AP Chem
... 3. Describe how alpha, beta, and gamma rays each behave when passed through an electric field. Draw a picture. 4. Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? ...
... 3. Describe how alpha, beta, and gamma rays each behave when passed through an electric field. Draw a picture. 4. Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? ...
Chapter 9 Nuclear Radiation 9.1 Natural Radioactivity Radioactive
... In a balanced nuclear equation, the sum of the mass numbers and the sum of the atomic numbers are equal for the nuclei of the reactants and the products. ...
... In a balanced nuclear equation, the sum of the mass numbers and the sum of the atomic numbers are equal for the nuclei of the reactants and the products. ...
Ch9
... In a balanced nuclear equation, the sum of the mass numbers and the sum of the atomic numbers are equal for the nuclei of the reactants and the products. ...
... In a balanced nuclear equation, the sum of the mass numbers and the sum of the atomic numbers are equal for the nuclei of the reactants and the products. ...
Half-life and Radioactive Decay guided notes
... amount of atoms in the sample. This means that the half-life of a specific isotope will be the whether you have one million moles or one mole of the atoms. The graph below represents the radioactive decay of a hypothetical element. Notice that the time it takes for the amount of radioactive material ...
... amount of atoms in the sample. This means that the half-life of a specific isotope will be the whether you have one million moles or one mole of the atoms. The graph below represents the radioactive decay of a hypothetical element. Notice that the time it takes for the amount of radioactive material ...
The Band of Stability
... Introduction: Radioactivity is the spontaneous emission of radiation by nuclei. Radioactive decay changes the nature and identity of an atom’s nucleus. This occurs for a specific reason. Elements from hydrogen to lead (atomic numbers 1-82) have stable isotopes in which the tendency of protons to rep ...
... Introduction: Radioactivity is the spontaneous emission of radiation by nuclei. Radioactive decay changes the nature and identity of an atom’s nucleus. This occurs for a specific reason. Elements from hydrogen to lead (atomic numbers 1-82) have stable isotopes in which the tendency of protons to rep ...
Purging - Sellafield Suppliers
... The primary use of stainless steel is their general corrosion resistance. Exposure to heat and oxygen during the welding process can severely reduce the corrosion resistance of stainless steel and results in what is known as heat tint (oxidisation). Shielding the weld during welding with an inert ga ...
... The primary use of stainless steel is their general corrosion resistance. Exposure to heat and oxygen during the welding process can severely reduce the corrosion resistance of stainless steel and results in what is known as heat tint (oxidisation). Shielding the weld during welding with an inert ga ...
1 AP Chemistry Chapter 21 - The Nucleus: A Chemist`s View 21.1
... AP Chemistry Chapter 21 - The Nucleus: A Chemist’s View 21.1 Nuclear Stability and Radioactive Decay A. Radioactive Decay 1. Decomposition forming a different nucleus and producing one or more particles a. Total mass number and atomic number must be conserved in any nuclear change ...
... AP Chemistry Chapter 21 - The Nucleus: A Chemist’s View 21.1 Nuclear Stability and Radioactive Decay A. Radioactive Decay 1. Decomposition forming a different nucleus and producing one or more particles a. Total mass number and atomic number must be conserved in any nuclear change ...
Chapter 18 Notes
... AP Chemistry Chapter 18 - The Nucleus: A Chemist’s View 18.1 Nuclear Stability and Radioactive Decay A. Radioactive Decay 1. Decomposition forming a different nucleus and producing one or more particles a. Total mass number and atomic number must be conserved in any nuclear change ...
... AP Chemistry Chapter 18 - The Nucleus: A Chemist’s View 18.1 Nuclear Stability and Radioactive Decay A. Radioactive Decay 1. Decomposition forming a different nucleus and producing one or more particles a. Total mass number and atomic number must be conserved in any nuclear change ...
Unit 3 – Atomic Structure
... is to reduce mass Nuclei shed mass in the form of a helium nucleus to become more stable Helium nucleus that is released is ionized and called and Alpha Particle ...
... is to reduce mass Nuclei shed mass in the form of a helium nucleus to become more stable Helium nucleus that is released is ionized and called and Alpha Particle ...
Unit 3 Study Guide: Atomic Structure and Nuclear
... _______________ 34. If more than a critical mass is present in a sample, that sample is said to have supercritical mass. ...
... _______________ 34. If more than a critical mass is present in a sample, that sample is said to have supercritical mass. ...
Chapter 9
... • energy is emitted from an unstable nucleus, indicated by m following the mass number. • the mass number and the atomic number of the new nucleus are the same. ...
... • energy is emitted from an unstable nucleus, indicated by m following the mass number. • the mass number and the atomic number of the new nucleus are the same. ...
radioisotopes and radiotherapy - video
... 10% Cosmic Rays from Sun and outer space, 35% medical and industrial use. 13. Ionising radiation can kill cells or change the way they function. 14. High doses of radiation from beta particles. 15. Low dose of gamma radiation. 16. In nuclear reactors (or cyclotrons, discussed later in video). In a n ...
... 10% Cosmic Rays from Sun and outer space, 35% medical and industrial use. 13. Ionising radiation can kill cells or change the way they function. 14. High doses of radiation from beta particles. 15. Low dose of gamma radiation. 16. In nuclear reactors (or cyclotrons, discussed later in video). In a n ...
Chapter 7 Worksheet
... Alpha decayAlpha particle Beta decay Beta particle CANDU Chain reaction Daughter isotope Decay curve FissionFusionGamma decay Gamma particle Half-life Hazardous wastes Induced nuclear reaction Isotopes Light Mass number Nuclear equation Nuclear reaction Parent isotope Potassium-40 clock Radiation Ra ...
... Alpha decayAlpha particle Beta decay Beta particle CANDU Chain reaction Daughter isotope Decay curve FissionFusionGamma decay Gamma particle Half-life Hazardous wastes Induced nuclear reaction Isotopes Light Mass number Nuclear equation Nuclear reaction Parent isotope Potassium-40 clock Radiation Ra ...
2 α
... and create a nuclear reaction that produces neutrons. The neutrons bombard everything in the reactor, including the stable isotope, and the it absorbs the neutrons into its nucleus. Radioactive Iridium-192 is produced in a small-scale nuclear reactor by bombarding naturally occurring Iridium-191 wit ...
... and create a nuclear reaction that produces neutrons. The neutrons bombard everything in the reactor, including the stable isotope, and the it absorbs the neutrons into its nucleus. Radioactive Iridium-192 is produced in a small-scale nuclear reactor by bombarding naturally occurring Iridium-191 wit ...
Nuclear Radiation1516
... one nuclear reaction causes an average of one or more nuclear reactions, thus leading to a self-propagating number of these reactions. The nuclear reaction is the fission of heavy isotopes. • The nuclear chain reaction is unique ...
... one nuclear reaction causes an average of one or more nuclear reactions, thus leading to a self-propagating number of these reactions. The nuclear reaction is the fission of heavy isotopes. • The nuclear chain reaction is unique ...
NUCLEAR CHEMISTRY
... and likely symptoms of nausea, fatigue and vomiting for one or two days. 5 to 50 RAD: Slight blood changes may be detected by medical evaluation. Less than 5 RAD: No immediate observable effects. ...
... and likely symptoms of nausea, fatigue and vomiting for one or two days. 5 to 50 RAD: Slight blood changes may be detected by medical evaluation. Less than 5 RAD: No immediate observable effects. ...
Radioactivity
... • Nuclear fuel is usually a supercritical mass of U-235 enriched uranium • Reaction is promoted by a moderator - a material that slows neutrons down so they will cause fission - usually carbon or D2O ...
... • Nuclear fuel is usually a supercritical mass of U-235 enriched uranium • Reaction is promoted by a moderator - a material that slows neutrons down so they will cause fission - usually carbon or D2O ...
Radioactivity
... • Nuclear fuel is usually a supercritical mass of U-235 enriched uranium • Reaction is promoted by a moderator - a material that slows neutrons down so they will cause fission - usually carbon or D2O ...
... • Nuclear fuel is usually a supercritical mass of U-235 enriched uranium • Reaction is promoted by a moderator - a material that slows neutrons down so they will cause fission - usually carbon or D2O ...
Topic 14 - Lloyd Crosby
... b. 1 rem = 1 rad x 1 RBE c. RBE is from Relative Biological Effectiveness. d. The RBE factor depends on how destructive to biological tissues a type of radiation happens to be for the same amount of energy delivered to the tissue e. RBEs for selected radiation (1) X-rays: RBE = 0.7 (2) beta: ...
... b. 1 rem = 1 rad x 1 RBE c. RBE is from Relative Biological Effectiveness. d. The RBE factor depends on how destructive to biological tissues a type of radiation happens to be for the same amount of energy delivered to the tissue e. RBEs for selected radiation (1) X-rays: RBE = 0.7 (2) beta: ...
E = mc2 (Einstein)
... happened at Three Mile Island or Chernobyl, or even to be able to take an informed stand on the future of nuclear power in our society. ...
... happened at Three Mile Island or Chernobyl, or even to be able to take an informed stand on the future of nuclear power in our society. ...
Lithium 6.941 - mrkearsley.com
... A classic example of a fission reaction is that of UU235 where stray neutron strikes an atom of UU-235. It absorbs the neutron and becomes an unstable atom of UU-236. 236 This undergoes fission. fission More neutrons are released in the reaction. These neutrons can strike other UU-235 atoms to initi ...
... A classic example of a fission reaction is that of UU235 where stray neutron strikes an atom of UU-235. It absorbs the neutron and becomes an unstable atom of UU-236. 236 This undergoes fission. fission More neutrons are released in the reaction. These neutrons can strike other UU-235 atoms to initi ...
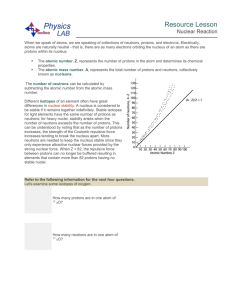
Resource Lesson Nuclear Reaction When we speak of atoms, we
... β represents an electron, with a charge of -1, which has been released from the nucleus when a neutron decays into a proton, an electron and an antineutrino by way of the weak force: ...
... β represents an electron, with a charge of -1, which has been released from the nucleus when a neutron decays into a proton, an electron and an antineutrino by way of the weak force: ...
Chapter 25 Nuclear Chemistry
... Nuclear power is good because the fuel lasts longer than fossil fuels with no emissions of CO2(greenhouse gas) or SO2(acid rain gas). 1 kg of Uranium-235 releases more energy than 20,000 tons of dynamite (that's 40 million pounds!) Nuclear power is bad because the nuclei left after fission are radio ...
... Nuclear power is good because the fuel lasts longer than fossil fuels with no emissions of CO2(greenhouse gas) or SO2(acid rain gas). 1 kg of Uranium-235 releases more energy than 20,000 tons of dynamite (that's 40 million pounds!) Nuclear power is bad because the nuclei left after fission are radio ...
Nuclear and radiation accidents and incidents

A nuclear and radiation accident is defined by the International Atomic Energy Agency (IAEA) as ""an event that has led to significant consequences to people, the environment or the facility."" Examples include lethal effects to individuals, large radioactivity release to the environment, or reactor core melt."" The prime example of a ""major nuclear accident"" is one in which a reactor core is damaged and significant amounts of radioactivity are released, such as in the Chernobyl disaster in 1986.The impact of nuclear accidents has been a topic of debate practically since the first nuclear reactors were constructed in 1954. It has also been a key factor in public concern about nuclear facilities. Some technical measures to reduce the risk of accidents or to minimize the amount of radioactivity released to the environment have been adopted. Despite the use of such measures, human error remains, and ""there have been many accidents with varying impacts as well near misses and incidents"".Worldwide there have been 99 accidents at nuclear power plants. Fifty-seven accidents have occurred since the Chernobyl disaster, and 57% (56 out of 99) of all nuclear-related accidents have occurred in the USA. Serious nuclear power plant accidents include the Fukushima Daiichi nuclear disaster (2011), Chernobyl disaster (1986), Three Mile Island accident (1979), and the SL-1 accident (1961).Nuclear-powered submarine core meltdown and other mishaps include the K-19 (1961), K-11 (1965), K-27 (1968), K-140 (1968), K-429 (1970), K-222 (1980), K-314 (1985), and K-431 (1985). Serious radiation accidents include the Kyshtym disaster, Windscale fire, radiotherapy accident in Costa Rica, radiotherapy accident in Zaragoza, radiation accident in Morocco, Goiania accident, radiation accident in Mexico City, radiotherapy unit accident in Thailand, and the Mayapuri radiological accident in India.The IAEA maintains a website reporting recent accidents.