forming o ...
... Severe blood changes will be noted and symptoms appear immediately. Approximately two weeks later, some exposed casualties may die. At 300-500 RAD, up to one half of the people exposed will die within 30 days without intensive medical attention. Death is due to the destruction of the blood forming o ...
Nuclear Chemistry
... a. Students know protons and neutrons in the nucleus are held together by nuclear forces that overcome the electromagnetic repulsion between the protons. b. Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The cha ...
... a. Students know protons and neutrons in the nucleus are held together by nuclear forces that overcome the electromagnetic repulsion between the protons. b. Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The cha ...
Nuclear Chemistry - Mona Shores Blogs
... bomb contained 5kg of plutonium, but created an explosion equivalent to18, 600 tons of TNT!!! • So, since we bent the law a little, just a little, we use a revised version of the law: – This law is referred to as the Law of Conservation of mass and energy. ...
... bomb contained 5kg of plutonium, but created an explosion equivalent to18, 600 tons of TNT!!! • So, since we bent the law a little, just a little, we use a revised version of the law: – This law is referred to as the Law of Conservation of mass and energy. ...
Name
... C. Beneficial uses of nuclear radiation 1. Smoke detectors help to save lives a. In a smoke alarm, a small alpha-emitting isotope detects smoke particles in the air. 2. Nuclear radiation is used to detect diseases a. A radioactive tracer is a radioactive material that is added to a substance so tha ...
... C. Beneficial uses of nuclear radiation 1. Smoke detectors help to save lives a. In a smoke alarm, a small alpha-emitting isotope detects smoke particles in the air. 2. Nuclear radiation is used to detect diseases a. A radioactive tracer is a radioactive material that is added to a substance so tha ...
Nuclear Fission and Fusion Notes
... *Stars and Sun: Energy is produced when H nuclei combine *We need a large amount of energy to start a fusion reaction so that the H nuclei can be brought together and overcome the electric forces ...
... *Stars and Sun: Energy is produced when H nuclei combine *We need a large amount of energy to start a fusion reaction so that the H nuclei can be brought together and overcome the electric forces ...
nuclear reactions
... rad = radiation absorbed dose = amount of radiation that results in absorption of 1 10-5 J g-1 of absorbing material. It varies with the type of radiation and its source. All sources of nuclear radiation are distinguishable by type and energy. ...
... rad = radiation absorbed dose = amount of radiation that results in absorption of 1 10-5 J g-1 of absorbing material. It varies with the type of radiation and its source. All sources of nuclear radiation are distinguishable by type and energy. ...
A Conceptual Introduction to Chemistry, First Edition
... Plutonium When nonfissionable U-238 captures a fast neutron, it eventually forms the fissionable nuclide of plutonium, Pu-239, which can support a chain reaction. Plutonium is a transuranium element, meaning that it has an atomic number greater than the 92 of uranium. The fissionable plutonium prod ...
... Plutonium When nonfissionable U-238 captures a fast neutron, it eventually forms the fissionable nuclide of plutonium, Pu-239, which can support a chain reaction. Plutonium is a transuranium element, meaning that it has an atomic number greater than the 92 of uranium. The fissionable plutonium prod ...
first lecture - الدكتورة / زينب بنت زكي الفل
... have isotopes of different mass numbers. Their nuclei contain the same number of protons and different number of neutrons. Isotopes were first discovered amongst naturally radioactive elements J.J. Thomson while exploring the properties of positive rays by the parabola method, was the first to disco ...
... have isotopes of different mass numbers. Their nuclei contain the same number of protons and different number of neutrons. Isotopes were first discovered amongst naturally radioactive elements J.J. Thomson while exploring the properties of positive rays by the parabola method, was the first to disco ...
Chapter 1
... • Half-life (t1/2) - the time required for one-half of a given quantity of a substance to undergo change • Each radioactive isotope has its own half-life – Ranges from a fraction of a second to a billion years – The shorter the half-life, the more unstable the isotope ...
... • Half-life (t1/2) - the time required for one-half of a given quantity of a substance to undergo change • Each radioactive isotope has its own half-life – Ranges from a fraction of a second to a billion years – The shorter the half-life, the more unstable the isotope ...
Nuclear Stability Notes
... and works within a very short distance. Neutrons act as insulation, since they have no charge, but have the strong force to bring other nucleons (protons and neutrons) together. ...
... and works within a very short distance. Neutrons act as insulation, since they have no charge, but have the strong force to bring other nucleons (protons and neutrons) together. ...
Chapter 3
... As the water boils, heat from the hot stove burner and pan radiates into the surrounding ...
... As the water boils, heat from the hot stove burner and pan radiates into the surrounding ...
APES-Chapter-3
... As the water boils, heat from the hot stove burner and pan radiates into the surrounding ...
... As the water boils, heat from the hot stove burner and pan radiates into the surrounding ...
Chemistry Standard 2A-Nucleus Section 20.1
... a. electric force c. tracer b. strong force d. Isotope 7. Nuclei with too many or too few neutrons are…. Page answer is found ____________ a. never found. c. unnatural. b. unstable. d. stable. 8. The process of nuclear change in a radioactive atom is called Page answer is found ____________ a. nucle ...
... a. electric force c. tracer b. strong force d. Isotope 7. Nuclei with too many or too few neutrons are…. Page answer is found ____________ a. never found. c. unnatural. b. unstable. d. stable. 8. The process of nuclear change in a radioactive atom is called Page answer is found ____________ a. nucle ...
GRAMMAR: verb tenses
... many neutrons cannot stick together either. As the Chart of the Nuclides indicates, roughly the same number of neutrons and protons are needed for light nuclei, while for heavier nuclei a few more neutrons are needed (about half again as many neutrons as protons for the heaviest .nuclides, such as U ...
... many neutrons cannot stick together either. As the Chart of the Nuclides indicates, roughly the same number of neutrons and protons are needed for light nuclei, while for heavier nuclei a few more neutrons are needed (about half again as many neutrons as protons for the heaviest .nuclides, such as U ...
Alpha decay
... Not all combinations of protons and neutrons are created equal Some are more unstable than others. If they are unstable they will do one of the ...
... Not all combinations of protons and neutrons are created equal Some are more unstable than others. If they are unstable they will do one of the ...
Alpha Beta Fission Fusion
... undergoing radioactive decay. For example, Bi210 can undergo decay to Tl206 with a T½ of five days. Bi215, by comparison, undergoes decay to Po215 with a T½ of 7.6 minutes, and Bi208 undergoes yet another mode of radioactive decay (called electron capture) with a T½ of 368,000 years! Stimulated ...
... undergoing radioactive decay. For example, Bi210 can undergo decay to Tl206 with a T½ of five days. Bi215, by comparison, undergoes decay to Po215 with a T½ of 7.6 minutes, and Bi208 undergoes yet another mode of radioactive decay (called electron capture) with a T½ of 368,000 years! Stimulated ...
Independent Study: Nuclear Chemistry
... on facts, about nuclear testing be sure to reference health and environmental issues. Go to the following website about North Korea and recent nuclear testing and think about the ramifications of what NK is doing and how it effects the world : http://www.businessinsider.com/north-koreas-last-nuclear ...
... on facts, about nuclear testing be sure to reference health and environmental issues. Go to the following website about North Korea and recent nuclear testing and think about the ramifications of what NK is doing and how it effects the world : http://www.businessinsider.com/north-koreas-last-nuclear ...
Independent Study: Nuclear Chemistry
... on facts, about nuclear testing be sure to reference health and environmental issues. Go to the following website about North Korea and recent nuclear testing and think about the ramifications of what NK is doing and how it effects the world : http://www.businessinsider.com/north-koreas-last-nuclear ...
... on facts, about nuclear testing be sure to reference health and environmental issues. Go to the following website about North Korea and recent nuclear testing and think about the ramifications of what NK is doing and how it effects the world : http://www.businessinsider.com/north-koreas-last-nuclear ...
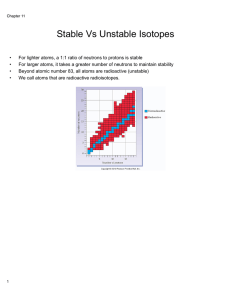
Stable Vs Unstable Isotopes
... 2. Beta decay: An atom emits a beta particle in the form of an electron when a neutron is changed into a proton. The atomic number increases by 1 while the mass number remains the same. ...
... 2. Beta decay: An atom emits a beta particle in the form of an electron when a neutron is changed into a proton. The atomic number increases by 1 while the mass number remains the same. ...
1. Nucleons Protons and neutrons 2. Nuclide A atom in
... • Positron emission- elements below the band of stability have too many protons. A proton can be turned into a neutron by emitting a positron – a particle that has the same mass as an electron, but has a positive charge and is emitted from the nucleus during some types of radioactive decay. ...
... • Positron emission- elements below the band of stability have too many protons. A proton can be turned into a neutron by emitting a positron – a particle that has the same mass as an electron, but has a positive charge and is emitted from the nucleus during some types of radioactive decay. ...
Nuclear Chemistry - Ector County ISD.
... example, the radioactive element bismuth (210Bi) can undergo alpha decay to form the element thallium (206Tl) with a reaction half-life equal to five days. If we begin an experiment starting with 100 g of bismuth in a sealed lead container, after five days we will have 50 g of bismuth and 50 g of th ...
... example, the radioactive element bismuth (210Bi) can undergo alpha decay to form the element thallium (206Tl) with a reaction half-life equal to five days. If we begin an experiment starting with 100 g of bismuth in a sealed lead container, after five days we will have 50 g of bismuth and 50 g of th ...
Radioactivity - Mrs. Sjuts` Science Site
... give the measured amounts of the isotope are calculated ! The number of half-‐lives is the amount of time that has passed since the isotope began to decay AND usually is the same as the age ...
... give the measured amounts of the isotope are calculated ! The number of half-‐lives is the amount of time that has passed since the isotope began to decay AND usually is the same as the age ...
Nuclear and radiation accidents and incidents

A nuclear and radiation accident is defined by the International Atomic Energy Agency (IAEA) as ""an event that has led to significant consequences to people, the environment or the facility."" Examples include lethal effects to individuals, large radioactivity release to the environment, or reactor core melt."" The prime example of a ""major nuclear accident"" is one in which a reactor core is damaged and significant amounts of radioactivity are released, such as in the Chernobyl disaster in 1986.The impact of nuclear accidents has been a topic of debate practically since the first nuclear reactors were constructed in 1954. It has also been a key factor in public concern about nuclear facilities. Some technical measures to reduce the risk of accidents or to minimize the amount of radioactivity released to the environment have been adopted. Despite the use of such measures, human error remains, and ""there have been many accidents with varying impacts as well near misses and incidents"".Worldwide there have been 99 accidents at nuclear power plants. Fifty-seven accidents have occurred since the Chernobyl disaster, and 57% (56 out of 99) of all nuclear-related accidents have occurred in the USA. Serious nuclear power plant accidents include the Fukushima Daiichi nuclear disaster (2011), Chernobyl disaster (1986), Three Mile Island accident (1979), and the SL-1 accident (1961).Nuclear-powered submarine core meltdown and other mishaps include the K-19 (1961), K-11 (1965), K-27 (1968), K-140 (1968), K-429 (1970), K-222 (1980), K-314 (1985), and K-431 (1985). Serious radiation accidents include the Kyshtym disaster, Windscale fire, radiotherapy accident in Costa Rica, radiotherapy accident in Zaragoza, radiation accident in Morocco, Goiania accident, radiation accident in Mexico City, radiotherapy unit accident in Thailand, and the Mayapuri radiological accident in India.The IAEA maintains a website reporting recent accidents.