Types of Materials
... compressive strength. Where additional tensile strength is required concrete is usually reinforced by steel. Common uses for concrete include all kinds of building work, railway lines and road building. ...
... compressive strength. Where additional tensile strength is required concrete is usually reinforced by steel. Common uses for concrete include all kinds of building work, railway lines and road building. ...
Heat Treating of Non Ferous Alloys
... Most heat treatments begin by heating an alloy beyond the upper transformation (A3) temperature. The alloy will usually be held at this temperature long enough for the heat to completely penetrate the alloy, thereby bringing it into a complete solid solution. Since a smaller grain size usually enhan ...
... Most heat treatments begin by heating an alloy beyond the upper transformation (A3) temperature. The alloy will usually be held at this temperature long enough for the heat to completely penetrate the alloy, thereby bringing it into a complete solid solution. Since a smaller grain size usually enhan ...
smart_materials_1 - Aldercar High School
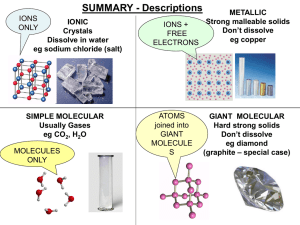
... Regular structure, layers slide CONDUCT: YES (very well) Free electrons between ions ...
... Regular structure, layers slide CONDUCT: YES (very well) Free electrons between ions ...
Kinds and Characteristics of Hydrogen Storage Alloy
... Hydrogen is readily incorporated in hydrogen storage alloys, and when the concentration of hydrogen in the alloy comes to a certain limit, metal hydrides are formed. As a result, hydrogen occupies certain sites among the metal lattices. The properties of hydrogen storage in an alloy are determined b ...
... Hydrogen is readily incorporated in hydrogen storage alloys, and when the concentration of hydrogen in the alloy comes to a certain limit, metal hydrides are formed. As a result, hydrogen occupies certain sites among the metal lattices. The properties of hydrogen storage in an alloy are determined b ...
History and Current Status of the Plastics Industry
... List various ingredients of cast iron, steels, and stainless steels Recognize and use the nomenclature associated with steels Recognize the major regions and ranges of the iron-carbon phase diagram List the major shapes in which ferrous metal products are available List and describe the various allo ...
... List various ingredients of cast iron, steels, and stainless steels Recognize and use the nomenclature associated with steels Recognize the major regions and ranges of the iron-carbon phase diagram List the major shapes in which ferrous metal products are available List and describe the various allo ...
Effects of Toxic Materials
... This means that some alloys of aluminium increase in hardness and strength over a period of a few days when they have been quenched from high temperatures. ...
... This means that some alloys of aluminium increase in hardness and strength over a period of a few days when they have been quenched from high temperatures. ...
Annealing Stages
... • Mention some factors that may affect the actual cooling rate of an air cooled specimen. • What effects do the alloying additives have on the microstructure and mechanical properties of air cooled (normalized) steel specimens? • What effects do the alloying additives have on the microstructure and ...
... • Mention some factors that may affect the actual cooling rate of an air cooled specimen. • What effects do the alloying additives have on the microstructure and mechanical properties of air cooled (normalized) steel specimens? • What effects do the alloying additives have on the microstructure and ...
Introduction to Dental Materials
... They are bonded by covalent bonding along the backbone, and ionic bonding . Polymers are prepared in form of dough then shaped into desired shapes. They harden by: -physical reaction,(cooling, or evapration). Waxes, -chemical reaction, ...
... They are bonded by covalent bonding along the backbone, and ionic bonding . Polymers are prepared in form of dough then shaped into desired shapes. They harden by: -physical reaction,(cooling, or evapration). Waxes, -chemical reaction, ...
Section3a - Lyle School of Engineering
... atoms are located in the Fe cells. – Solid solubility increases with temperature as it does with liquid solutions. Correspondingly, solute atoms precipitate on cooling (by diffusion) out of the atoms of the solvent. – Substitutional solid solutions in which there is a long-range order of the two met ...
... atoms are located in the Fe cells. – Solid solubility increases with temperature as it does with liquid solutions. Correspondingly, solute atoms precipitate on cooling (by diffusion) out of the atoms of the solvent. – Substitutional solid solutions in which there is a long-range order of the two met ...
Applied Materials Science
... Utilize a variety of analytical skills in the interpretation of material properties. Collect laboratory data concerning material properties and analyze it using statistical methods. Function as part of a laboratory team. Communicate the results of laboratory experiments effectively. Understand some ...
... Utilize a variety of analytical skills in the interpretation of material properties. Collect laboratory data concerning material properties and analyze it using statistical methods. Function as part of a laboratory team. Communicate the results of laboratory experiments effectively. Understand some ...
Made in the Stars Notes
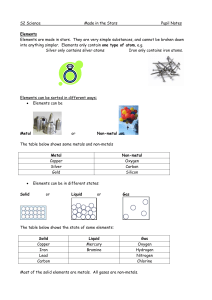
... at room temperature except for mercury, which is a liquid. Non-metal solids are usually brittle (they break easily). Non-metals can be solids, liquids or gases at room temperature. Non-metals usually have low melting and boiling points. They are poor conductors of electricity. The exception is graph ...
... at room temperature except for mercury, which is a liquid. Non-metal solids are usually brittle (they break easily). Non-metals can be solids, liquids or gases at room temperature. Non-metals usually have low melting and boiling points. They are poor conductors of electricity. The exception is graph ...
Aluminum Alloy Designation
... The first digit designates the major alloy Description of Alloy 1xxx Series designates alloy free (no alloys) and is at least 99% Aluminum of 99 percent or higher purity has many applications, especially in the electrical and chemical fields. These compositions are characterized by excellent corrosi ...
... The first digit designates the major alloy Description of Alloy 1xxx Series designates alloy free (no alloys) and is at least 99% Aluminum of 99 percent or higher purity has many applications, especially in the electrical and chemical fields. These compositions are characterized by excellent corrosi ...
9F Reactivity - Parrs Wood High School
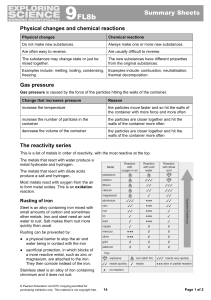
... Rusting of iron Steel is an alloy containing iron mixed with small amounts of carbon and sometimes other metals. Iron and steel need air and water to rust. Salt makes them rust more quickly than usual. Rusting can be prevented by: ● a physical barrier to stop the air and water being in contact with ...
... Rusting of iron Steel is an alloy containing iron mixed with small amounts of carbon and sometimes other metals. Iron and steel need air and water to rust. Salt makes them rust more quickly than usual. Rusting can be prevented by: ● a physical barrier to stop the air and water being in contact with ...
Document
... 3% Lead, 40% Zinc) are more properly regarded as brass alloys because they contain zinc as the main alloying ingredient. They are commonly used in architectural applications. Bismuth bronze is a bronze alloy with a composition of 52% copper, 30% nickel, 12% zinc, 5% lead, and 1% bismuth. It is able ...
... 3% Lead, 40% Zinc) are more properly regarded as brass alloys because they contain zinc as the main alloying ingredient. They are commonly used in architectural applications. Bismuth bronze is a bronze alloy with a composition of 52% copper, 30% nickel, 12% zinc, 5% lead, and 1% bismuth. It is able ...
THEMPROCESS_technology_GB
... temperature. One outstanding example is iron, the atoms of which from two types of three-dimensional lattice depending on the temperature: body centered cubic and, at higher temperatures, face centered cubic. The “Atomium”, the landmark of the Belgian capital of Brussels, is a model of a body center ...
... temperature. One outstanding example is iron, the atoms of which from two types of three-dimensional lattice depending on the temperature: body centered cubic and, at higher temperatures, face centered cubic. The “Atomium”, the landmark of the Belgian capital of Brussels, is a model of a body center ...
The subject of " Engineering Materials " deals with the study of
... crystals of regular shapes which called crystallites or grains of the metal . Metals in the solid state , and to same extent in the liqnid state possess high thermal and electrical conductivity . the electrical resistance of pure metals increases with the temperature Many metals display duper condu ...
... crystals of regular shapes which called crystallites or grains of the metal . Metals in the solid state , and to same extent in the liqnid state possess high thermal and electrical conductivity . the electrical resistance of pure metals increases with the temperature Many metals display duper condu ...
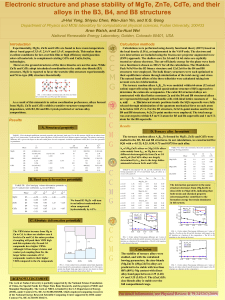
Electronic structure and phase stability of MgTe, ZnTe, CdTe, and
... relaxed through minimization of the quantum mechanical force on each atom to be below 0.02 eV/A. For the B3 structure, 3x3x3 k-point meshes and for the B4 and B8 structures, 4x2x2 k-point meshes were employed. The total energy was converged to within 0.5 meV/2-atom for B3 and B4 supercells and 1 meV ...
... relaxed through minimization of the quantum mechanical force on each atom to be below 0.02 eV/A. For the B3 structure, 3x3x3 k-point meshes and for the B4 and B8 structures, 4x2x2 k-point meshes were employed. The total energy was converged to within 0.5 meV/2-atom for B3 and B4 supercells and 1 meV ...
Ferrous and Non-Ferrous Metals
... A blast furnace is a type of metallurgical furnace used for smelting industrial metals, generally iron. In a blast furnace, fuel, ore and limestone as flux are continuously supplied through the top of the furnace, while air (sometimes with oxygen enrichment) is blown into the bottom of the chambe ...
... A blast furnace is a type of metallurgical furnace used for smelting industrial metals, generally iron. In a blast furnace, fuel, ore and limestone as flux are continuously supplied through the top of the furnace, while air (sometimes with oxygen enrichment) is blown into the bottom of the chambe ...
Ferrous and Non-Ferrous Metals
... A blast furnace is a type of metallurgical furnace used for smelting industrial metals, generally iron. In a blast furnace, fuel, ore and limestone as flux are continuously supplied through the top of the furnace, while air (sometimes with oxygen enrichment) is blown into the bottom of the chambe ...
... A blast furnace is a type of metallurgical furnace used for smelting industrial metals, generally iron. In a blast furnace, fuel, ore and limestone as flux are continuously supplied through the top of the furnace, while air (sometimes with oxygen enrichment) is blown into the bottom of the chambe ...
2.1.2 Alloys
... This is pretty much an empirical law, it does not pay to justify it theoretically. Again, it is not possible to produce an alloy with a resistivity smaller than one of its components. If you have intermetallic compounds in your phase diagram, use Nordheim's rule with the intermetallic phases as XA a ...
... This is pretty much an empirical law, it does not pay to justify it theoretically. Again, it is not possible to produce an alloy with a resistivity smaller than one of its components. If you have intermetallic compounds in your phase diagram, use Nordheim's rule with the intermetallic phases as XA a ...
2.2.2 Alloys
... This is pretty much an empirical law, it does not pay to justify it theoretically. Again, it is not possible to produce an alloy with a resistivity smaller than one of its components. If you have intermetallic compounds in your phase diagram, use Nordheim's rule with the intermetallic phases as XA a ...
... This is pretty much an empirical law, it does not pay to justify it theoretically. Again, it is not possible to produce an alloy with a resistivity smaller than one of its components. If you have intermetallic compounds in your phase diagram, use Nordheim's rule with the intermetallic phases as XA a ...
Syllabus High Temperature Structural Materials
... temperature materials is introduced. At high temperature, materials deform gradually when they are exposed even under low stress. This phenomenon is called creep behavior and governs material’s life. General introduction of creep behavior is then shown. Typical high temperature materials such as hea ...
... temperature materials is introduced. At high temperature, materials deform gradually when they are exposed even under low stress. This phenomenon is called creep behavior and governs material’s life. General introduction of creep behavior is then shown. Typical high temperature materials such as hea ...
Inweld Nibral, Aluminum Bronze 46
... Inweld Nibral is sometimes referred to its generic name, Nickel-Aluminum-Bronze. This filler metal is used for MIG and TIG welding of cast and wrought nickel-aluminum-bronze parts where there is a requirement for high resistance to corrosion, erosion and cavitation in salt or brackish water. The mos ...
... Inweld Nibral is sometimes referred to its generic name, Nickel-Aluminum-Bronze. This filler metal is used for MIG and TIG welding of cast and wrought nickel-aluminum-bronze parts where there is a requirement for high resistance to corrosion, erosion and cavitation in salt or brackish water. The mos ...
Metals & Metallurgy
... Oil was produced from the decomposition of once living life forms. Since crude oil is not found near any naturally occurring deposits of vanadium, then how did it find its way into this important resource? ...
... Oil was produced from the decomposition of once living life forms. Since crude oil is not found near any naturally occurring deposits of vanadium, then how did it find its way into this important resource? ...
Alloy

An alloy is a mixture of metals or a mixture of a metal and another element. Alloys are defined by metallic bonding character. An alloy may be a solid solution of metal elements (a single phase) or a mixture of metallic phases (two or more solutions). Intermetallic compounds are alloys with a defined stoichiometry and crystal structure. Zintl phases are also sometimes considered alloys depending on bond types (see also: Van Arkel-Ketelaar triangle for information on classifying bonding in binary compounds).Alloys are used in a wide variety of applications. In some cases, a combination of metals may reduce the overall cost of the material while preserving important properties. In other cases, the combination of metals imparts synergistic properties to the constituent metal elements such as corrosion resistance or mechanical strength. Examples of alloys are steel, solder, brass, pewter, duralumin, phosphor bronze and amalgams.The alloy constituents are usually measured by mass. Alloys are usually classified as substitutional or interstitial alloys, depending on the atomic arrangement that forms the alloy. They can be further classified as homogeneous (consisting of a single phase), or heterogeneous (consisting of two or more phases) or intermetallic.