Chapter 3, Elements, Atoms, Ions, and the Periodic Table Ans
... In what way(s) are the three orbitals in the 2p sublevel similar; in what way(s) are they different? they have the same shape and the same energy; they are oriented differently in space What requirement must be met in order for two electrons to coexist in the same orbital? ...
... In what way(s) are the three orbitals in the 2p sublevel similar; in what way(s) are they different? they have the same shape and the same energy; they are oriented differently in space What requirement must be met in order for two electrons to coexist in the same orbital? ...
Classification and Periodic Properties of Elements
... order of atomic weights and placed the elements with similar chemical properties together. For example, iodine having atomic weight 127 was placed after tellurium (atomic weight 128), together with fluorine, chlorine and bromine due to similarities in properties. The general plan of the modified Men ...
... order of atomic weights and placed the elements with similar chemical properties together. For example, iodine having atomic weight 127 was placed after tellurium (atomic weight 128), together with fluorine, chlorine and bromine due to similarities in properties. The general plan of the modified Men ...
Topic 1 notes - A
... 1 unit of charge is 1.602 x 10-19 coulombs. A proton is given a charge of +1 and an electron a charge of -1. All charges are measured in these units. 1 unit of mass is 1.661 x 10-27 kg. This is also not a convenient number, so we use “atomic mass units”. Since the mass of protons and neutrons varies ...
... 1 unit of charge is 1.602 x 10-19 coulombs. A proton is given a charge of +1 and an electron a charge of -1. All charges are measured in these units. 1 unit of mass is 1.661 x 10-27 kg. This is also not a convenient number, so we use “atomic mass units”. Since the mass of protons and neutrons varies ...
Periodic Table
... 'I'he arrangement of elements in the periodic table is useful as it is possible to predict the properties of an individual element from its position. -l'he Modern Periodic table is a table of all the chemical elements, in order of their atomic numbers so that elements with similar properties arc clo ...
... 'I'he arrangement of elements in the periodic table is useful as it is possible to predict the properties of an individual element from its position. -l'he Modern Periodic table is a table of all the chemical elements, in order of their atomic numbers so that elements with similar properties arc clo ...
Std 10th, Science and Technology, Maharashtra Board, English
... In addition to this, solved and practice problems are included which not only aim at covering the topic but also make students ready to face the competition. The topic-wise classified “question and answer” format of this book helps students in easy comprehension. Numerical problems included at the e ...
... In addition to this, solved and practice problems are included which not only aim at covering the topic but also make students ready to face the competition. The topic-wise classified “question and answer” format of this book helps students in easy comprehension. Numerical problems included at the e ...
Science - ExamResults.net
... In addition to this, solved and practice problems are included which not only aim at covering the topic but also make students ready to face the competition. The topic-wise classified “question and answer” format of this book helps students in easy comprehension. Numerical problems included at the e ...
... In addition to this, solved and practice problems are included which not only aim at covering the topic but also make students ready to face the competition. The topic-wise classified “question and answer” format of this book helps students in easy comprehension. Numerical problems included at the e ...
CH 9 blackboard
... The number of valence electrons for any main-group element is equal to the group number of its column. (Helium is an exception.) ...
... The number of valence electrons for any main-group element is equal to the group number of its column. (Helium is an exception.) ...
Document
... No more than two electrons can occupy one orbital Electrons occupy the lowest energy orbitals available, the ground state. They enter a higher energy orbital only after the lower orbitals are filled. (Aufbau again.) For the atoms beyond hydrogen, orbital energies vary as “s
... No more than two electrons can occupy one orbital Electrons occupy the lowest energy orbitals available, the ground state. They enter a higher energy orbital only after the lower orbitals are filled. (Aufbau again.) For the atoms beyond hydrogen, orbital energies vary as “s
60. Write the electron configuration for Zn
... 53. Why does atomic radius decrease as you go across a period? Atomic radius decreases because as you move left to right on the periodic table, the elements are all in the same quantum level but have increasing numbers of protons in the nucleus. That means that the farther right the element, the str ...
... 53. Why does atomic radius decrease as you go across a period? Atomic radius decreases because as you move left to right on the periodic table, the elements are all in the same quantum level but have increasing numbers of protons in the nucleus. That means that the farther right the element, the str ...
Spring Semester
... 64. Why does atomic radius decrease as you go across a period? Atomic radius decreases because as you move left to right on the periodic table, the elements are all in the same quantum level but have increasing numbers of protons in the nucleus. That means that the farther right the element, the str ...
... 64. Why does atomic radius decrease as you go across a period? Atomic radius decreases because as you move left to right on the periodic table, the elements are all in the same quantum level but have increasing numbers of protons in the nucleus. That means that the farther right the element, the str ...
Presentation
... b. Removal of an electron from 4s level needs less energy. c. Removal of one electron from 4s orbital of Cr does not change the d configuration. ...
... b. Removal of an electron from 4s level needs less energy. c. Removal of one electron from 4s orbital of Cr does not change the d configuration. ...
Unit 3 - Youngstown City Schools
... square to each group. Students fill the squares with any 25 of the 36 elements in any order. Teacher reads the student-made clues and allows each group of students to mark an X through each element they identify. The first group to correctly complete a vertical, horizontal, or diagonal row is the wi ...
... square to each group. Students fill the squares with any 25 of the 36 elements in any order. Teacher reads the student-made clues and allows each group of students to mark an X through each element they identify. The first group to correctly complete a vertical, horizontal, or diagonal row is the wi ...
1 Electron Configuration and the Periodic Table
... Chapter 1. Electron Configuration and the Periodic Table ...
... Chapter 1. Electron Configuration and the Periodic Table ...
Lectures 12-13 - U of L Class Index
... quantum numbers: n, l, and ml An electron in the atomic orbital is also described by these three quantum numbers as well as a fourth, the spin magnetic quantum number, ms. Electrons behave as if they spin on their axes. Because they are charged, this generates a _________________________ around the ...
... quantum numbers: n, l, and ml An electron in the atomic orbital is also described by these three quantum numbers as well as a fourth, the spin magnetic quantum number, ms. Electrons behave as if they spin on their axes. Because they are charged, this generates a _________________________ around the ...
Lectures 12-13
... quantum numbers: n, l, and ml An electron in the atomic orbital is also described by these three quantum numbers as well as a fourth, the spin magnetic quantum number, ms. Electrons behave as if they spin on their axes. Because they are charged, this generates a _________________________ around the ...
... quantum numbers: n, l, and ml An electron in the atomic orbital is also described by these three quantum numbers as well as a fourth, the spin magnetic quantum number, ms. Electrons behave as if they spin on their axes. Because they are charged, this generates a _________________________ around the ...
Lectures 8-9 - U of L Class Index
... quantum numbers: n, l, and ml An electron in the atomic orbital is also described by these three quantum numbers as well as a fourth, the spin magnetic quantum number, ms. Electrons behave as if they spin on their axes. Because they are charged, this generates a _________________________ around the ...
... quantum numbers: n, l, and ml An electron in the atomic orbital is also described by these three quantum numbers as well as a fourth, the spin magnetic quantum number, ms. Electrons behave as if they spin on their axes. Because they are charged, this generates a _________________________ around the ...
CfE Higher Chemistry Unit 1 Chemical changes and structures
... Iron – used to make ammonia by the Haber Process Platinum – used in manufacture of nitric acid (Ostwald Process) Rhodium and Platinum - in catalytic converters Nickel – to make margarine by hardening vegetable oil Vanadium (V) Oxide – in the contact process, to make sulphuric acid e) Poten ...
... Iron – used to make ammonia by the Haber Process Platinum – used in manufacture of nitric acid (Ostwald Process) Rhodium and Platinum - in catalytic converters Nickel – to make margarine by hardening vegetable oil Vanadium (V) Oxide – in the contact process, to make sulphuric acid e) Poten ...
Chapter 5 Section 2 Electron Configuration and the Periodic Table
... • The number of the highest occupied energy level is 5, so the element is in the fifth period. • There are five electrons in the d sublevel, which means that it is incompletely filled. The d sublevel can hold 10 electrons. Therefore, the element is in the d block. • For d-block elements, the number ...
... • The number of the highest occupied energy level is 5, so the element is in the fifth period. • There are five electrons in the d sublevel, which means that it is incompletely filled. The d sublevel can hold 10 electrons. Therefore, the element is in the d block. • For d-block elements, the number ...
`atoms`.
... • All the elements have only one valence electron which is found in an s-orbital. • The electronic configuration is similar because in all cases there is only one electron outside a noble gas configuration. • Because compound formation involves valence electrons, elements in a group, due to the pre ...
... • All the elements have only one valence electron which is found in an s-orbital. • The electronic configuration is similar because in all cases there is only one electron outside a noble gas configuration. • Because compound formation involves valence electrons, elements in a group, due to the pre ...
Periodic Trends Superblock
... The metallic elements are grouped on the left side of the periodic table. Metals are elements that have a high luster when clean and a high electrical conductivity. They are ductile (can be drawn into wire) and malleable (can be beaten into sheets). Most of the elements are metals. They include the ...
... The metallic elements are grouped on the left side of the periodic table. Metals are elements that have a high luster when clean and a high electrical conductivity. They are ductile (can be drawn into wire) and malleable (can be beaten into sheets). Most of the elements are metals. They include the ...
Problem Solving Drill - Rapid Learning Center
... needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 2. Electronegativity is one of the most important concepts in chemistry, specifically in the description of bonding. It is defined as the tendency of an atom to attract (or pull) electrons to itself. The higher the ...
... needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 2. Electronegativity is one of the most important concepts in chemistry, specifically in the description of bonding. It is defined as the tendency of an atom to attract (or pull) electrons to itself. The higher the ...
Chapter 2 Classification of the Elements
... electrons will half-fill before completely filling the orbitals for a maximum of six electrons. – The d sublevel has five orbitals. The electrons will half-fill before completely filling the orbitals for a maximum of ten electrons. ...
... electrons will half-fill before completely filling the orbitals for a maximum of six electrons. – The d sublevel has five orbitals. The electrons will half-fill before completely filling the orbitals for a maximum of ten electrons. ...
The Bohr Atom The Bohr Atom Electronic Transitions 2.3 Light, Atom
... • Ionization energy - The energy required to remove an electron from an isolated atom • The magnitude of ionization energy correlates with the strength of the attractive force between the nucleus and the outermost electron • The lower the ionization energy, the easier it is to form a cation ionizati ...
... • Ionization energy - The energy required to remove an electron from an isolated atom • The magnitude of ionization energy correlates with the strength of the attractive force between the nucleus and the outermost electron • The lower the ionization energy, the easier it is to form a cation ionizati ...
Lorna Merklinger
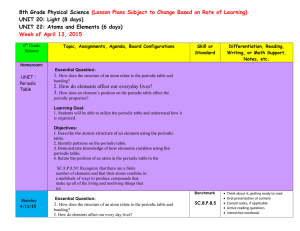
... SC.8.P.8.5#: Recognize that there are a finite number of elements and that their atoms combine in a multitude of ways to produce compounds that make up all of the living and nonliving things that we Benchmark ...
... SC.8.P.8.5#: Recognize that there are a finite number of elements and that their atoms combine in a multitude of ways to produce compounds that make up all of the living and nonliving things that we Benchmark ...
Period 2 element
The period 2 elements are the chemical elements in the second row (or period) of the periodic table. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behavior of the elements as their atomic number increases; a new row is started when chemical behavior begins to repeat, creating columns of elements with similar properties.The second period contains the elements lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine, and neon. This situation can be explained by modern theories of atomic structure. In a quantum mechanical description of atomic structure, this period corresponds to the filling of the 2s and 2p orbitals. Period 2 elements obey the octet rule in that they need eight electrons to complete their valence shell. The maximum number of electrons that these elements can accommodate is ten, two in the 1s orbital, two in the 2s orbital and six in the 2p orbital. All of the elements in the period can form diatomic molecules except beryllium and neon.