Elements and Compounds
... • Atoms of the same element can have different masses. • They always have the same number of protons, but they can have different numbers of neutrons in their nuclei. • The difference in the number of neutrons accounts for the difference in ...
... • Atoms of the same element can have different masses. • They always have the same number of protons, but they can have different numbers of neutrons in their nuclei. • The difference in the number of neutrons accounts for the difference in ...
Chapter 4 PPT
... If two elements form more than one compound between them, then the ratios of the masses of the second element which combine with a fixed mass of the first element will be ratios of small whole numbers. (not decimals) For example, H2O and ...
... If two elements form more than one compound between them, then the ratios of the masses of the second element which combine with a fixed mass of the first element will be ratios of small whole numbers. (not decimals) For example, H2O and ...
Chapter 4, Lesson 2: The Periodic Table
... Each student should find and present some basic information about their element to the class. The presentation can be in the form of a poster, pamphlet, PowerPoint presentation or other form. The presentations should be short and can include: atom name, atomic number, derivation of name, when and wh ...
... Each student should find and present some basic information about their element to the class. The presentation can be in the form of a poster, pamphlet, PowerPoint presentation or other form. The presentations should be short and can include: atom name, atomic number, derivation of name, when and wh ...
Chapter 4 Atomic Structure
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
The Periodic Table of the Elements
... C. How many electrons are in the atom? An atom is neutral, which means that the number of electrons is equal to the number of protons. An atom of Pb has 82 protons and ...
... C. How many electrons are in the atom? An atom is neutral, which means that the number of electrons is equal to the number of protons. An atom of Pb has 82 protons and ...
The Atom - TeacherWeb
... Isotopes are atoms of the same element that have different masses (ie same protons, different neutrons) Isotopes are written using the name of the element followed by a hyphen and then the mass number. Ex: Hydrogen-1, Hydrogen-2, & Hydrogen-3 are isotopes ...
... Isotopes are atoms of the same element that have different masses (ie same protons, different neutrons) Isotopes are written using the name of the element followed by a hyphen and then the mass number. Ex: Hydrogen-1, Hydrogen-2, & Hydrogen-3 are isotopes ...
Homework #1 Atoms
... 1. The number of ____________ in the nucleus of an atom is the atomic ____________ of that element. Because atoms are electrically neutral, the number of protons and ____________ in an atom are equal. 2. The sum of the _____________ and neutrons is the mass number. 3. Atoms of the same element are i ...
... 1. The number of ____________ in the nucleus of an atom is the atomic ____________ of that element. Because atoms are electrically neutral, the number of protons and ____________ in an atom are equal. 2. The sum of the _____________ and neutrons is the mass number. 3. Atoms of the same element are i ...
Atom
... • Atoms of an element don’t always have the same # of neutrons. • These atoms are called isotopes. ...
... • Atoms of an element don’t always have the same # of neutrons. • These atoms are called isotopes. ...
Chemistry: Unit Organizer Name 6-__ Matter has physical properties
... undergo a change that results in a new substance. (Remember: there are only two kinds of chemical properties: ex. Eukaryotes ability to react with other substances or flammability). Chemical change: change in a chemical property in which the identity of the matter does change into a new substance (e ...
... undergo a change that results in a new substance. (Remember: there are only two kinds of chemical properties: ex. Eukaryotes ability to react with other substances or flammability). Chemical change: change in a chemical property in which the identity of the matter does change into a new substance (e ...
2.1 The Nature of Matter Notes
... Ionic Bonds These oppositely charged ions have a strong attraction for each other, forming an ionic bond. ...
... Ionic Bonds These oppositely charged ions have a strong attraction for each other, forming an ionic bond. ...
7 - Edmodo
... using Table 5.2 on page 182 answer the following in your notebook: 1) Find and list the names and symbols of 5 elements beginning with the letter C 2) Find and list the names and symbols of 3 elements named after places 3) Find and list the names and symbols for any 5 non-metals with symbols compose ...
... using Table 5.2 on page 182 answer the following in your notebook: 1) Find and list the names and symbols of 5 elements beginning with the letter C 2) Find and list the names and symbols of 3 elements named after places 3) Find and list the names and symbols for any 5 non-metals with symbols compose ...
Chapter 4 Atomic Structure
... 3) Atoms of different elements combine in simple whole-number ratios to form chemical compounds 4) In chemical reactions, atoms are combined, separated, or rearranged – but never changed into atoms of another element. ...
... 3) Atoms of different elements combine in simple whole-number ratios to form chemical compounds 4) In chemical reactions, atoms are combined, separated, or rearranged – but never changed into atoms of another element. ...
Atomic masses are weighted averages.
... 1. All elements are composed of tiny indivisible particles called atoms. Atoms are not indivisible – they are made of subatomic particles 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. Every atom has at least one isotope; one a ...
... 1. All elements are composed of tiny indivisible particles called atoms. Atoms are not indivisible – they are made of subatomic particles 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. Every atom has at least one isotope; one a ...
Chapter 4 PowerPoint
... atoms of any one element differ from those of any other element. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. Chemical reactions occur when atoms are separated, joined, or rearranged. Atoms of one element, however, ...
... atoms of any one element differ from those of any other element. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. Chemical reactions occur when atoms are separated, joined, or rearranged. Atoms of one element, however, ...
Oxidation numbers
... individual oxidation numbers of the elements adds up to the charge on the ion Eg in CO32- C = +4 and O = -2 sum = +4 + (3 x -2) = -2 ...
... individual oxidation numbers of the elements adds up to the charge on the ion Eg in CO32- C = +4 and O = -2 sum = +4 + (3 x -2) = -2 ...
Atoms and Elements: Are they Related?
... • What are the most commonly occurring elements in the food labels? • What items seemed to have the most amount of elements in them? • Can you predict what that means about the food item? • Why do you think the baby formula has such a variety of elements? • Can you predict what the other items on th ...
... • What are the most commonly occurring elements in the food labels? • What items seemed to have the most amount of elements in them? • Can you predict what that means about the food item? • Why do you think the baby formula has such a variety of elements? • Can you predict what the other items on th ...
Unit 2 Spiraling
... 18. If the average atomic mass of C is 12.011, which isotope do you think is most abundant C-12, C-13 or C-14? Explain your answer. The chemical reaction where elements react (bond together) to form a compound is called synthesis. Example: Hydrogen gas and oxygen gas react to synthesize water. 2H2 + ...
... 18. If the average atomic mass of C is 12.011, which isotope do you think is most abundant C-12, C-13 or C-14? Explain your answer. The chemical reaction where elements react (bond together) to form a compound is called synthesis. Example: Hydrogen gas and oxygen gas react to synthesize water. 2H2 + ...
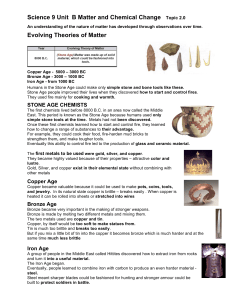
An understanding of the nature of matter has developed
... reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 98 elements exist naturally although some are found only in trace amounts and were synthesized in laboratories before being found in nature.[n 1] Elements with atomic numbers from 99 to 118 have only b ...
... reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 98 elements exist naturally although some are found only in trace amounts and were synthesized in laboratories before being found in nature.[n 1] Elements with atomic numbers from 99 to 118 have only b ...
- Catalyst
... Question 7: Fill in the blanks of the statements below with the words in the box. Note, you will only use each word once. 1. A positively charged ion is a(n) ____________________. 2. A proton has a +1 _______________________________. 3. Like charges _________________________ each other. ...
... Question 7: Fill in the blanks of the statements below with the words in the box. Note, you will only use each word once. 1. A positively charged ion is a(n) ____________________. 2. A proton has a +1 _______________________________. 3. Like charges _________________________ each other. ...
24 Sept 08 - Seattle Central College
... Of the following, which is most likely to become a cation as a result of a chemical reaction? What would be the charge on that cation? 1. N; –3 2. Ne; +1 3. Na: +1 ...
... Of the following, which is most likely to become a cation as a result of a chemical reaction? What would be the charge on that cation? 1. N; –3 2. Ne; +1 3. Na: +1 ...
Chapter 4, Lesson 2: The Periodic Table
... or neutron). The majority of the atomic mass is contributed by the protons and neutrons. For any element in the periodic table, the number of electrons in an atom of that element always equals the number of protons in the nucleus. But this is not true for neutrons. Atoms of the same element can have ...
... or neutron). The majority of the atomic mass is contributed by the protons and neutrons. For any element in the periodic table, the number of electrons in an atom of that element always equals the number of protons in the nucleus. But this is not true for neutrons. Atoms of the same element can have ...
Chapter 5 Atomic Structure & the Periodic Table
... • Magnesium = region in Greece known as Magnesia • Lithium = Greek word lithos, meaning stone • Neptunium = after the planet Neptune • Hydrogen (H), Sulfur (S), Carbon (C) • Gold (Au), Lead (Pb), Iron (Fe), Copper (Cu) = symbols come from latin names. ...
... • Magnesium = region in Greece known as Magnesia • Lithium = Greek word lithos, meaning stone • Neptunium = after the planet Neptune • Hydrogen (H), Sulfur (S), Carbon (C) • Gold (Au), Lead (Pb), Iron (Fe), Copper (Cu) = symbols come from latin names. ...
ChemCh4of2011
... Dalton’s Atomic Theory (1808) Matter is composed of extremely small particles called atoms ...
... Dalton’s Atomic Theory (1808) Matter is composed of extremely small particles called atoms ...
Bean Bag Lab
... and equal in mass was simple yet revolutionary. Unfortunately, it was not quite right. More research started to show that atoms of the same element could have different masses. These atoms were call isotopes. Isotopes have the same # of protons and electrons but different # of neutrons. Isotopes are ...
... and equal in mass was simple yet revolutionary. Unfortunately, it was not quite right. More research started to show that atoms of the same element could have different masses. These atoms were call isotopes. Isotopes have the same # of protons and electrons but different # of neutrons. Isotopes are ...
3.1 The Element A. Abundances of Eleme B. Names and Symbols
... he chemical elements are essential to each of us in our daily lives. The most important element is carbon, which is found in virtually all of the molecules that make up the living cell. Although certain elements are present in our bodies in tiny amounts, these elements can have profound effects on o ...
... he chemical elements are essential to each of us in our daily lives. The most important element is carbon, which is found in virtually all of the molecules that make up the living cell. Although certain elements are present in our bodies in tiny amounts, these elements can have profound effects on o ...