Chapter 6 Review
... 1. The elements of the periodic table are sorted into groups according to what? 2. Who was the scientist that first arranged the elements into a periodic table? 3. He arranged the elements according to their what? 4. In the modern periodic table the elements are arranged according to their _________ ...
... 1. The elements of the periodic table are sorted into groups according to what? 2. Who was the scientist that first arranged the elements into a periodic table? 3. He arranged the elements according to their what? 4. In the modern periodic table the elements are arranged according to their _________ ...
Periodic Table Trends - Peoria Public Schools
... Periodic Table Trends Atomic Radii, Ionization Energy, & Electronegativity ...
... Periodic Table Trends Atomic Radii, Ionization Energy, & Electronegativity ...
The Periodic Table
... from the left to the right across a period. Group - reactivity decreases as you go down the group. Why? The farther right and up you go on the periodic table, the higher the electronegativity, resulting in a more vigorous exchange of electron. ...
... from the left to the right across a period. Group - reactivity decreases as you go down the group. Why? The farther right and up you go on the periodic table, the higher the electronegativity, resulting in a more vigorous exchange of electron. ...
The Periodic Table - Anderson High School
... from the left to the right across a period. Group - reactivity decreases as you go down the group. Why? The farther right and up you go on the periodic table, the higher the electronegativity, resulting in a more vigorous exchange of electron. ...
... from the left to the right across a period. Group - reactivity decreases as you go down the group. Why? The farther right and up you go on the periodic table, the higher the electronegativity, resulting in a more vigorous exchange of electron. ...
Document
... X(g) X+(g) +eThis is the number of vibrations per second. The units are Hertz (Hz). E=h. h is the Planck constant and is 6.6.3 x 10-34 J Hz-1.. E is energy in joules (J). The joining together of 2 small nuclei to make one larger one. Approach is at high speed to overcome repulsion between positiv ...
... X(g) X+(g) +eThis is the number of vibrations per second. The units are Hertz (Hz). E=h. h is the Planck constant and is 6.6.3 x 10-34 J Hz-1.. E is energy in joules (J). The joining together of 2 small nuclei to make one larger one. Approach is at high speed to overcome repulsion between positiv ...
Ch.4 Notes Powerpoint Version
... • Recall that groups 1 and 7 are the most reactive metals and nonmetals. • As we move down group 1, the alkali metals become more reactive- this is because of the trend seen in ionization energy! • As we move down group 7, the halogens become less reactive- this is because of the trend seen in elect ...
... • Recall that groups 1 and 7 are the most reactive metals and nonmetals. • As we move down group 1, the alkali metals become more reactive- this is because of the trend seen in ionization energy! • As we move down group 7, the halogens become less reactive- this is because of the trend seen in elect ...
Year 10 Chemistry File
... • We can use the periodic table to help us work out the charges on ions. • Atoms with 1,2 or 3 electrons in their outer shell (groups 1,2 and 3) lose electrons to become positive ions. • Atoms with 5,6 or 7 electrons in their outer shell (groups 15,16 and 17) gain electrons to become negative ions. ...
... • We can use the periodic table to help us work out the charges on ions. • Atoms with 1,2 or 3 electrons in their outer shell (groups 1,2 and 3) lose electrons to become positive ions. • Atoms with 5,6 or 7 electrons in their outer shell (groups 15,16 and 17) gain electrons to become negative ions. ...
AP Chemistry Chapter 7
... • The elements in this family are fluorine, chlorine, bromine, iodine, and astatine. • Halogens have 7 valence electrons, which explains why they are the most active nonmetals. They are never found free in nature. • Halogen atoms only need to gain 1 electron to fill their outermost energy level. • T ...
... • The elements in this family are fluorine, chlorine, bromine, iodine, and astatine. • Halogens have 7 valence electrons, which explains why they are the most active nonmetals. They are never found free in nature. • Halogen atoms only need to gain 1 electron to fill their outermost energy level. • T ...
CH04_Tro_LectureNotes_081 - Tutor
... Although metals are usually solid, nonmetals may be solids, liquids, or gases. Unlike metals, they do not conduct electricity well. They have a very wide variety of physical characteristics such as color and form. On an atomic level, nonmetals tend to gain electrons in chemical reactions. When a non ...
... Although metals are usually solid, nonmetals may be solids, liquids, or gases. Unlike metals, they do not conduct electricity well. They have a very wide variety of physical characteristics such as color and form. On an atomic level, nonmetals tend to gain electrons in chemical reactions. When a non ...
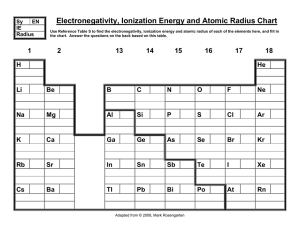
Electronegativity, Ionization Energy and Atomic Radius Chart
... - They are only found bonded to other elements in compounds in their natural state. - They can be separated from those compounds using electricity (electrolytic decomposition). - They combine with hydrogen, to form strong acids (like hydrochloric acid, HCl). Fluorine and chlorine are extremely corro ...
... - They are only found bonded to other elements in compounds in their natural state. - They can be separated from those compounds using electricity (electrolytic decomposition). - They combine with hydrogen, to form strong acids (like hydrochloric acid, HCl). Fluorine and chlorine are extremely corro ...
the atomic theory
... The number of protons in an atom determines its identity, and is called atomic number (Z). In a neutral atom, the number of protons (+) are equal to the number of electrons (–). Almost all the mass of the atom rests in the nucleus. The number of protons and neutrons in an atom is called the ...
... The number of protons in an atom determines its identity, and is called atomic number (Z). In a neutral atom, the number of protons (+) are equal to the number of electrons (–). Almost all the mass of the atom rests in the nucleus. The number of protons and neutrons in an atom is called the ...
Unit 4 Pack
... Why is Si bigger than S (explain why – do not just write because it is further left!) Why is Se bigger than S? (explain why) Why is Cu2+ smaller than Cu? Identify the two ions that are most important in the human body. Where are they found? What is their function? Given Al, I and F a. Place them in ...
... Why is Si bigger than S (explain why – do not just write because it is further left!) Why is Se bigger than S? (explain why) Why is Cu2+ smaller than Cu? Identify the two ions that are most important in the human body. Where are they found? What is their function? Given Al, I and F a. Place them in ...
Science 2nd prep 1st term 1st lesson Many attempts are made by
... Science 2nd prep 1st term 1st lesson - He arranged elements in an ascending order according to their atomic numbers. - he added the inert gases in the (0) zero group. - He classified the elements of each group into two subgroups (A&B) as they differ in their ...
... Science 2nd prep 1st term 1st lesson - He arranged elements in an ascending order according to their atomic numbers. - he added the inert gases in the (0) zero group. - He classified the elements of each group into two subgroups (A&B) as they differ in their ...
Nov 9 Agenda
... 2) The size of atoms in the same period, as the relative positive charge increases due to Z increasing, the size of the atoms decreases. ...
... 2) The size of atoms in the same period, as the relative positive charge increases due to Z increasing, the size of the atoms decreases. ...
Periodic Table Funsheet
... 9. Where is the highest electronegativity found? _________________________________________________ 10. Where is the lowest electronegativity found? __________________________________________________ 11. Elements of Group 1 are called _____________________________________________________________. 12. ...
... 9. Where is the highest electronegativity found? _________________________________________________ 10. Where is the lowest electronegativity found? __________________________________________________ 11. Elements of Group 1 are called _____________________________________________________________. 12. ...
Chemistry_Review_Packet - AP-Biology
... 2. Name the six most abundant elements found in the human body. The universe is composed of about 92 naturally occurring elements. In nature, most of these elements are found in combination with one or more other elements. These combinations of elements are called compounds. Twenty-five of the known ...
... 2. Name the six most abundant elements found in the human body. The universe is composed of about 92 naturally occurring elements. In nature, most of these elements are found in combination with one or more other elements. These combinations of elements are called compounds. Twenty-five of the known ...
Power point notes - Social Circle City Schools
... Because they do not readily combine with other elements to form compounds, the noble gases are called inert. The family of noble gases includes helium, neon, argon, krypton, xenon, and radon. All the noble gases are found in small amounts in the earth's ...
... Because they do not readily combine with other elements to form compounds, the noble gases are called inert. The family of noble gases includes helium, neon, argon, krypton, xenon, and radon. All the noble gases are found in small amounts in the earth's ...
C Carbon Cu Copper
... electrons in their very outermost energy level (This is called the rule of octet.) Atoms bond until this level is complete. Atoms with few valence electrons lose them during bonding. Atoms with 6, 7, or 8 valence electrons gain electrons during bonding. ...
... electrons in their very outermost energy level (This is called the rule of octet.) Atoms bond until this level is complete. Atoms with few valence electrons lose them during bonding. Atoms with 6, 7, or 8 valence electrons gain electrons during bonding. ...
PeriodicTrends
... • SC4a. Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements. ...
... • SC4a. Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements. ...
Families on the Periodic Table
... He noticed that every _______ element had ___________________ chemical and physical properties; their properties were repeating. This became known as __________________________________________________ o Li Be B C N O F o Na Mg Al Si P S Cl ...
... He noticed that every _______ element had ___________________ chemical and physical properties; their properties were repeating. This became known as __________________________________________________ o Li Be B C N O F o Na Mg Al Si P S Cl ...
Unit 4 Periodic Table Packet 2016-2017
... He noticed that every _______ element had ___________________ chemical and physical properties; their properties were repeating. This became known as __________________________________________________ o Li Be B C N O F o Na Mg Al Si P S Cl ...
... He noticed that every _______ element had ___________________ chemical and physical properties; their properties were repeating. This became known as __________________________________________________ o Li Be B C N O F o Na Mg Al Si P S Cl ...
Chemistry Chapter 5 The Periodic Law
... and their meaning. Know the definitions of ionization energy, electronegativity, and atomic radius and their trends both across a period and down a group. Know the definition of valence electrons & how to find their number in s & p groups. ...
... and their meaning. Know the definitions of ionization energy, electronegativity, and atomic radius and their trends both across a period and down a group. Know the definition of valence electrons & how to find their number in s & p groups. ...
History of Periodic Table
... their atomic mass Wanted to keep elements with similar properties in the same columns Left gaps in his early tables; predicted elements that had not been discovered would fill in those gaps ...
... their atomic mass Wanted to keep elements with similar properties in the same columns Left gaps in his early tables; predicted elements that had not been discovered would fill in those gaps ...
columns
... due to the addition of energy levels, electrons further away from the nucleus have less attraction to the positive nucleus. Atoms and ions get bigger as you go down the columns because the shielding effect outweighs the effects of the nuclear charge, so the attraction between the nucleus and electro ...
... due to the addition of energy levels, electrons further away from the nucleus have less attraction to the positive nucleus. Atoms and ions get bigger as you go down the columns because the shielding effect outweighs the effects of the nuclear charge, so the attraction between the nucleus and electro ...
Period 3 element
A period 3 element is one of the chemical elements in the third row (or period) of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when the periodic table skips a row and a chemical behaviour begins to repeat, meaning that elements with similar behavior fall into the same vertical columns. The third period contains eight elements: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, and argon. The first two, sodium and magnesium, are members of the s-block of the periodic table, while the others are members of the p-block. Note that there is a 3d orbital, but it is not filled until Period 4, such giving the period table its characteristic shape of ""two rows at a time"". All of the period 3 elements occur in nature and have at least one stable isotope.